 |
 |
 |
| |
Effects of eplerenone on myocardial perfusion and function: the Miracle HIV study: May Stall Worsening Subclinical Heart Disease With HIV
|
| |
| |
30th CROI, Conference on Retroviruses and Opportunistic Infections, February 19-22, 2023, Seattle
Mark Mascolini
Eplerenone, an aldosterone antagonist licensed to treat hypertension and congestive heart failure after myocardial infarction, may prevent worsening of subclinical myocardial disease in people with HIV, according to results of 33-person randomized, placebo-controlled trial. Eplerenone also significantly improved coronary flow reserve in people with impaired total flow capacity.
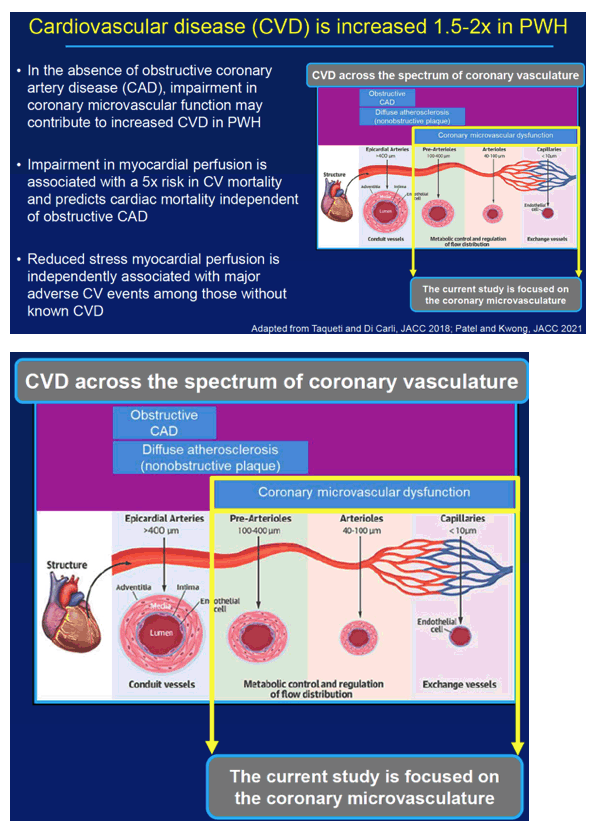
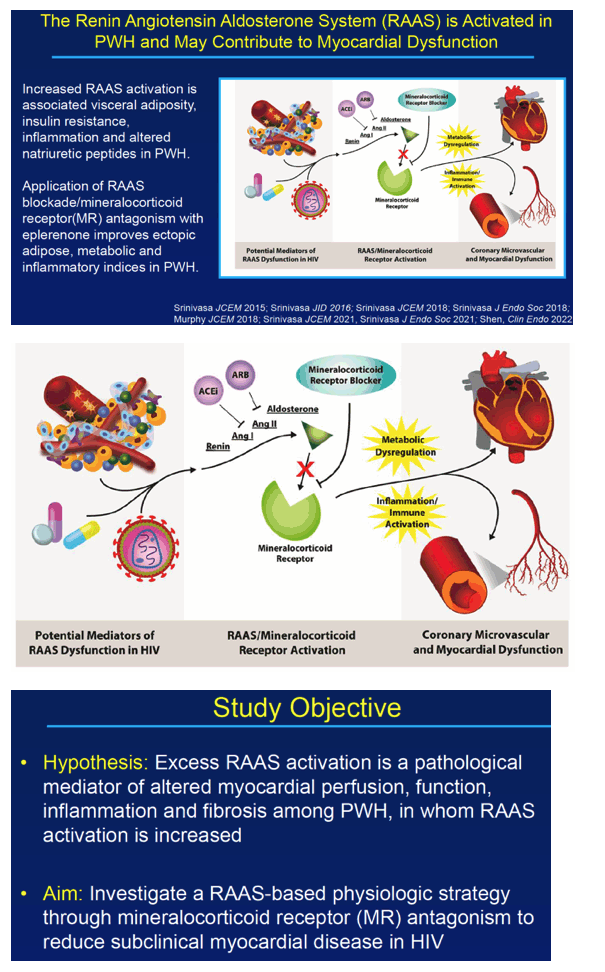
Researchers at Massachusetts General Hospital and Harvard Medical School reminded colleagues that people with HIV have a 1.5- to 2-fold higher cardiovascular disease risk than HIV-negative people. They noted that impaired myocardial perfusion boosts cardiovascular mortality risk 5-fold and predicts mortality independently of obstructive coronary artery disease. The renin angiotensin aldosterone system (RAAS) becomes activated in people with HIV and may contribute to weakened heart function.
