 |
 |
 |
| |
Plasma proteomic signatures capture unique cardiovascular risk phenotypes among persons living with and without HIV in the United States:
Plasma Proteome [biomarkers] Offers Insights Beyond Traditional CVD Risk Factors
|
| |
| |
International Workshop on HIV & Aging 2023, October 26-27, Washington, DC
Reported by Mark Mascolini for NATAP and Academic Medical Education
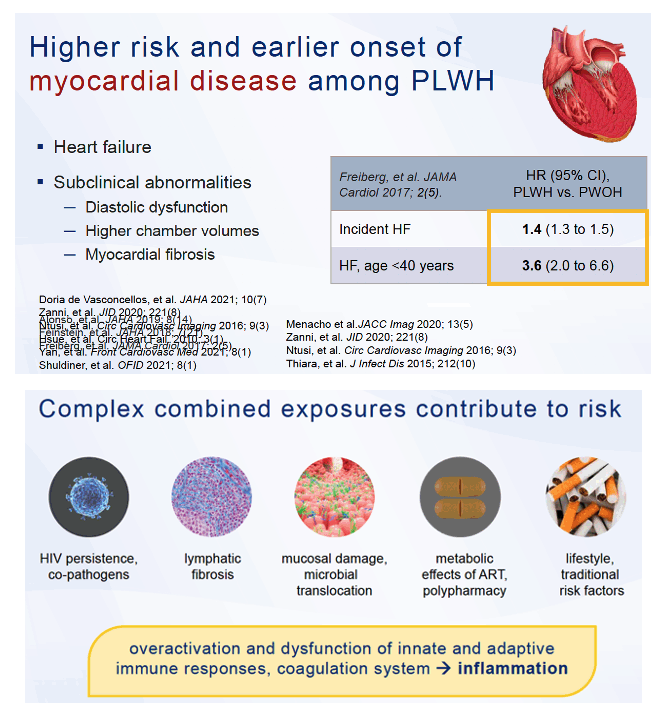
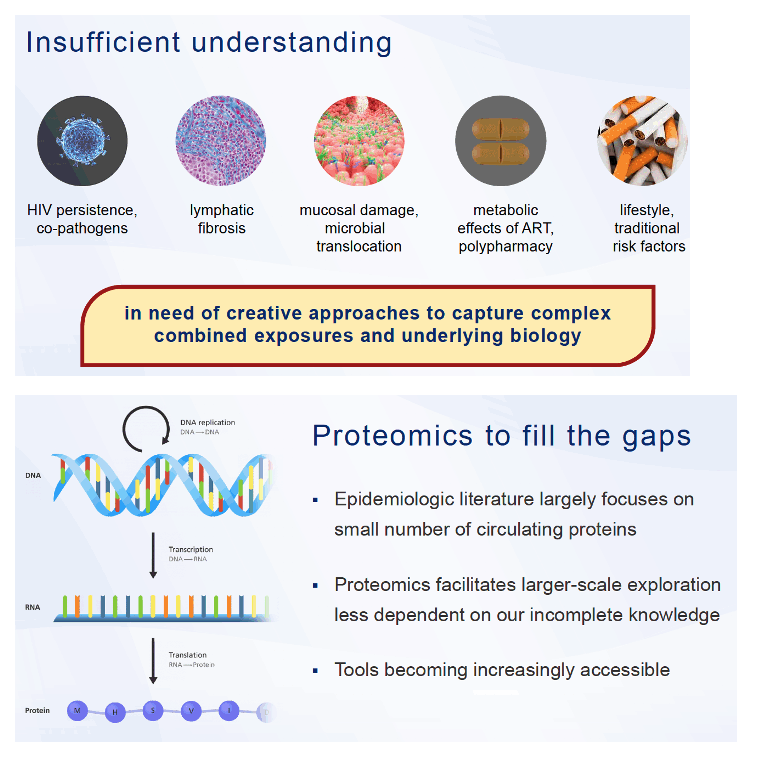
Plasma proteomics-a high-throughput analysis of thousands of biomarkers in plasma-may offer a broader yet sharper way to assess cardiovascular disease (CVD) risk factors in people with or without HIV infection [1,2]. A study by researchers at Johns Hopkins University, the National Institute on Aging (NIA), and other centers in 352 HIV-positive and -negative people explored the plasma proteome to define three distinct CVD subpopulations that differed in nontraditional and HIV-related CVD risk factors [1].
