 |
 |
 |
| |
Good Rectal TAF/EVG Levels With Rectal Inserts in HIV-Negative Men and Women
|
| |
| |
30th CROI, Conference on Retroviruses and Opportunistic Infections, February 19-22, 2023, Seattle
Mark Mascolini
Rectal inserts containing tenofovir alafenamide (TAF) and elvitegravir (EVG) provided high rectal fluid and tissue concentrations of EVG, tenofovir (TFV), and tenofovir diphosphate (TFV-DP, the active form of TAF) in a study of 17 men and 6 women without HIV [1]. Being studied for preexposure prophylaxis (PrEP), the inserts were safe and participants tolerated them well in this short study.
These fast-dissolving rectal inserts to protect people from sexual transmission of HIV contain 20 mg of the reverse transcriptase inhibitor TAF and 16 mg of the integrase inhibitor EVG. The inserts protected monkeys from vaginal [2] or rectal [3] simian HIV (SHIV) infection, and researchers have completed a phase 1 pharmacokinetic and safety study in women [4].
The new trial, MTN-039, set out to explore the PKs and safety of the TAF/EVG rectal insert at two doses—one insert and two inserts [1]. The study group consisted of healthy HIV-uninfected adults at low risk for HIV infection and not taking preexposure prophylaxis (PrEP) against HIV.
MTN researchers aimed to finish the trial with 20 evaluable participants with one rectal insert and 20 with two inserts. Twenty-one of 23 participants completed the first dosing visit evaluating the impact of one insert. After at least a 7-day washout, volunteers used two inserts, and 19 completed this phase of the trial. The MTN team measured EVG, TAF, and TFV in blood, rectal fluid, and rectal tissue homogenates. They measured TFV-DP in rectal tissue cell isolates.
Seventeen participants were male at birth and 6 female; 8 were black, 13 white, and 2 some other race or ethnicity. Median age stood at 34 years (interquartile range 24 to 43) and median body mass index at 30.8 kg/m2, just over the obesity threshold (interquartile range 26 to 37).
Nine participants reported 17 adverse events, only 1 of which (mild anal erythema) was related to the insert. The only two grade 2 adverse events (reactogenicity to the COVID vaccine and gastritis) were not related to the insert.
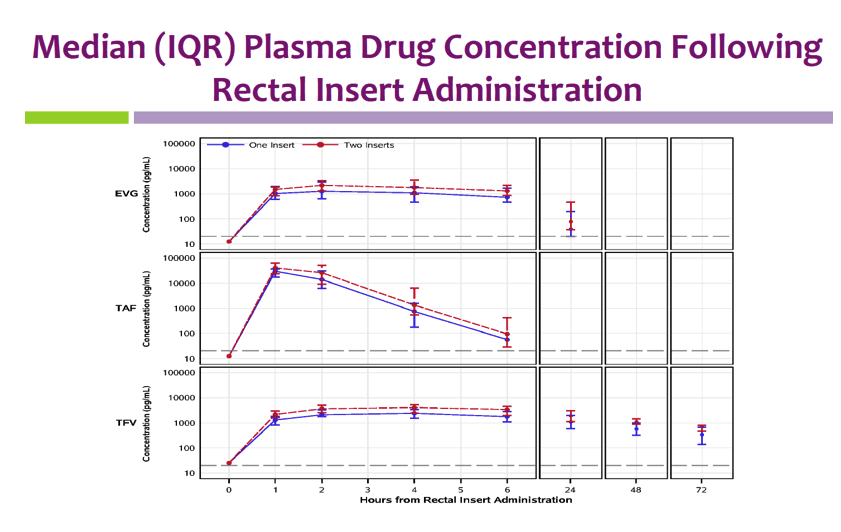
Median peak blood plasma concentrations with 1 and 2 inserts were 1.73/2.39 ng/mL for EVG, 29.5/41.9 ng/mL for TAF, and 2.64/4.42 ng/mL for TFV. Concentrations of EVG and TFV plateaued near the peak concentration for 6 hours.
