 |
 |
 |
| |
TAF and Cobicistat-Boosted Drugs Tied to More Somatic Depressive Symptoms
|
| |
| |
30th CROI, Conference on Retroviruses and Opportunistic Infections, February 19-22, 2023, Seattle
Mark Mascolini
A study of 1538 US women with HIV independently linked tenofovir alafenamide (TAF) combined with a cobicistat (COBI)-boosted integrase inhibitor (INSTI) or protease inhibitor (PI) to somatic depressive symptoms like sleep and appetite problems [1]. The analysis tied tenofovir disoproxil fumarate (TDF) plus a nonnucleoside (efavirenz or rilpivirine) to fewer somatic depressive symptoms. If confirmed in men and other cohorts, the results could further complicate the complex balancing act behind antiretroviral prescribing. But findings in two recent small trials in Spain [2,3] and a prospective cohort study in China [4]-all three involving mostly men-perhaps contradicted some of the WIHS results in US women. The Spanish and Chinese studies are outlined below.
Neuropsychiatric conditions like depression often go undiagnosed and untreated in people with HIV. Few studies have explored potential associations between today's antiretroviral combinations and depression. To address that shortcoming, researchers working with the Women's Interagency HIV Study (WIHS) in the United States assessed the combined impact of antiretroviral regimens on somatic depressive symptoms (like sleep and appetite problems) and nonsomatic depressive symptoms (like sadness) in WIHS women with HIV.
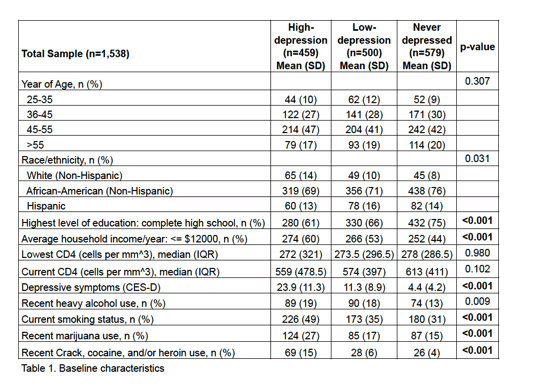
The analysis focused on WIHS women who had at least two study visits while taking a contemporary antiretroviral regimen. Researchers used longitudinal Center for Epidemiologic Studies Depression (CES-D) scale scores to divide the study group in 459 women with high depression, 500 with low depression, and 579 never-depressed women.
At their first WIHS visit, women classified as having high depression had a higher average number of depressive symptoms on the CES-D than women with low depression or never depression (23.9, 11.3, 4.4, P < 0.001). The three groups did not differ much in age (about 30% 36 to 45, about 45% 45 to 55). Slightly higher proportions of white women had high versus low or never depression (14%, 10%, 8%), compared with black women (69%, 71%, 76%) and Hispanic women (13%, 16%, 14%), but black women made up more than 70% of the whole study group (P = 0.031).
A higher proportion of never-depressed women than women with low depression or high depression completed high school (75%, 66%, 61%, P < 0.001), and a lower proportion of never-depressed women than women with low depression or high depression averaged under $12,000 in yearly household income (44%, 53%, 60%, P < 0.001). Significantly higher proportions of women with high depression than low depression or never depression currently smoked (P < 0.001), recently used marijuana (P < 0.001), or recently used crack, cocaine, or heroin (P < 0.001).
Novel Bayesian machine-learning methods accounting for relevant variables estimated associations between current antiretroviral regimens and somatic or nonsomatic depressive symptoms.
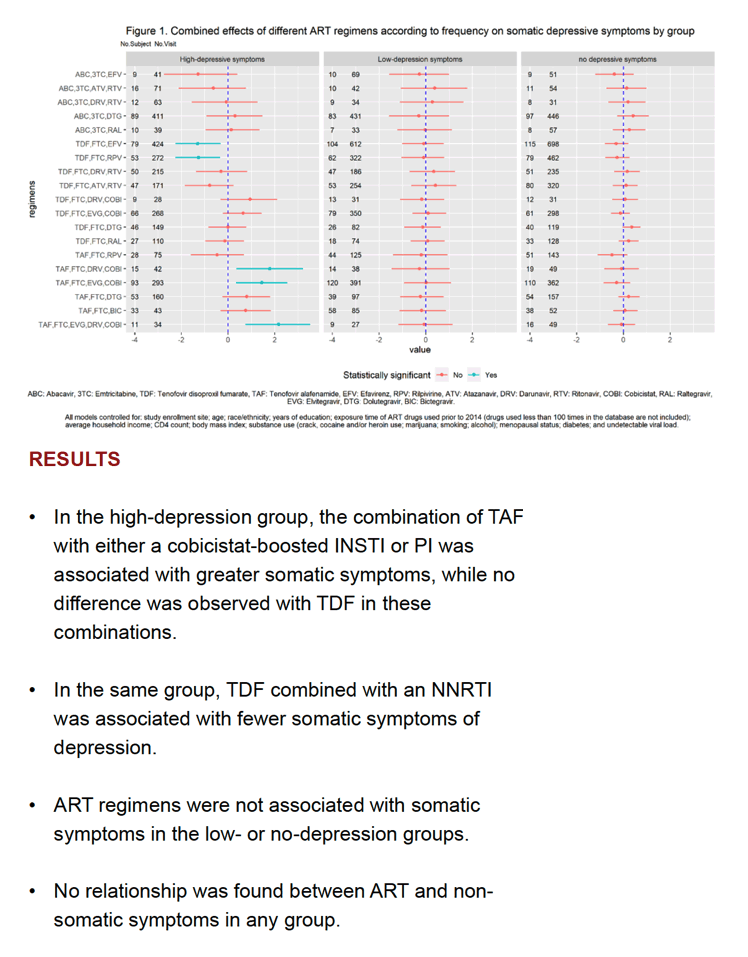
Regimens independently linked to more somatic depressive symptoms in the high-depression group:
- TAF, FTC, darunavir/COBI
- TAF, FTC, elvitegravir/COBI
- TAF, FTC, elvitegravir, darunavir/COBI
Regimens independently linked to fewer somatic depressive symptoms in the high-depression group :
- TDF, FTC, efavirenz
- TDF, FTC, rilpivirine
This analysis linked no other antiretroviral regimens to more or fewer somatic symptoms in the high-depression group, the low-depression group, or the never-depression group, including all combinations including abacavir (ABC)/3TC and any others including TDF and FTC or TAF and FTC (even TAF plus FTC plus unboosted dolutegravir or bictegravir). None of these regimens-including the five independently linked to more or fewer somatic depressive symptoms-were associated with nonsomatic depressive symptoms.