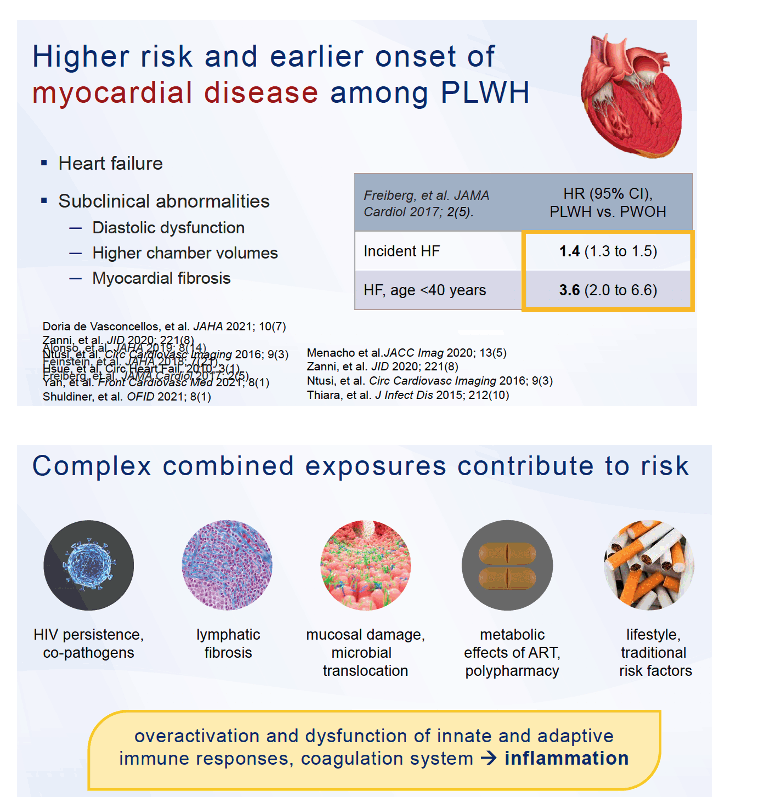
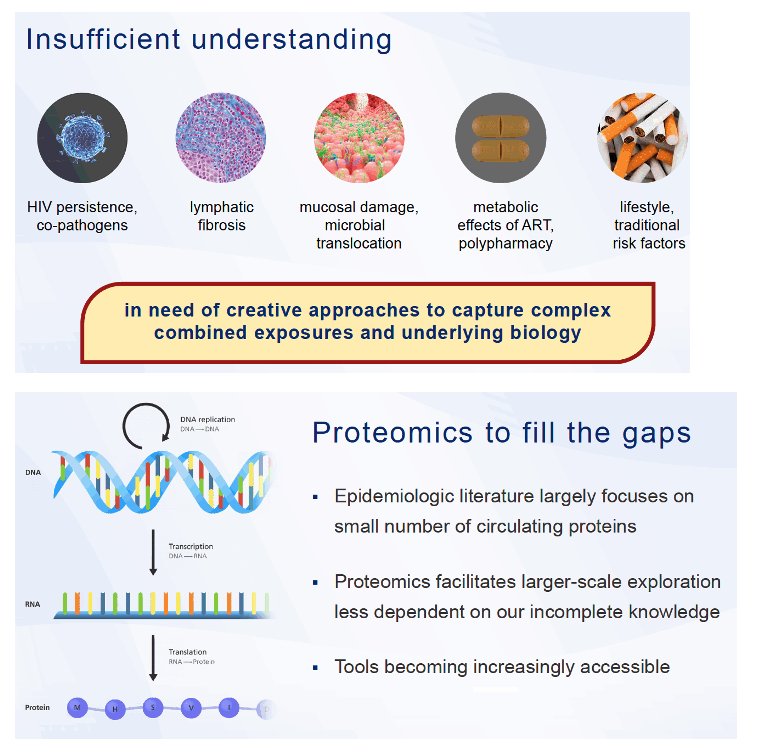
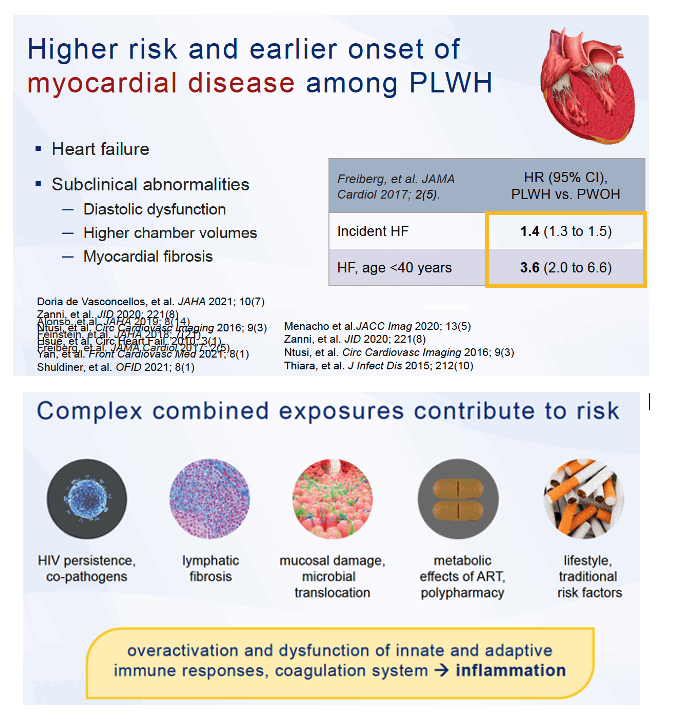
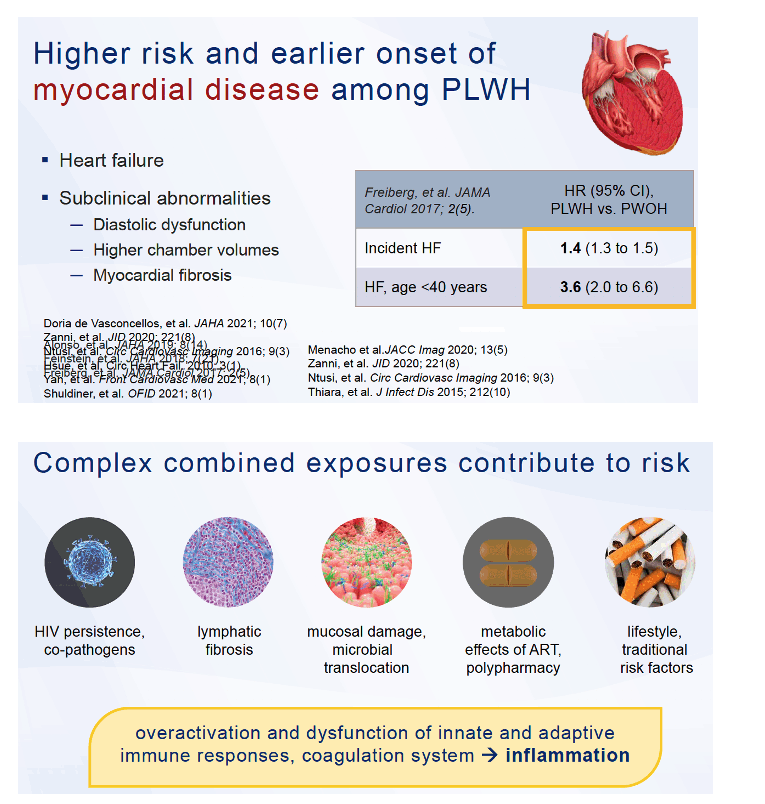
Analyzing biomarkers to diagnose and monitor CVD has long been a standard tool of clinical medicine. Recent developments in high-throughput plasma proteomics offer an opportunity to identify biomarkers that will help in managing heart diseases increasing in frequency yet lacking biomarkers, like heart failure and abdominal aortic aneurysm [3], to enhance biomarker understanding of cardiovascular diseases with some already characterized biomarkers, and to define differences in protein clusters specific to certain populations, like people with HIV infection.