The Mass General/Harvard investigators hypothesized that excess RAAS activation mediates altered myocardial perfusion, function, inflammation, and fibrosis in people with HIV. They conducted this study to explore a RAAS-based strategy via mineralocorticoid receptor (MR) antagonism to ease subclinical myocardial disease in people with HIV. Eplerenone blocks aldosterone binding at the MR.
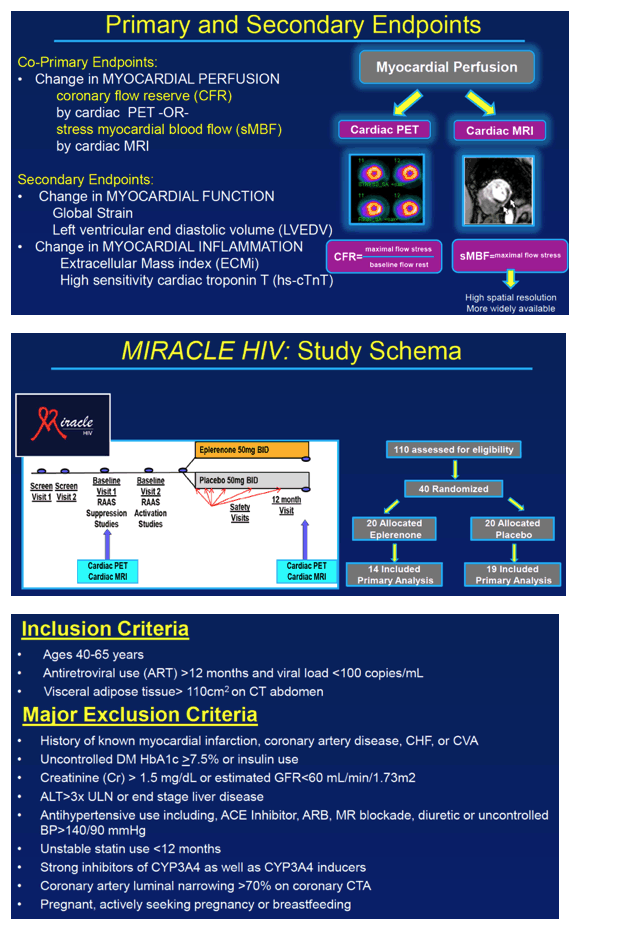
The trial's primary endpoints were to measure the impact of eplerenone on myocardial perfusion gauged as (1) coronary flow reserve (by cardiac PET scan) or (2) stress myocardial blood flow (by cardiac MRI). The study included HIV-positive people 40 to 65 years old taking antiretroviral therapy for more than 12 months with a viral load below 100 copies and with visceral adipose tissue greater than 110 cm2 on abdominal CT. Researchers excluded people with a history of myocardial infarction, coronary artery disease, congestive heart failure, or cerebrovascular disease. Participants could not have uncontrolled diabetes or coronary artery luminal narrowing greater than 70%.