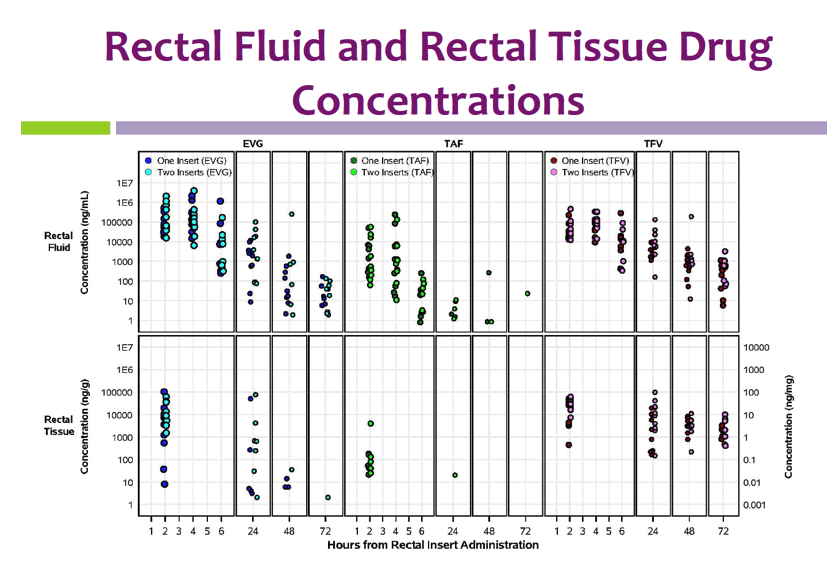
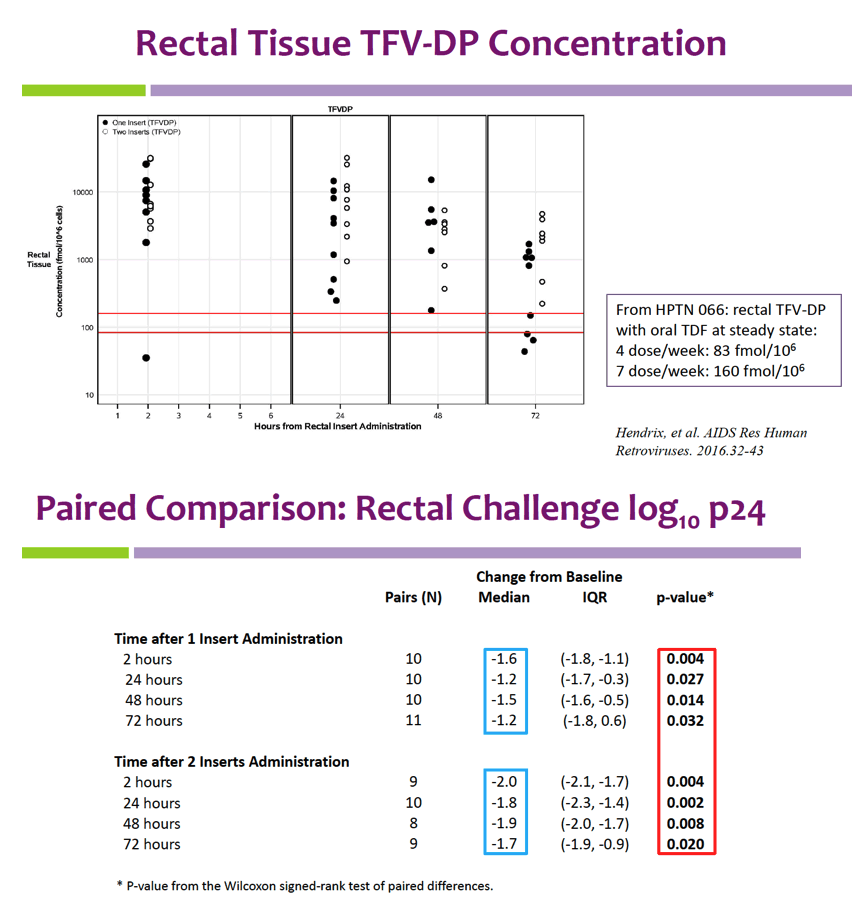
Insert administration yielded higher rectal fluid and tissue levels than blood plasma levels of TFV and EVG. Median rectal fluid concentrations with 1 and 2 inserts at 24 hours were 2234/2597 ng/mL for EVG and 2909/8277 ng/mL for TFV. Rectal tissue TFV-DP levels almost always exceeded rectal tissue levels after oral dosing of tenofovir disoproxil fumarate (TDF) in an earlier trial.
After HIV challenge of rectal tissue, HIV p24 antigen levels proved significantly lower than baseline p24 at every interval measured—2, 24, 48, and 72 hours after insertion of both the first and second TAF/EVG insert. At those four time points, median change from baseline p24 ranged from -1.2 to -1.6 log10 with a single insert, and from -1.7 to -2.0 log10 with 2 inserts.
MTN researchers proposed that these pharmacokinetic findings, coupled with protection from SHIV challenge in monkeys [2,3], justify continued study of TAF/EVG rectal inserts.
References
1. Riddler SA, Kelly C, Hoesley C, et al. Safety and PK/PD of a tenofovir alafenamide/ elvitegravir insert administered rectally. 30th CROI, Conference on Retroviruses and Opportunistic Infections, February 19-22, 2023, Seattle. Abstract 164.
2. Dobard CW, Peet MM, Nishiura K, et al. Single dose topical inserts containing tenofovir alafenamide fumarate and elvitegravir provide pre- and post-exposure protection against vaginal SHIV infection in macaques. EBioMedicine. 2022;86:104361. doi: 10.1016/j.ebiom.2022.104361. https://www.thelancet.com/journals/ebiom/article/PIIS2352-3964(22)00543-6/fulltext
3. Makarova N, Singletary T, Peet MM, et al. Pharmacokinetics and efficacy of topical inserts containing tenofovir alafenamide fumarate and elvitegravir administered rectally in macaques. EBioMedicine. 2022;86:104338. doi: 10.1016/j.ebiom.202.104338. https://www.thelancet.com/journals/ebiom/article/PIIS2352-3964(22)00520-5/fulltext
4. Thurman A, Ouattara AL, Yousefieh N, et al. A phase I study to assess safety, pharmacokinetics and pharmacodynamics of a topical multipurpose prevention insert containing tenofovir alafenamide fumarate and elvitegravir dosed vaginally. HIVR4P. 2021. Abstract OA06.05. https://onlinelibrary.wiley.com/doi/full/10.1002/jia2.25659 [search for OA06.05].





|
| |
|
 |
 |
|
|