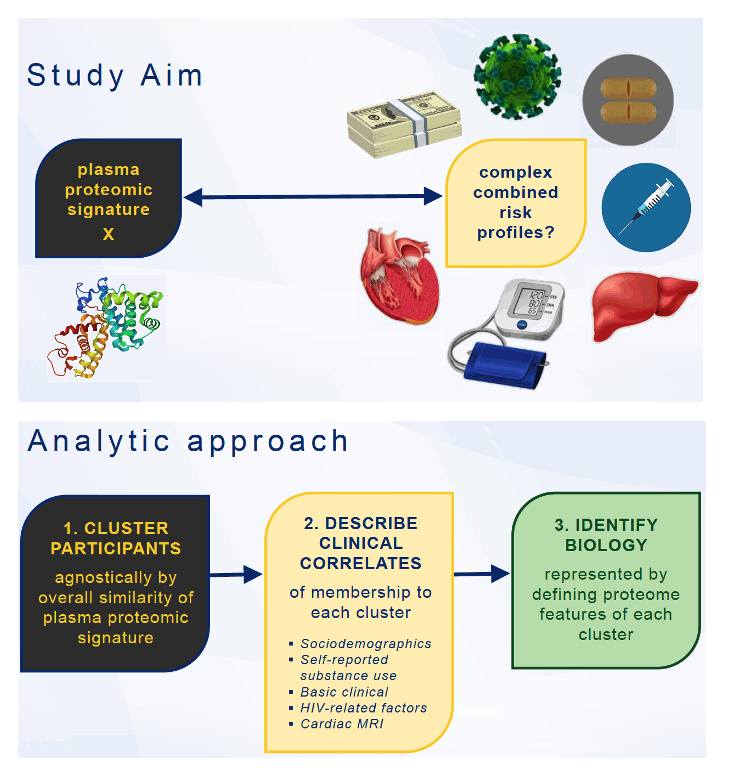
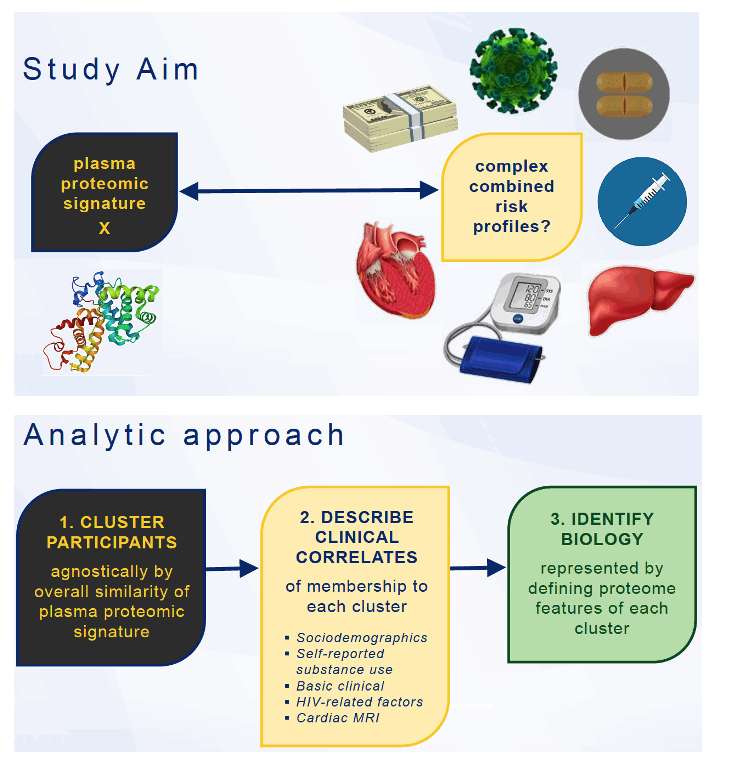
The Hopkins/NIA group noted that subclinical structural and functional abnormalities in the cardiovascular system can foretell clinical diseases such as heart failure. Because biological mechanisms driving development of such diseases remain poorly understood and probably reflect complex risk factor interactions, these researchers set out to discern subpopulations of people with or without HIV with distinct risk profiles that plasma proteomics could identify.