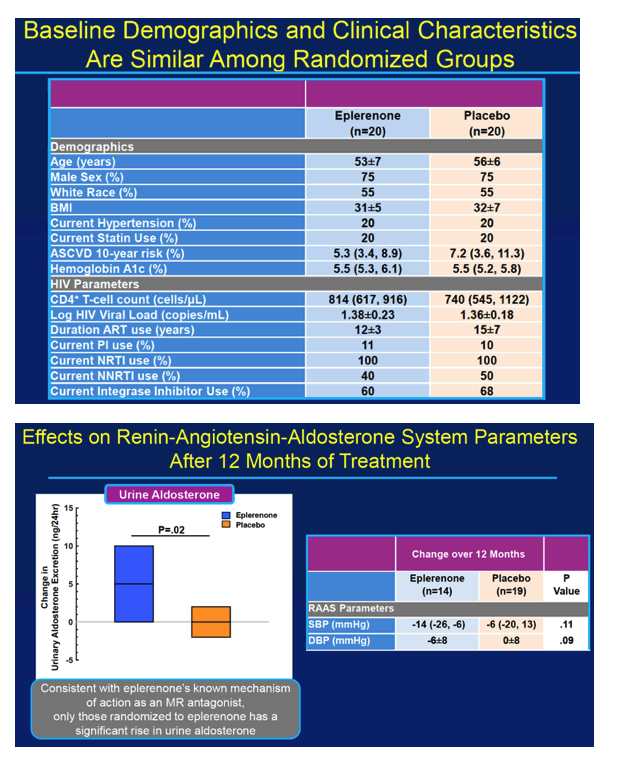
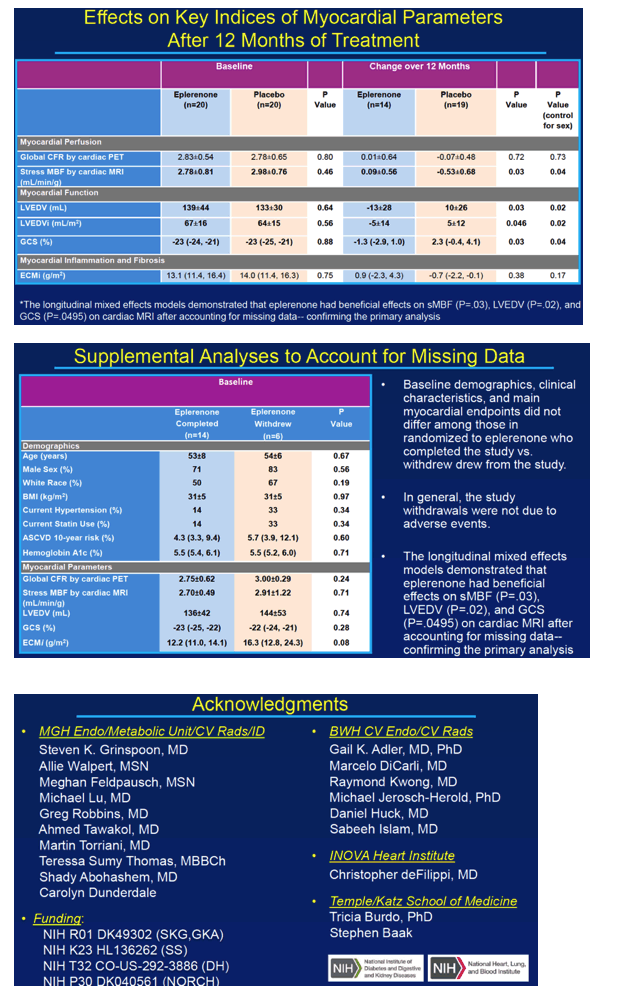
Forty study participants got randomized evenly to eplerenone or placebo. The primary analysis included 14 people assigned to eplerenone and 19 assigned to placebo. Among the 40 participants randomized to eplerenone or placebo, age averaged 53 and 56, proportions of men 75% in both groups, and proportions of whites 55% in both groups. In each study arm, 20% had hypertension and 20% used a statin. Ten-year risk of atherosclerotic cardiovascular disease was 5.3% in the eplerenone arm and 7.2% in the placebo arm.
