 |
 |
 |
| |
CROI 2023: Lenacapavir Treatment, Prevention
|
| |
| |
CROI: Long-Acting Lenacapavir in a Combination Regimen for Treatment Naïve PWH: Week 80 - (02/25/23)
CROI: Week 52 Subgroup Efficacy of Lenacapavir in Heavily Treatment-Experienced PWH - (02/24/23)
CROI: LENACAPAVIR WITH bNAbs GS-5423 AND GS-2872 DOSED EVERY 6 MONTHS IN PEOPLE WITH HIV - (02/23/23)
Susceptibility Screening to bNAbs Teropavimab (GS-5423) and Zinlirvimab (GS-2872) in ART-Suppressed Participants - (02/23/23)
CROI: Lenacapavir Protects Against Rectal SHIV Acquisition in Macaque Model - (02/24/23)
CROI: Antiviral Activity of Lenacapavir Against HIV-2 Isolates - (02/24/23)
Study to Compare Bictegravir/Lenacapavir Versus Current Therapy in People With HIV-1 Who Are Successfully Treated With a Complicated Regimen
The Phase 2 clinical trial (NCT05052996) evaluating an investigational oral once-weekly combination treatment regimen of Merck’s islatravir and Gilead's lenacapavir in adults with HIV-1 infection who are virologically suppressed will resume.
https://www.natap.org/2022/HIV/092022_01.htm
Update on Gilead's HIV Prevention Trials - (12/12/22)
New PrEP Studies - Lenacapavir; Adolescent Girls, Young Women - (12/12/22)
--------------------------------------
Lenacapavir in first-line therapy
Chloe Orkin
January, 2023
These data suggest that lenacapavir combined with a second potent agent can be effective in maintaining virological suppression, which is consistent with previous studies of two-drug regimens.18, 19
Lenacapavir administered every 26 weeks or daily in combination with oral daily antiretroviral therapy for initial treatment of HIV: a randomised, open-label, active-controlled, phase 2 trial
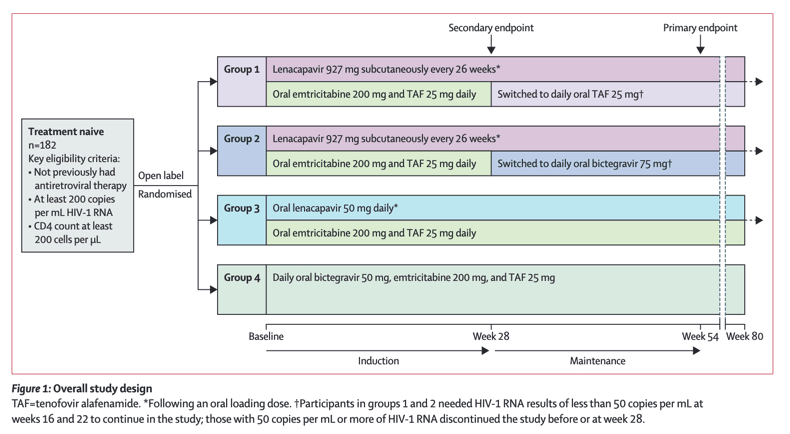
Lenacapavir is flexible in its route of administration and dosing intervals, with oral (daily to weekly) and injectable (up to every 6 months) dosing. When combined with other antiretroviral agents with synchronous dosing schedules, a range of dosing options could be possible for people with HIV. There are currently no phase 3 trials planned for any of the specific regimens evaluated in this study, all of which include antiretrovirals requiring daily administration. Oral lenacapavir is being investigated in an all-oral, once-weekly dual combination regimen (NCT05052996). For subcutaneous injectable lenacapavir, there are ongoing efforts to develop a long-acting partner agent that can be combined with lenacapavir as a complete synchronous regimen, offering the best potential for sustained adherence.
Where are we now with long-acting therapy? Based on an extensive registrational trial programme, long-acting injectable cabotegravir and rilpivirine is the first complete regimen to be recommended for the treatment of HIV-1 infection.1, 2, 3 The regimen is indicated for the treatment of virally suppressed individuals without hepatitis B co-infection who meet other virological suitability criteria.2 It is not yet universally available.
Lenacapavir is the second long-acting injectable treatment for HIV-1 infection to receive a license.4
It is a potent, first-in-class capsid inhibitor that is active at multiple points in the viral lifecycle, with a flexible pharmacokinetic profile that facilitates subcutaneous injection every 6 months with potential for weekly oral therapy.5 The licensed indication for lenacapavir is different from long-acting injectable cabotegravir and rilpivirine. Lenacapavir has been licensed by the European Agency for the Evaluation of Medicinal Products for the treatment of individuals with HIV-1 with extensive treatment experience and for whom it is otherwise impossible to construct a viable regimen.4 The CAPELLA study5 in heavily treatment-experienced individuals showed that when lenacapavir is combined with an optimised backbone, impressive levels of viral suppression over 52 weeks are possible, as is significant immune restoration. Injection site reactions were frequent but mild and there were very few resulting discontinuations. Capsid inhibitor resistance occurred in eight participants, almost all of whom were either non-adherent to the optimised backbone or had no other active agents.
|
| |
|
 |
 |
|
|