 |
 |
 |
| |
Gilead Demonstrates Transformative Potential
Impact Of Antiviral Innovation at CROI 2023
|
| |
| |
- Clinical Data Include Analyses of Novel, Investigational Long-acting Therapies and Combination Cure Strategy Research with the Potential to Help Address the Diverse and Unmet Needs of People with HIV -
- Real-World Evidence Evaluating the Effectiveness of Veklury in Broad Range of Hospitalized Patient Populations Being Presented, Including Those at High Risk for COVID-19 Disease Progression -
Feb 14 2023
FOSTER CITY, Calif.--(BUSINESS WIRE)-- Gilead Sciences, Inc. (Nasdaq: GILD) today announced the upcoming presentation of new clinical and real-world data from the company's HIV, COVID-19 and viral hepatitis research and development programs at the 30th Conference on Retroviruses and Opportunistic Infections (CROI) from February 19-22. Seventy abstracts, including late-breaking data, demonstrate Gilead's continued expertise and leadership in virology with a commitment to advance the next wave of scientific discovery to treat, prevent, cure and help eradicate viral diseases.
"Positioning people at the center of the research and development process is helping us to fuel the next wave of innovation in virology," said Frank Duff, MD, Senior Vice President, Virology Therapeutic Area Head, Gilead Sciences. "Our scientific advances are grounded in collaboration with community and research partners around the world and highlight the importance of developing innovative solutions for the unmet and evolving needs of those most affected by viral diseases. The data selected for presentation at CROI include real-world evidence in the treatment of COVID-19, the investigational evaluation of existing therapies for HIV/hepatitis B co-infection, and new HIV treatment, prevention, and cure data evaluating long-acting options as well as novel combination therapies."
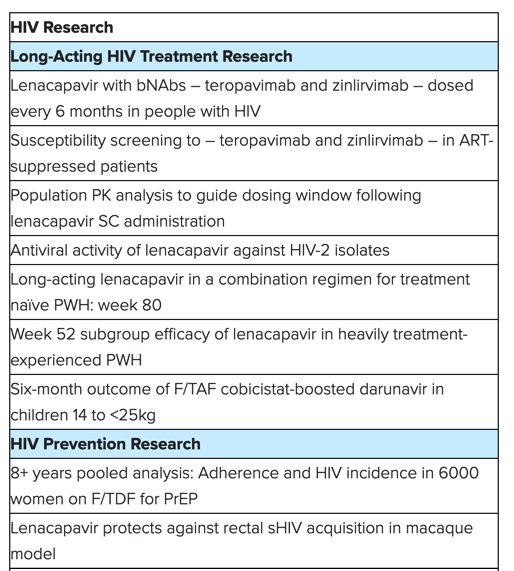
HIV Research
Gilead is committed to continuous scientific discovery to provide solutions for the evolving needs of people affected by HIV around the world, with the goal of ending the HIV epidemic for everyone, everywhere. At CROI 2023, Gilead will share new findings on HIV treatment and prevention strategies, as well as the latest updates from the company's continued pursuit of an HIV cure.
HIV treatment research and development data include proof-of-concept Phase 1b data on the safety and efficacy profile of lenacapavir in combination with investigational broadly neutralizing antibodies (bNAbs) - teropavimab and zinlirvimab - dosed twice-yearly in virologically suppressed adults for the treatment of HIV.
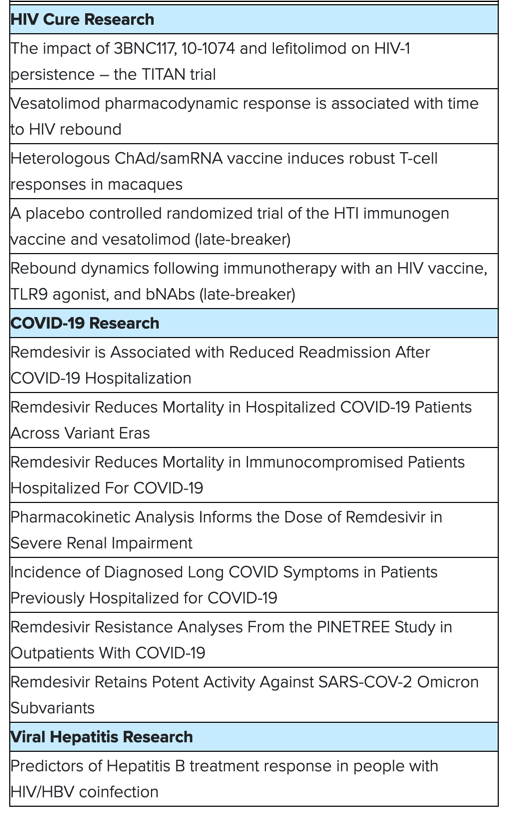
Insights from Gilead's cure research program include new data from multiple collaborative studies evaluating novel combination therapies aimed at targeting the HIV viral reservoir and enhancing the immune response to maintain virologic control in the absence of antiretroviral therapy. The findings support the continued investigational evaluation of broadly neutralizing antibodies, vaccine candidates, and toll-like receptor agonists.