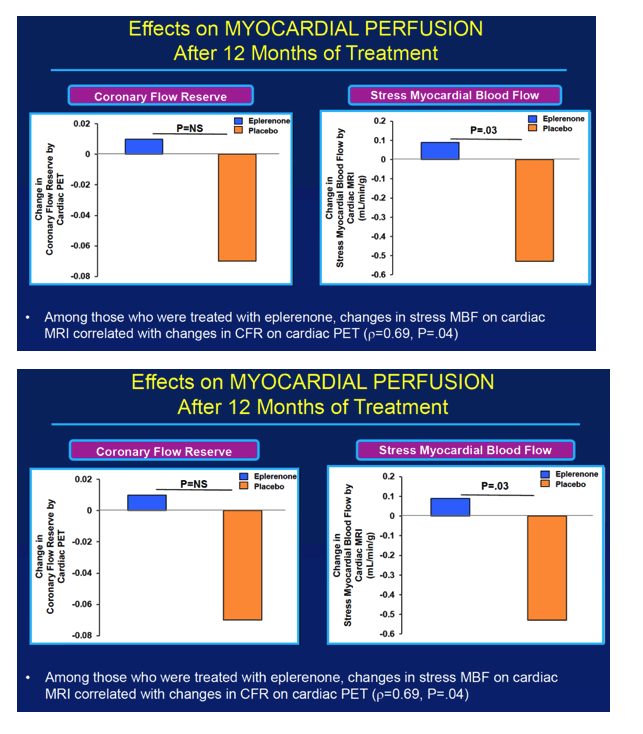
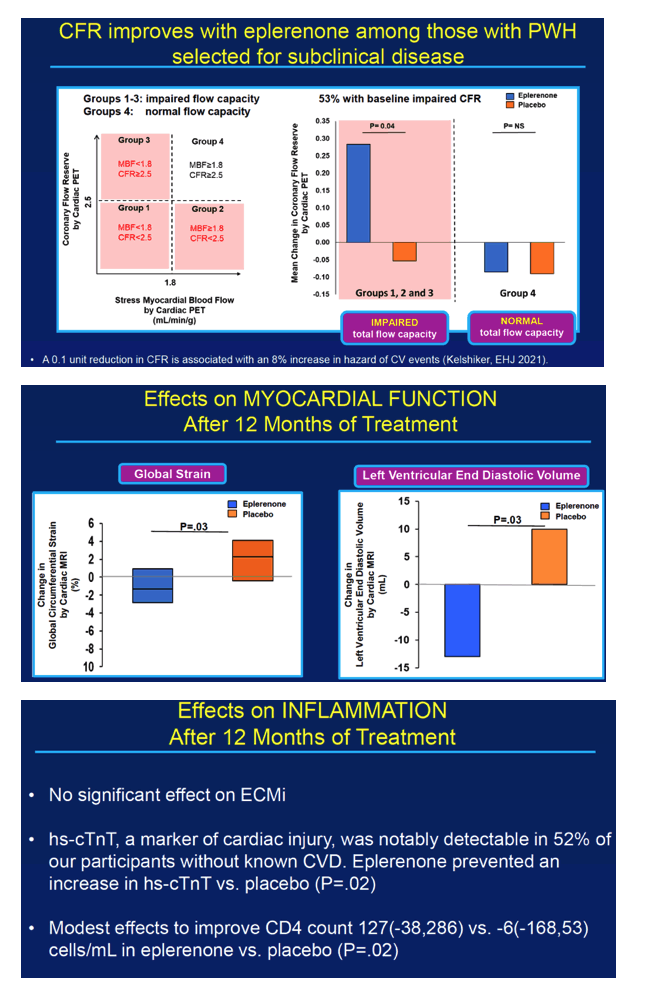
Because eplerenone's mechanism involves MR antagonism, only people randomized to the drug had a significant rise in urine aldosterone through 12 months (P = 0.02 vs placebo). At the 12-month point, the two measures of myocardial perfusion—coronary flow reserve and stress myocardial blood flow—dropped sharply in the placebo arm but inched upward in the eplerenone arm (P = 0.03 for change in stress myocardial blood flow with eplerenone versus placebo). In participants treated with eplerenone, change in stress myocardial blood flow correlated significantly with change in coronary flow reserve (rho = 0.69, P = 0.04). And coronary flow reserve improved significantly in the 53% of eplerenone recipients who had impaired coronary flow reserve before treatment began (P = 0.04 vs placebo).
