 |
 |
 |
| |
LA CAB + RPV Every 4 Weeks Controls HIV in 131 of 133 With High Nonadherence Risk, Unstable Housing, Mental Illness, Homelessness, drug use
|
| |
| |
30th CROI, Conference on Retroviruses and Opportunistic Infections, February 19-22, 2023, Seattle
Mark Mascolini
Long-acting cabotegravir plus rilpivirine (CAB + RPV) injected every 4 weeks made viral loads undetectable in 131 of 133 San Francisco residents with a high risk of poor adherence to antiretroviral therapy [1]. Fifty-seven of these people (43%) began CAB + RPV with a detectable viral load. All used public insurance, and high proportions had unstable housing, used stimulants, or had a major mental illness.
Long-acting injected CAB + RPV initially won approval for antiretroviral-naive or experienced people who kept their viral load undetectable for at least 5 months with oral therapy. Researchers at the University of California, San Francisco (UCSF) noted that a substantial number of people with HIV face a steep challenge in reaching or sustaining an undetectable viral load with oral medication because of problems like mental illness and homelessness.They conducted this study of adherence-challenged people using San Francisco's Ward 86 HIV Clinic, all of whom rely on public insurance (Medicaid, Medicare, or an AIDS Drug Assistance Program). Since the researchers began this study, the FDA licensed direct-to-inject CAB + RPV, eliminating the oral therapy lead-in requirement. The San Francisco researchers published earlier results of this analysis with a smaller number of patients in August 2022 [2].
Registrational trials for long-acting CAB + RPV required participants to have an undetectable viral load for at least 16 weeks while taking an oral regimen. They could have no history of virologic failure, no integrase inhibitor mutations, and only the K103N nonnucleoside mutation. In the San Francisco demonstration project, participants did not need an undetectable viral load with oral therapy before starting CAB + RPV injections. They could have no integrase inhibitor or RPV mutations. To enter the program, people had to express a willingness to come to the clinic every 4 weeks for their injections. This rigorous protocol reviewed participant progress every 2 weeks.
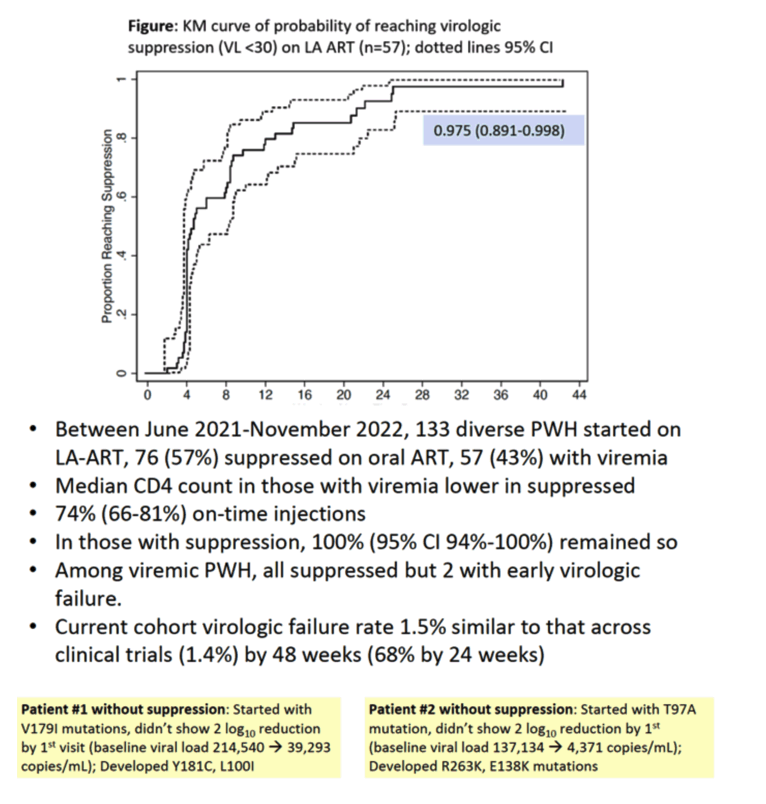
From June 2021 through November 2022, 133 people started long-acting CAB + RPV, 76 of them with an undetectable viral load and 57 with a detectable load. Median age stood at 45 years, 88% of participants were cisgender men, 8% cisgender women, and 4% transgender women. This ethnically mixed population was 38% Hispanic, 32% white, 16% black, and 14% multiracial. More than half of participants, 58%, had unstable housing, and 8% were homeless. While 130 people used Medicare or Medicaid, 3 paid for treatment through an AIDS Drug Assistance Program. One third of participants used a stimulant at the time of enrollment, and 38% had a major mental illness. Median CD4 count stood at 616 in people who began CAB + RPV with an undetectable viral load and at 215 in those who began with a detectable load (above 30 copies).
Three quarters of CAB + RPV injections were given on schedule. Kaplan-Meier analysis determined that 97.5% of all participants would have an undetectable viral load at a median of 26 weeks. All 76 people who began CAB + RPV with an undetectable viral load maintained that status throughout follow-up of 40 weeks or more. Among the 57 people who started CAB + RPV injections with a detectable viral load, 55 had an undetectable load at a median of 33 days and 2 had early virologic failure. At 48 weeks 2 of 133 people had virologic failure to yield a failure rate of 1.5%, similar to the 1.4% rate seen in clinical trials of CAB + RPV through 48 weeks.
The two people with virologic failure before week 24 had evidence of minor mutations that led the researchers to make the protocol stricter. One of these two people had the V179I nonnucleoside accessory mutation when treatment began. As the viral load fell from 214,540 to 39,293 copies, the nonnucleoside mutations Y181C and L100I arose. The other person with virologic failure began treatment with the T97A integrase inhibitor accessory mutation. As the viral load dropped from 137,134 to 4371 copies, the R263K integrase inhibitor mutation and the and E138K RPV-associated mutation evolved.
The UCSF investigators concluded that long-acting injectable CAB + RPV proved virologically successful in a large majority of people who had high rates of traditional adherence risk factors such as substance use, housing instability, and mental illness, and many of whom had a detectable viral load when they began getting injections. They proposed that long-acting injectable antiretroviral therapy "may be indicated for patients with adherence challenges unable to achieve or maintain viral suppression on oral antiretroviral therapy to extend its benefits."

References
1. Gandhi M, Salazar J, Hickey MD, et al. High virologic suppression rates on long-acting ART in a safety-net clinic population. 30th CROI, Conference on Retroviruses and Opportunistic Infections, February 19-22, 2023, Seattle. Abstract 518.
2. Christopoulos KA, Grochowski J, Mayorga-Munoz F, et al. First demonstration project of long-acting injectable antiretroviral therapy for persons with and without detectable human immunodeficiency virus (HIV) viremia in an urban HIV clinic. Clin Infect Dis. 2023;76:375-381. https://academic.oup.com/cid/article/76/3/e645/6652895
CROI: HIGH VIROLOGIC SUPPRESSION RATES ON LONG-ACTING ART IN A SAFETY-NET CLINIC POPULATION - (02/23/23)
CROI: Projected Benefits of Long-Acting ART (LA-ART) in PWH Viremic Not Taking Oral Therapy - (02/23/23)
|
| |
|
 |
 |
|
|