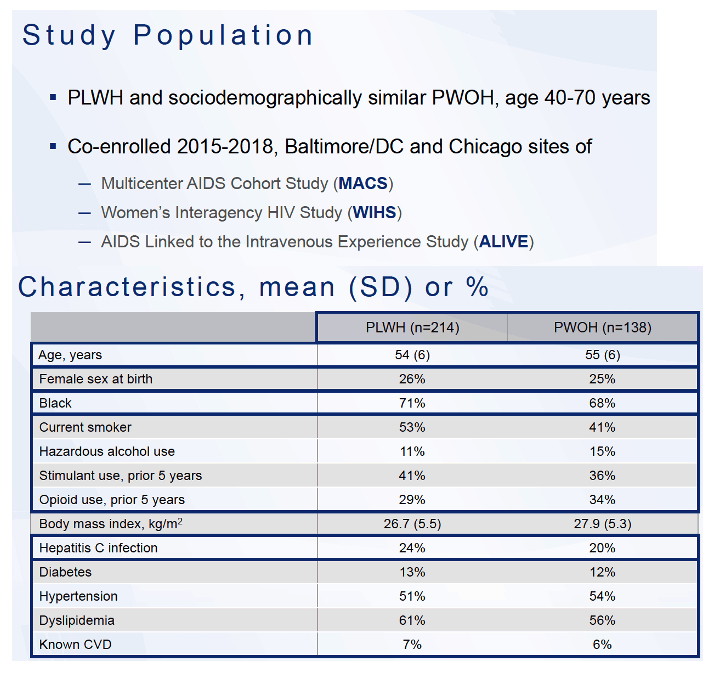
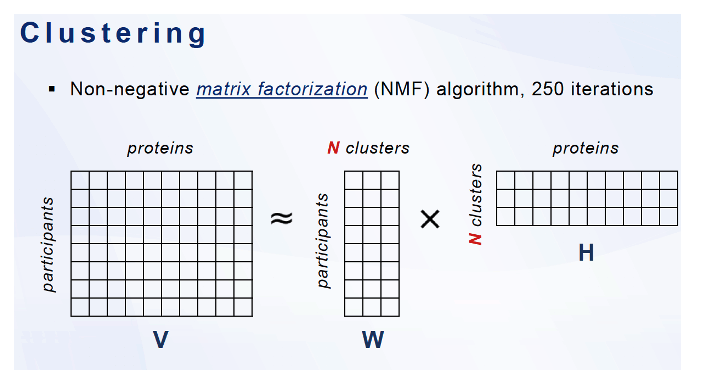
The researchers planned a 3-step analytic approach: (1) to cluster participants by the overall similarity of plasma proteome signature, (2) to describe clinical correlates of each proteome cluster, and (3) to identify the biology represented by defining proteomic features of each cluster. They collected plasma for proteomic analysis at the same time they performed cardiac magnetic resonance imaging of study participants in Baltimore, Washington, DC, and Chicago already enrolled in the Subclinical Myocardial Abnormalities in HIV (SMASH) study.
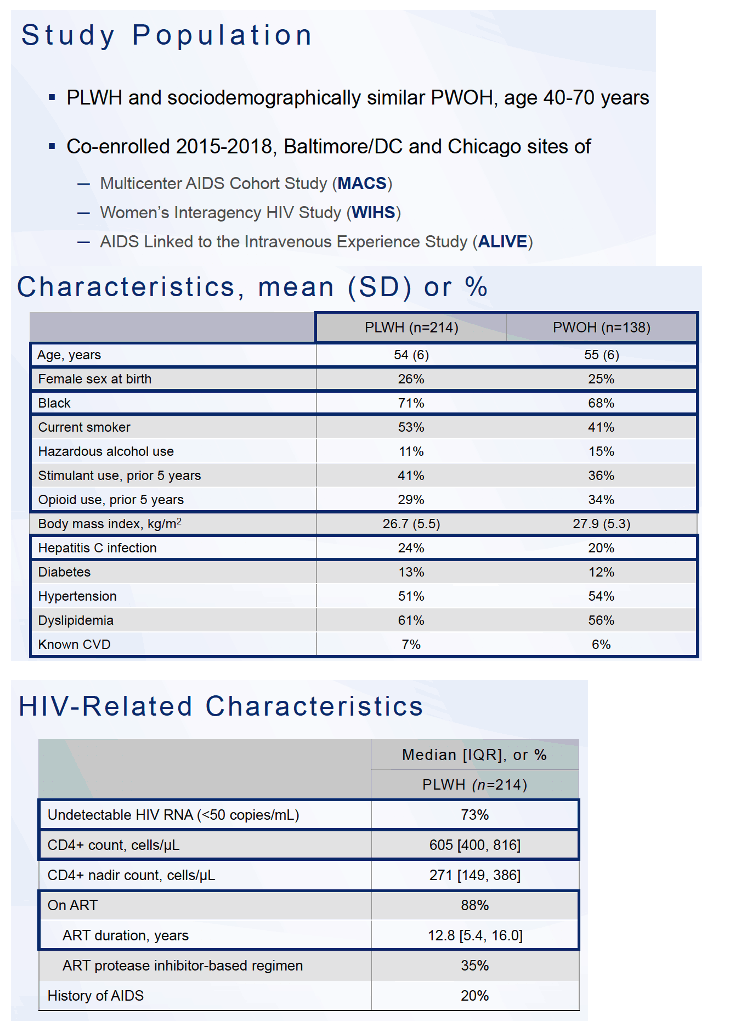
The analysis involved 214 people with HIV and 138 without HIV. The HIV group did not differ from the HIV-negative group substantially in age (average 54 and 55), proportion female at birth (26% and 25%), proportion of blacks (71% and 68%), body mass index, or proportions with hepatitis C infection, diabetes, hypertension, dyslipidemia, or known CVD (7% and 6%). The HIV and non-HIV groups did differ in proportions who currently smoked (53% vs 41%) or had hazardous alcohol use (11% vs 15%), or opioid use in the past 5 years (29% vs 34%). In the group with HIV infection, 88% took antiretrovirals for a median 12.8 years, 73% had an undetectable viral load, and median CD4 count stood at 605.