At the 12-month point, two measures of myocardial function improved significantly with eplerenone versus placebo: left ventricular end diastolic volume (P = 0.03) and global circumference strain [2] (P = 0.03).
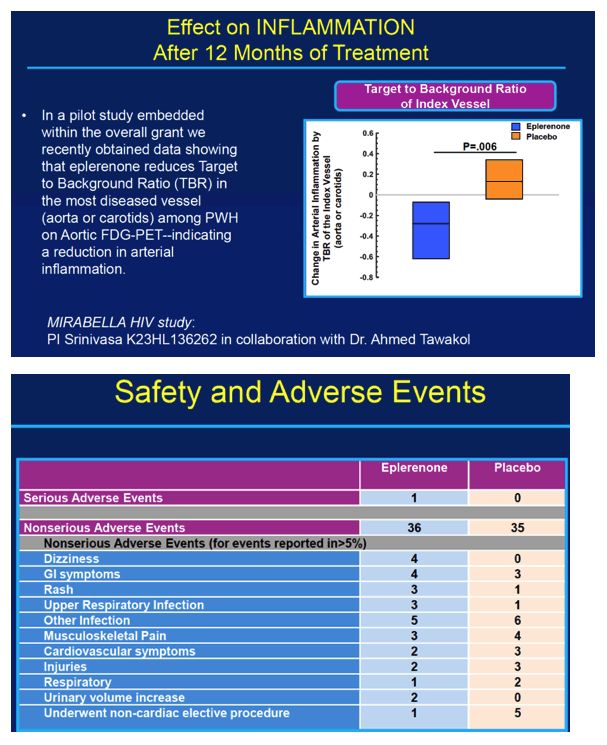
Among these study participants without known cardiovascular disease, the researchers detected hs-cTnT, a cardiac injury marker, in 52%. Eplerenone kept levels of this marker from rising during the 12-month trial, but placebo did not (P = 0.02). CD4 count rose by a median 127 cells in people assigned to eplerenone while dropping 6 cells in the placebo group (P = 0.02).
One serious adverse event arose in the eplerenone group and none with placebo. The most frequent nonserious adverse events in the eplerenone group were another infection in 5, dizziness in 4, and gastrointestinal symptoms in 4.
The Mass General/Harvard team proposed that MR antagonism with eplerenone "may have unique benefits" that prevent worsening subclinical myocardial disease in people with HIV infection. They recommended that studies should explore the impact of eplerenone-mediated RAAS blockade in the large group of people with HIV who have evidence of subclinical cardiac disease and impaired myocardial perfusion.
References
1. Srinivasa S, Walpert AR, Thomas TS, et al. Effects of eplerenone on myocardial perfusion and function: the Miracle HIV study. 30th CROI, Conference on Retroviruses and Opportunistic Infections, February 19-22, 2023, Seattle. Abstract 144.
2. Pu C, Fei J, Lv S, et al. Global circumferential strain by cardiac magnetic resonance tissue tracking associated with ventricular arrhythmias in hypertrophic cardiomyopathy patients. Front Cardiovasc Med. 2021;8. https://doi.org/10.3389/fcvm.2021.670361

|
| |
|
 |
 |
|
|