 |
 |
 |
| |
Long-Acting CAB for PrEP Acceptable, Safe in Adolescent Female Africans
|
| |
| |
30th CROI, Conference on Retroviruses and Opportunistic Infections, February 19-22, 2023, Seattle
Mark Mascolini
Cisgender female Africans younger than 18 years old found long-acting cabotegravir (CAB) injections for preexposure prophylaxis (PrEP) highly tolerable, and 92% would prefer getting the injections every 1 or 2 months to taking daily oral TDF/FTC [1]. Among 53 young people who started CAB injections in HPTN 084-01, a three-country trial, no one stopped getting the shots because of intolerance through 29 weeks of treatment.
HPTN 084-01 investigators noted that stopping the HIV epidemic depends on preventing infection in key young populations-including adolescents, young women, and sexual and gender minority youth. Although global guidelines recommend oral PrEP for these young people, they face "profound inequalities in PrEP access" and stern challenges to adherence to tablet PrEP. Because long-acting CAB has proved successful in other populations, HPTN researchers conducted this single-arm, open-label, phase 2 safety study in African cisgender adolescent females.
Participants had to be assigned female at birth, under 18 years old, and weigh at least 35 kg (72 lbs). These young people had to report oral, anal, or vaginal sex with a male in the past 12 months. They could not be pregnant or breastfeeding and had to be willing to use a reliable long-acting form of contraception.
Everyone took 30 mg of oral CAB once daily for 5 weeks then switched to 3 mL (600 mg) gluteal injections for 29 weeks-the first 2 shots given 4 weeks apart and the remaining 3 shots 8 weeks apart. After this injection phase, participants could take daily oral TDF/FTC for 48 weeks or continue injected CAB in an open-label extension, HPTN 084 OLE. The presentation at CROI gave results for the first two phases of this trial.
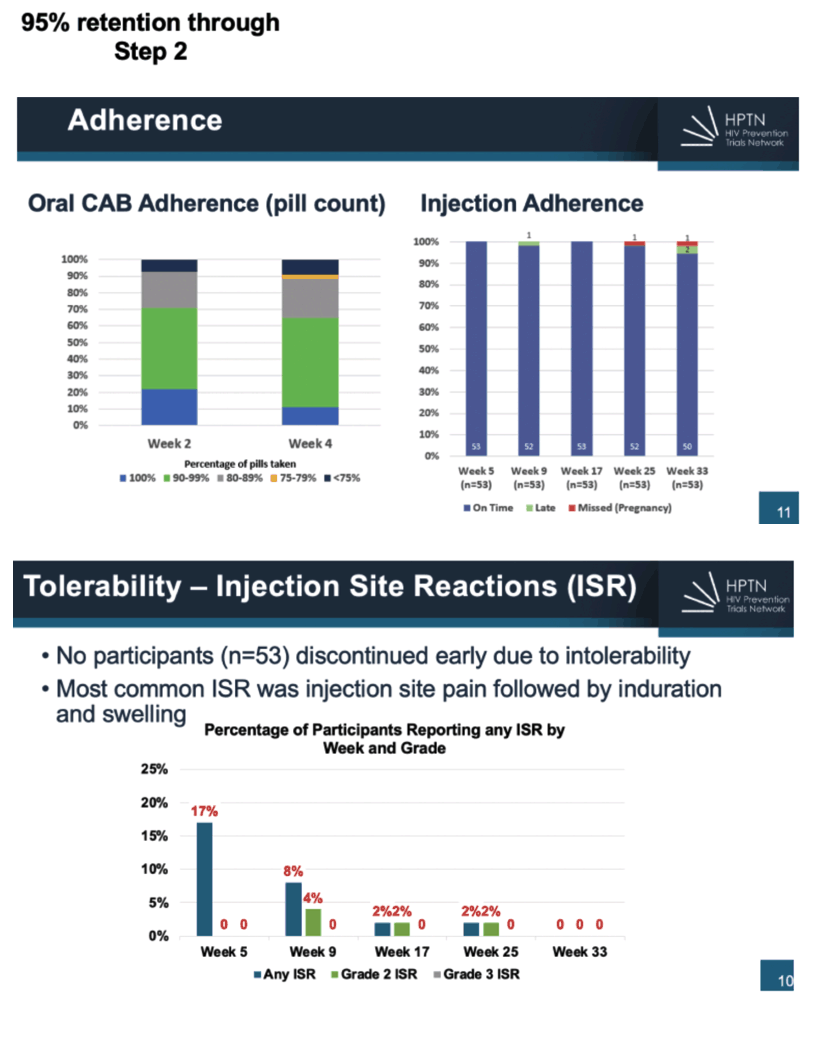
Researchers enrolled 55 participants from sites in Uganda, Zimbabwe, and South Africa, completing accrual 3 months ahead of schedule. Two participants stopped oral CAB because of grade 3 increased alanine aminotransferase or grade 3 increased lipase-neither change judged related to study drug. Of the 53 young people who entered the injection phase of the trial, 1 dropped out when she became pregnant. Among the 52 people who completed the injection phase, 48 (92%) opted to continue CAB injections and 4 chose oral TDF/FTC.
All participants were black African. They averaged 16 years in age (range 12 to 17), 73% weighed 50 kg or more, and 25% had at least one sex partner with HIV. The group had a median of 2 vaginal sex acts in the past month. Almost one quarter of the group, 22%, had transactional sex in the past month, and 36% had significant depressive symptoms on the CES-D-10.
During treatment, study clinicians recorded no product-related serious adverse events, and no one stopped treatment because of adverse events. No participants had significant weight gain, liver toxicity, hypersensitivity, rash, seizures, or pancreatitis. The most frequent adverse events in in the oral and injection phases of the trial were decreased creatinine clearance in 41 participants, higher blood glucose in 22, and increased blood creatinine in 9. All of these adverse events resolved spontaneously. Investigators recorded one grade 4 event, suicidal behavior, which responded to counseling.
Injection site reactions of any grade affected 17% of participants at week 5, 8% at week 9, 2% at week 17, 2% at week 25, and 0 at week 33. Respective proportions of grade 2 and 3 injection site reactions were 0, 4%, 2%, 2%, and 0.
No study participant became infected with HIV during the first two phases of the trial.
Pill counts showed that only 20% of participants took all pills through week 2 and only 10% through week 4. About 50% took 90% to 99% of pills at both points, and another 25% took 80% to 89% of pills. Almost all participants got their CAB shot on time at all 5 study visits. One person got her shot late at week 9, as did 2 at week 33.
The most frequent concerns about CAB injections were possible pain (mentioned by 28.3% of participants), possible harmful side effects (18.9%), and inability to reverse effects once CAB is injected (13.2%). About 11% of participants feared CAB injections may not protect against HIV, while 35.8% voiced no concerns about injected CAB.
Reference
1. Hosek S, Hamilton EL, Ngo J, et al. CAB LA for HIV prevention in African cisgender female adolescents (HPTN 084-01). 30th CROI, Conference on Retroviruses and Opportunistic Infections, February 19-22, 2023, Seattle. Abstract 162.
CAB LA FOR HIV PREVENTION IN AFRICAN CISGENDER FEMALE ADOLESCENTS (HPTN 084-01) - (02/23/23)
|
| |
|
 |
 |
|
|