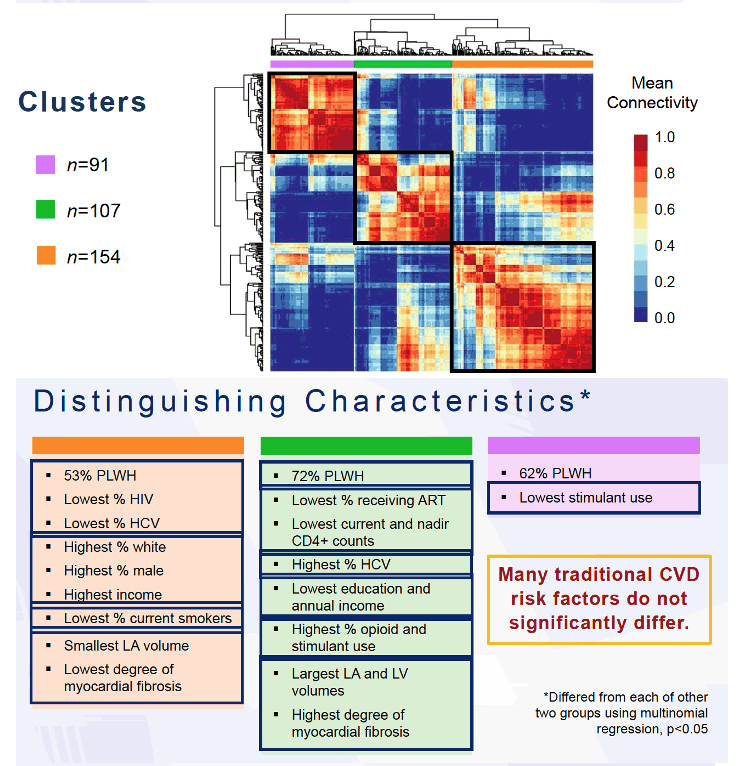
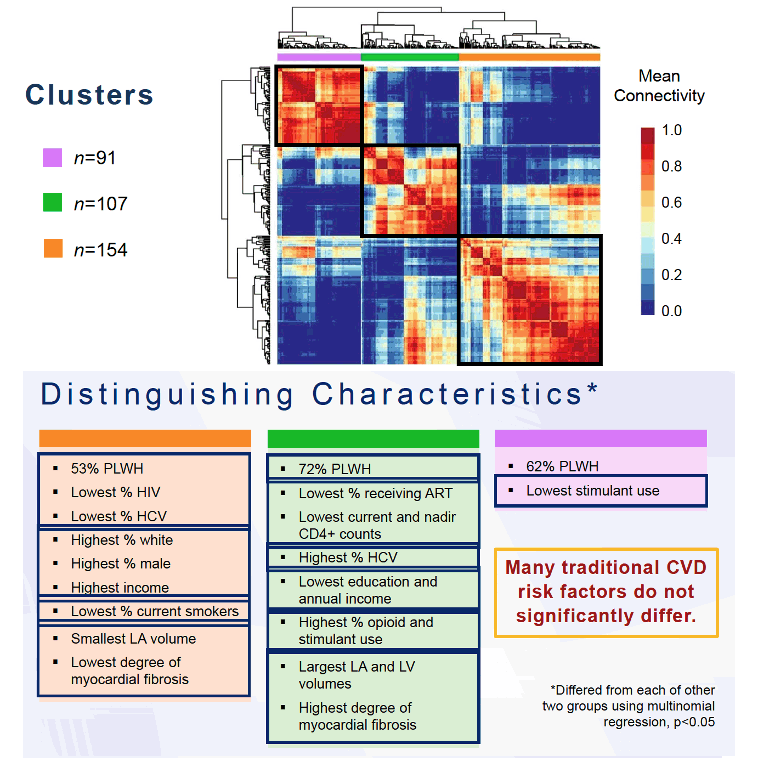
The study group used the Olink Explore 3072 plasma proteomics platform because of its extensive validation, high specificity, and high reproducibility. Proteomics identified 3 clusters: Cluster 1 had 154 members (53% with HIV), cluster 2 had 107 (72% with HIV), and cluster 3 had 154 (62% with HIV). Cluster 2, with the highest proportion of HIV-positive people, had a significantly higher prevalence of subclinical cardiovascular abnormalities-myocardial fibrosis/inflammation and both left atrial and left ventricular enlargement.
Cluster 2 also had the lowest percentage of people with HIV taking antiretrovirals, the lowest current and nadir CD4 counts, the least education and lowest annual income, the highest proportion with HCV infection, the highest proportion of opioid and stimulant users, the largest left atrial and ventricular volumes, and the highest degree of myocardial fibrosis.
Cluster 1 had the lowest proportion with HIV (53%) or HCV, the highest proportion of whites and males, the highest income, the lowest proportion of current smokers, the smallest left atrial volume, and the lowest degree of myocardial fibrosis. Cluster 3 had 62% with HIV and the least stimulant use.