Additionally, Gilead will present data from a pre-clinical in vivo assessment of a single subcutaneous administration of lenacapavir for pre-exposure prophylaxis (PrEP). As part of the company's HIV prevention research program, Gilead will also present the results of a multinational pooled analysis of real-world adherence to PrEP and HIV incidence in cisgender women.
COVID-19 Research
Gilead will present data that reinforce the critical role Veklury® (remdesivir) plays as the antiviral standard of care for the treatment of hospitalized patients with COVID-19. Real-world insights on Veklury's use will provide important, up-to-date evidence from large, hospitalized populations - including patients with immunocompromised conditions - across different periods of the pandemic and different variants of concern. A separate study will evaluate the likelihood of all cause readmission among hospitalized patients who received Veklury compared to those who did not.
Additionally, Gilead will present a new in vitro analysis that evaluates the antiviral activity of Veklury against common variants of concern, and data assessing the resistance profile of Veklury and other antiviral compounds.
Viral Hepatitis Research
Gilead will present new data from the ALLIANCE trial, which is an ongoing Phase 3 trial evaluating Biktarvy®(bictegravir 50 mg/emtricitabine 200 mg/tenofovir alafenamide 25 mg tablets, B/F/TAF) versus dolutegravir (50 mg, DTG) + emtricitabine 200 mg/tenofovir disoproxil fumarate 300 mg, F/TDF, DTG+F/TDF, in adults with HIV-1/HBV co-infection who are initiating treatment. The sub-analysis of Week 48 results complements the initial data presented at the 24th International AIDS Conference (AIDS 2022), with a focus on the HBV outcomes in this coinfected population.
Accepted abstracts at the 30th Conference on Retroviruses and Opportunistic Infections include:
For more information, including a complete list of abstracts and their corresponding oral and poster sessions, please visit https://www.croiconference.org.
Teropavimab, zinlirvimab, lefitolimod, vesatolimod, GS-5423, GS-2872, VRC07-523LS are investigational compounds and are not approved by the U.S. Food and Drug Administration or any other regulatory authority for any use. Their safety and efficacy are unknown.
The use of lenacapavir for HIV prevention is investigational and the safety and efficacy of lenacapavir for this use have not been established. Gilead is studying the safety and efficacy of lenacapavir for HIV prevention in multiple ongoing clinical studies.
The use of Biktarvy in individuals with HIV-1/HBV coinfection is investigational, and the safety and efficacy of Biktarvy for this use have not been established.
Please see below for the U.S. Indication and Important Safety Information for Veklury and Sunlenca. Please also see below for U.S. Indication and Important Safety Information, including Boxed Warning, for Biktarvy.
There is currently no cure for HIV or AIDS.
About Biktarvy
Biktarvy is a complete HIV treatment that combines three powerful medicines to form the smallest 3-drug, integrase strand transfer inhibitor (INSTI)-based single-tablet regimen (STR) available, offering simple once-daily dosing with or without food, with a limited drug interaction potential and a high barrier to resistance. Biktarvy combines the novel, unboosted INSTI bictegravir, with the Descovy® (emtricitabine 200 mg/tenofovir alafenamide 25 mg tablets, F/TAF) backbone. Biktarvy is a complete STR and should not be taken with other HIV medicines.
About Sunlenca ®
Sunlenca (300 mg tablet and 463.5 mg/1.5 mL injection) [(lenacapavir)] is a first-in-class, long-acting HIV capsid inhibitor approved in the United States, the United Kingdom, Canada and the European Union, for the treatment of HIV infection, in combination with other antiretroviral(s), in people with multi-drug resistant HIV who are heavily treatment-experienced. Sunlenca tablets are approved for oral loading during initiation of Sunlenca treatment, prior to or at the time of the first long-acting lenacapavir injection depending on initiation option. Sunlenca is the only HIV treatment option administered twice-yearly. The multi-stage mechanism of action of Sunlenca's active pharmaceutical agent, lenacapavir, is distinguishable from other currently approved classes of antiviral agents. While most antivirals act on just one stage of viral replication, Sunlenca is designed to inhibit HIV at multiple stages of its lifecycle and has no known cross resistance exhibited in vitro to other existing drug classes.
Lenacapavir is an investigational agent being evaluated as a long-acting option in multiple ongoing and planned early and late-stage clinical studies in Gilead's prevention and treatment research program. Lenacapavir is being developed as a foundation for future HIV therapies with the goal of offering both long-acting oral and injectable options with several dosing frequencies, in combination or as monotherapy, that help address individual patient needs and preferences.
U.S. Indication for Biktarvy
Biktarvy is indicated as a complete regimen for the treatment of human immunodeficiency virus type 1 (HIV-1) infection in adults and pediatric patients weighing at least 14 kg who have no antiretroviral treatment history or to replace the current antiretroviral regimen in those who are virologically-suppressed (HIV-1 RNA less than 50 copies per mL) on a stable antiretroviral regimen with no history of treatment failure and no known substitutions associated with resistance to the individual components of Biktarvy.
Link to full press release https://www.gilead.com/news-and-press/press-room/press-releases/2023/2/gilead-demonstrates-transformative-potential-impact-of-antiviral-innovation-at-croi-2023
|
| |
|
 |
 |
|
|