 |
 |
 |
| |
THE IMPACT OF 3BNC117, 10-1074, AND
LEFITOLIMOD ON HIV-1 PERSISTENCE: THE TITAN TRIAL
|
| |
| |
CROI 2023 Feb 20-23
Jesper D. Gunst1, Dag Henrik Reikvam2, James H. McMahon3, Marie H. Pahus1, Jesper F. Højen1, Isik S. Johansen4, Henrik Nielsen5, Thomas Lars Benfield6, Jan Gerstoft7, Sharon Lewin8, Martin Tolstrup1, Thomas A. Rasmussen1, Marina Caskey9, Michel C. Nussenzweig9, Ole S. Søgaard1
1Aarhus University Hospital, Aarhus, Denmark, 2Oslo University Hospital, Oslo, Norway, 3Monash University, Melbourne, Australia, 4Odense University Hospital, Odense, Denmark, 5Aalborg University Hospital, Aalborg, Denmark, 6Hvidovre Hospital, Hvidovre, Denmark, 7Copenhagen University Hospital, Copenhagen, Denmark, 8University of Melbourne, Melbourne, Australia, 9The Rockefeller University, New York, NY, USA
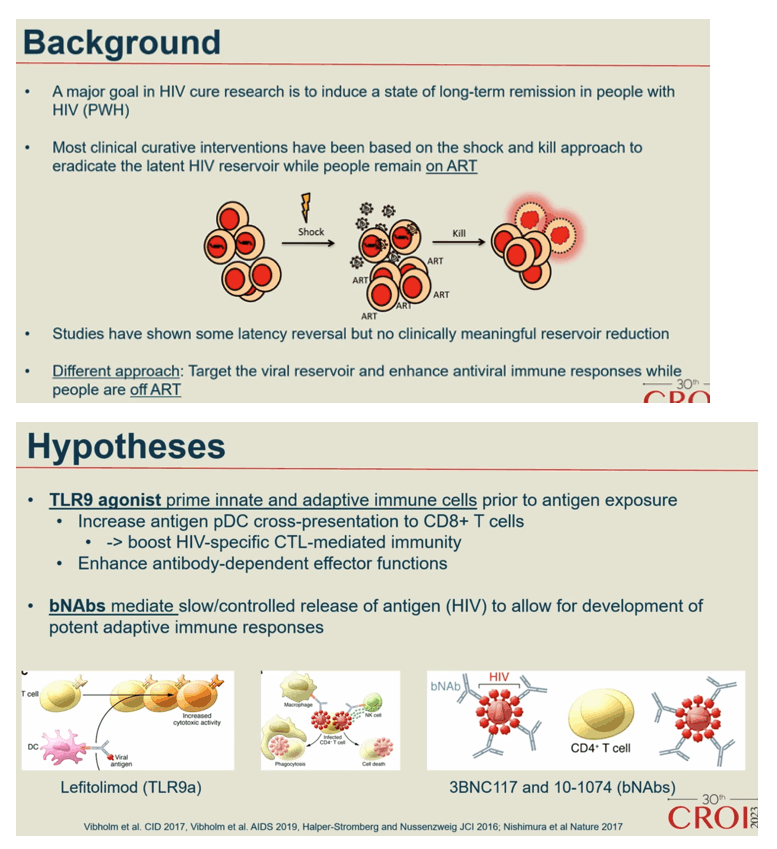
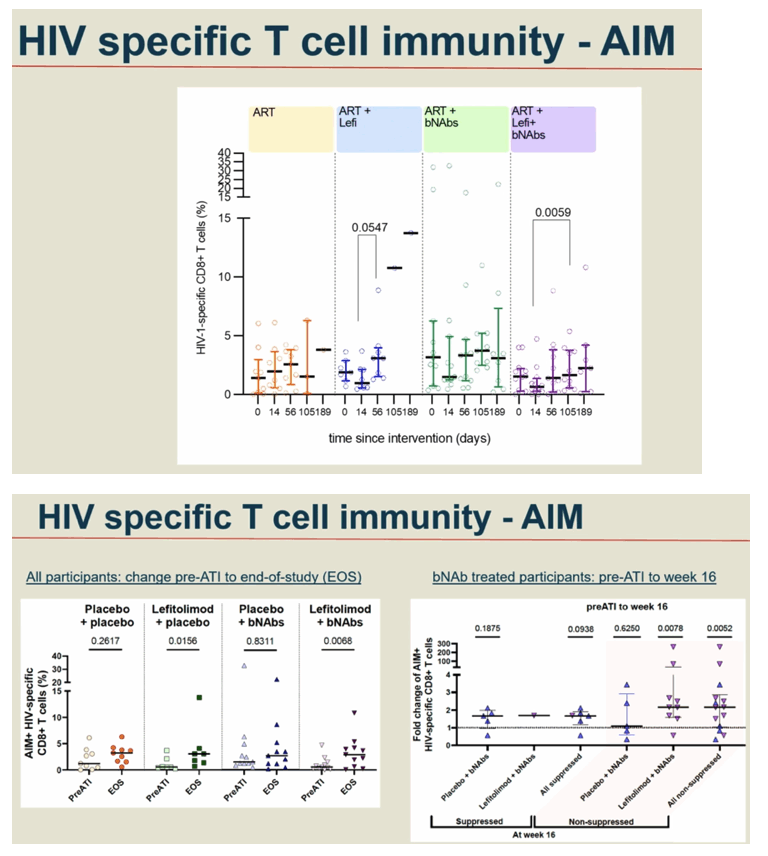
Background: Toll-like receptor 9 (TLR9) agonists stimulate innate antiviral immunity, prime adaptive immune responses and reverse latency in people with HIV-1 infection (PWH). Further, broadly neutralizing anti-HIV-1 antibodies (bNAbs) can boost HIV-specific immunity and enhance killing of infected cells through