These characteristics in each cluster differed significantly from the other two clusters in multinomial regression (P < 0.05). Reviewing these distinguishing traits, the researchers pointed out that the 3 clusters did not differ substantially in many traditional CVD risk factors.
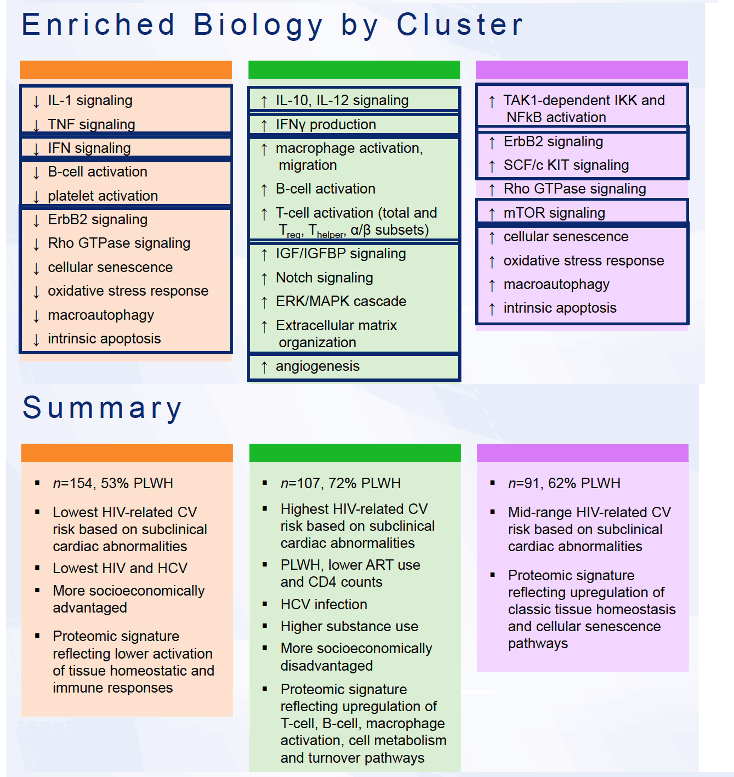
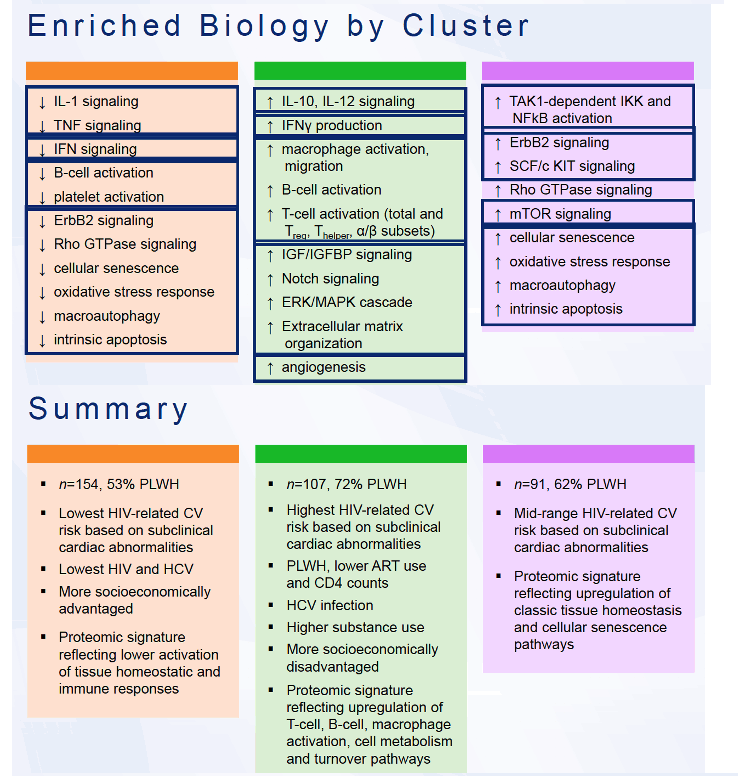
The three clusters differed in notable markers of inflammation and activation and in other biomarkers. Cluster 2, for example, had increased IL-10 and IL-12 signaling, increased interferon-gamma production, increased macrophage activation and migration, and increased B- and T-cell activation. Cluster 1, in contrast, had decreased IL-1, TNF, and interferon signaling, decreased B-cell and platelet activation, decreased cellular senescence, and decreased oxidative stress response. The cluster 1 proteomic signature reflected lower activation of tissue homeostatic and immune responses, while the cluster 3 proteomic signature indicated upregulation of classic tissue homeostasis and cellular senescence pathways. In contrast, with its high proportion of HIV-positive people, cluster 2 had a proteomic signature reflecting upregulation of T- and B-cell activation, macrophage activation, and cell metabolism and turnover pathways.

The researchers offered two conclusions:
"We've identified subpopulations of people living with HIV and people without HIV that may reflect systemic biology underlying interactions of exposures to risk factors for both HIV and CVD, which are manifest by subclinical abnormalities in cardiac structure.
"The proteome may objectively capture complex combined exposures beyond traditional cardiovascular risk factors that could potentially be leveraged toward improved risk prediction and targeted interventions."
link to oral session
https://www.youtube.com/watch?v=i2P7Qt2SjYk
References
1. Peterson T, Hahn V, Moaddel R, et al. Plasma proteomic signatures capture unique cardiovascular risk phenotypes among persons living with and without HIV in the United States. International Workshop on HIV & Aging 2023, October 26-27, Washington, DC. Abstract 18.
2. Thomas L. Plasma proteomics. News Medical. Last updated February 23, 2023. https://www.news-medical.net/health/Plasma-Proteomics.aspx
3. Palstrom NB, Matthiesen R, Rasmussen LM, Beck HC. Recent developments in clinical plasma proteomics-applied to cardiovascular research. Biomedicines. 2022;10:162. doi: 10.3390/biomedicines10010162. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8773619/







|
| |
|
 |
 |
|
|