The WIHS investigators believe their findings imply "complex associations between antiretroviral therapy combinations [and depression] . . . rather than individual agents" and depressive symptoms. But their poster offered no guesses on why the five regimens their analyses singled out are associated with more or fewer somatic depressive symptoms.
A small multicenter, open-label Spanish trial randomized 72 people (85% men) with poor sleep quality while taking ABC/3TC plus dolutegravir to keep that regimen or switch to TAF, FTC plus darunavir/COBI [2], one of the combos linked to more somatic depressive symptoms like sleep problems in the WIHS study. After 4 weeks Pittsburgh Sleep Quality Index (PSQI) scores stayed the same in people who kept taking ABC/3TC plus dolutegravir but improved significantly in people switching to TAF and FTC plus darunavir/COBI. Switchers also improved in Hospital Anxiety and Depression scale (HADS) scores and neuropsychiatric symptoms.
An even smaller multicenter, open-label Spanish trial randomized 38 people (almost all men) with neuropsychiatric symptoms while taking ABC/3TC plus dolutegravir to keep that combination or switch to TAF and FTC plus elvitegravir/COBI [3], another of the three implicated regimens in the WIHS analysis. Four, 12, and 24 weeks after the switch, people in the switch group had significant improvement in PSQI, HADS, and neuropsychiatric symptoms while no one maintaining the initial regimen improved or worsened.
A prospective Chinese cohort study involved 196 people, 97% of them men, with ongoing central nervous system toxicity at or above grade 2 while taking an efavirenz regimen [4]. They all had 48 weeks of follow-up after switching to single-pill elvitegravir/COBI/FTC/TAF, one of the regimens tied to somatic depressive symptoms in the WIHS analysis. Chinese cohort members had significant improvements in anxiety by HADS and sleep disturbance symptoms by PSQI 12, 24, and 48 weeks after trading their efavirenz combination for COBI-boosted elvitegravir plus TAF/FTC. But they had no improvement in depression symptom score.
These four studies had different designs and used different tools and statistical methods to assess changing depression symptoms with contemporary antiretroviral regimens. And the study populations could hardly be more dissimilar. But can US women with HIV differ diametrically from Spanish and Chinese men with HIV in depression symptoms related to some of the same regimens? Researchers who conducted the WIHS study may have more to say about potential gender-based differences because they plan to explore associations between contemporary antiretroviral regimens and depressive symptoms in men.
In response to an email query, Luis Parra-Rodriguez (Washington University in St. Louis), the presenting author for the WIHS study, noted a lack of evidence that TAF or COBI has neuronal toxicity. Some unidentified mechanism, he added, may explain the associations between these two agents and somatic depressive symptoms, "for example, recent evidence suggests that TAF may impair bioenergetics of immune cells." Other contributing factors may be a modulating effect of TAF or COBI (more likely COBI) on nonantiretroviral medications a person is taking, an off-target effect of TAF or COBI, or channeling bias in the study (although the researchers attempted to correct for channeling bias in their model).
References
1. Parra-Rodriguez L, O'Halloran J, Wang Y, et al. Effect of common antiretroviral combinations on depressive symptoms in women with HIV. 30th CROI, Conference on Retroviruses and Opportunistic Infections, February 19-22, 2023, Seattle. Abstract 469.
2. Cabello-Úbeda A, González Baeza A, Troya García J, et al. Changes in quality of sleep, mood, and other neuropsychiatric symptoms after switching dolutegravir/lamivudine/abacavir to darunavir/cobicistat/emtricitabine/tenofovir alafenamide in a randomized study of people with human immunodeficiency virus with poor sleep quality: GESIDA 10418. Open Forum Infect Dis. 2022, ofac345, https://doi.org/10.1093/ofid/ofac345
3. Perez Valero I, Cabello A, Ryan P, et al. Randomized trial evaluating the neurotoxicity of dolutegravir/abacavir/lamivudine and its reversibility after switching to elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide: GESIDA 9016. Open Forum Infect Dis. 2020;7:ofaa482. DOI: 10.1093/ofid/ofaa482
4. Xia H, Huang XJ, Hu Y, et al. Switching from efavirenz to elvitegravir/cobicistat/emtricitabine/tenofovir alafenamide reduces central nervous system symptoms in people living with HIV. Chin Med J (Engl). 2021;134:2850-2856. doi: 10.1097/CM9.0000000000001824


|
| |
|
 |
 |
|
|