Fc-dependent mechanisms. Here, we evaluated the impact of the TLR9 agonist Lefitolimod and dual bNAb treatment on ART-free virological control.
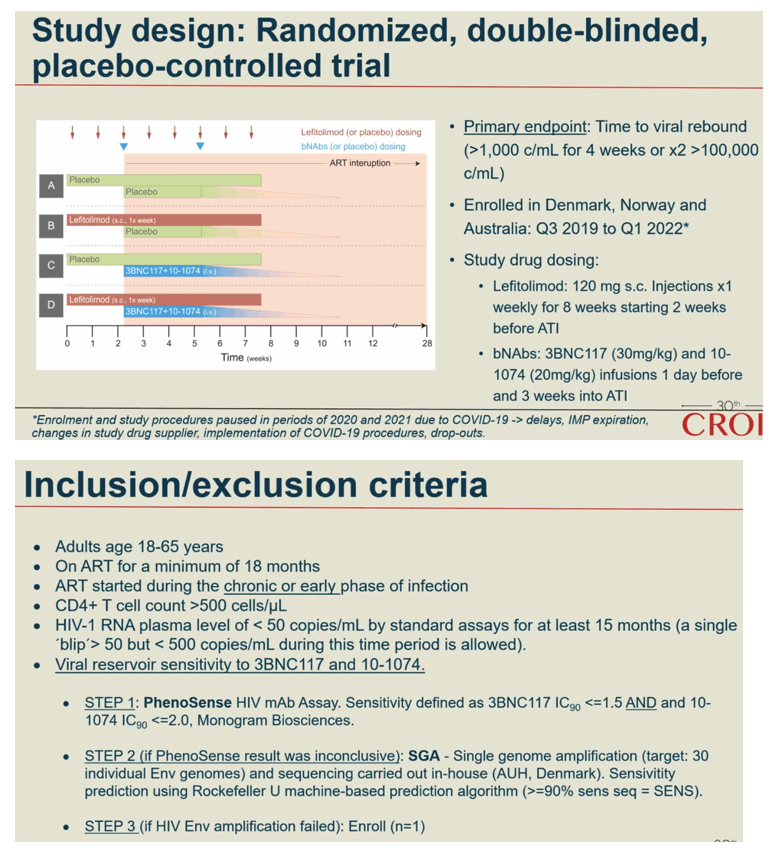
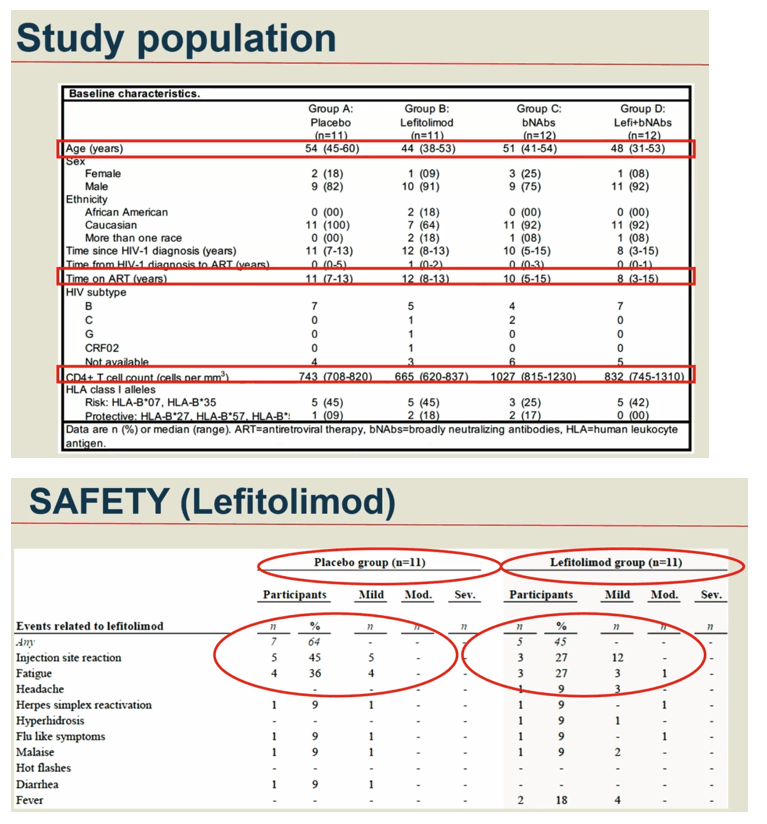
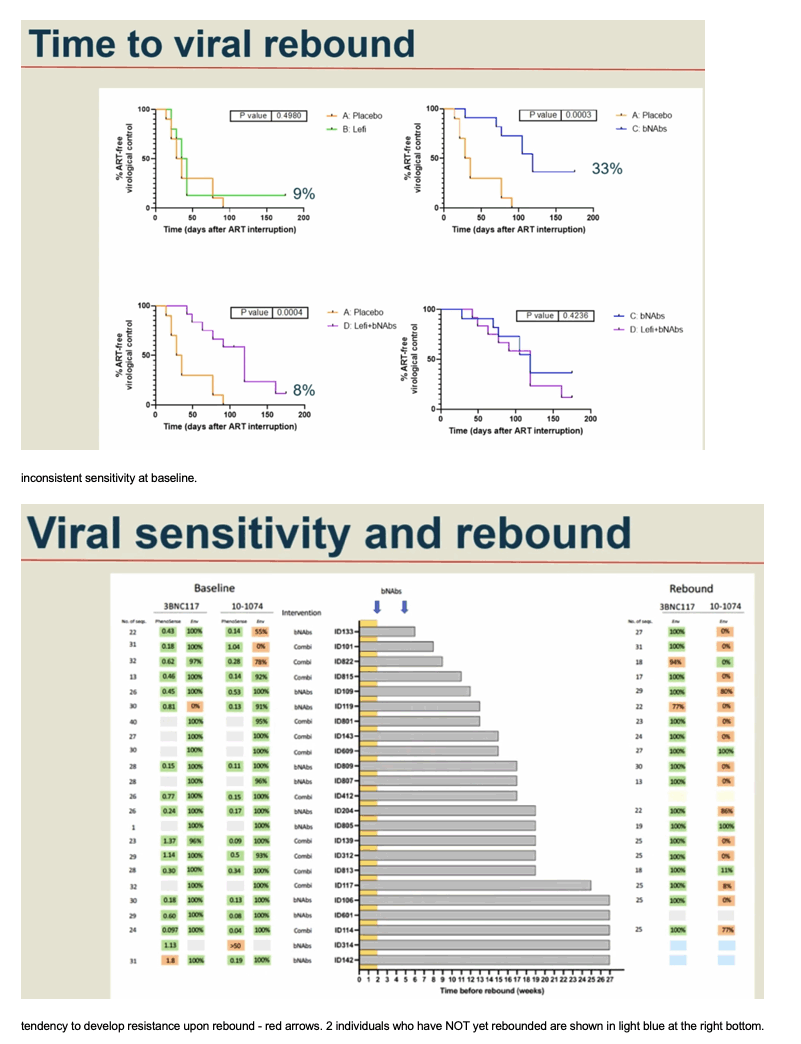
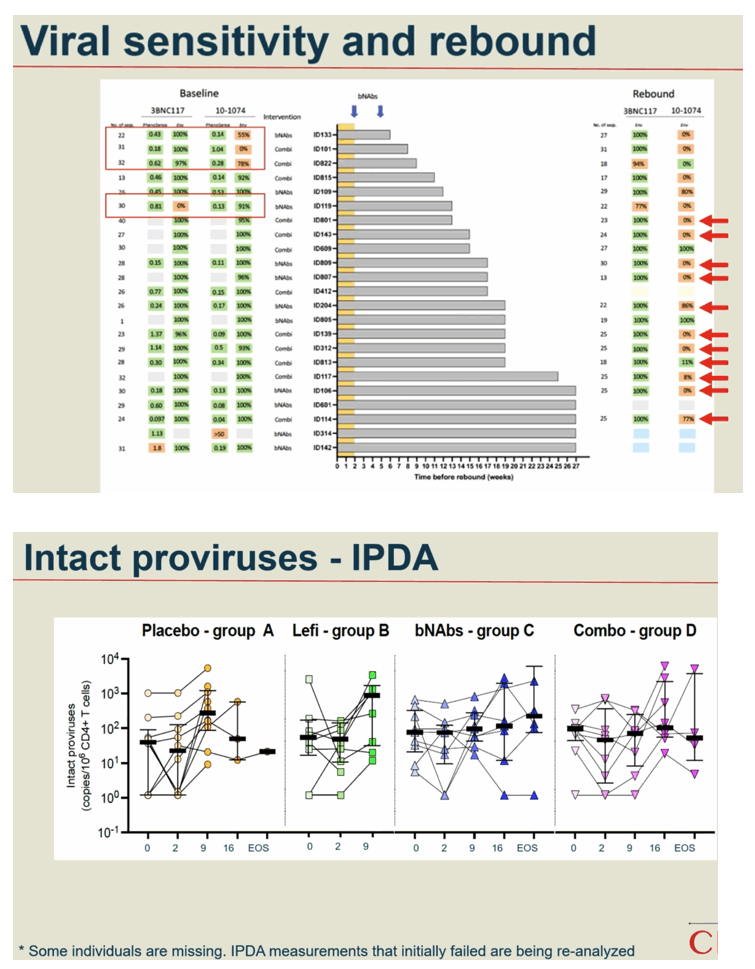
Methods: In a phase IIa multicenter, randomized placebo-controlled, double-blinded trial (TITAN study; NCT03837756), consenting PWH on suppressive ART were pre-screened for bNAb sensitivity and randomized into one of four groups: A) placebo /placebo; B) lefitolimod (LEFI) /placebo; C) placebo /bNAbs; or D) LEFI /bNAbs. LEFI (120 mg) or placebo were given subcutaneously (SC) weekly from week 1 through week 8. 3BNC117 at 30 mg/kg and 10-1074 at 20 mg/kg or placebo were given intravenously (IV) at week 3 and at week 6. Analytical treatment interruption (ATI) of antiretroviral therapy went from week 3 to 26, or viral rebound, whichever came first. The primary endpoint was time to loss of virological control (plasma HIV-RNA >1,000 c/mL for 4 weeks or confirmed >100,000 c/mL). Secondary endpoints were safety and rebound viral kinetics. bNAb sensitivity was assessed by PhenoSense and HIV envelope sequencing.
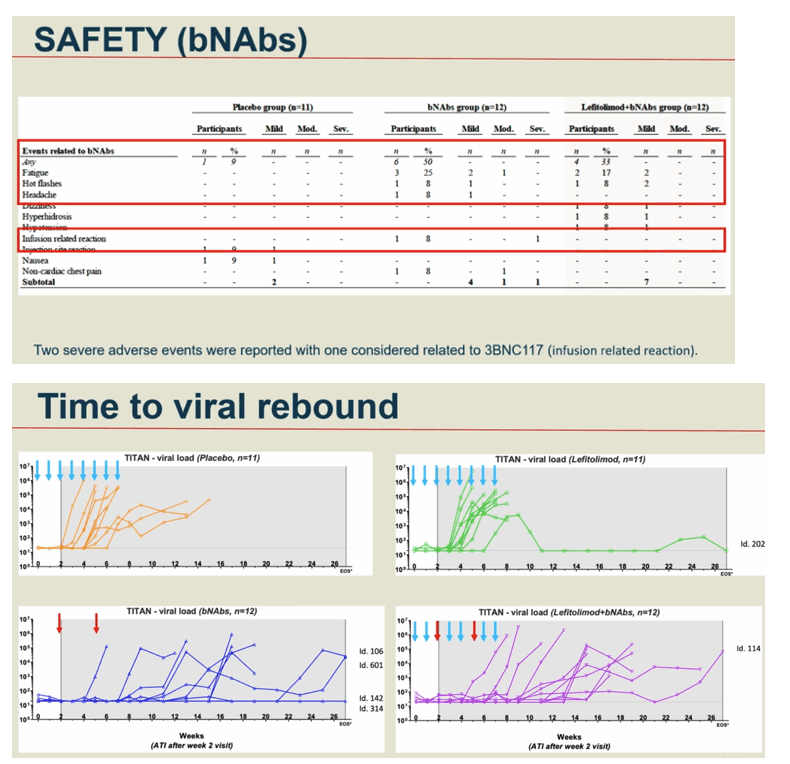
Results: 46 participants were randomized to either placebo/placebo (n=11), LEFI/placebo (n=11), placebo/bNAbs (n=12), or LEFI/bNAbs (n=12). The median time to loss of virological control was 32 days (interquartile range [IQR]:21-77) in group A, 35 days (IQR:28-42) in group B, 119 days (IQR:77-175) in group C, and 98 days (IQR:70-119) in group D. The groups receiving bNAbs had significant longer time to loss of virological control compared to placebo (log-rank, P< 0.0005). Six individuals did not meet ART restart criteria during the 24-week ATI (1 in B; 4 in C; 1 in D). The combination of LEFI and bNAbs vs. bNAbs alone did not result in improved virological control (log-rank, P=0.42). More mild adverse events were reported in group D. Two severe adverse events were reported (one vasovagal reaction [unrelated] and one infusion related reaction [considered related to 3BNC117]).
Conclusion: In a double-blinded trial among PWH on suppressive ART, harbouring virus sensitive to study bNAbs, the dual bNAb treatment led to a significant delay in viral rebound during ATI, but there was no added effect on virological control of the TLR9 agonist, lefitolimod.

webcast: https://www.croiwebcasts.org/console/player/51667?mediaType=slideVideo&


|
| |
|
 |
 |
|
|