 |
 |
 |
| |
EFFECTIVENESS OF SMALLPOX VACCINATION TO PREVENT MPOX IN MILITARY PERSONNEL
|
| |
| |
CROI 2023 Feb 20-23
program abstract
Boghuma K. Titanji1, Angelia Eick-Cost2, Lauren Epstein3, Natalie Wells2, Jeremy Smith4, Shauna L. Stahlman2, Abirami Balajee4, Saiju Pyarajan5, Chris W. Woods6, Mark Holodniy7, Elizabeth J. Partan5, Victoria J. Davey8, Robert A. Bonomo9, Yinong Young-Xu4, Vincent C. Marconi1
1Emory University, Atlanta, GA, USA, 2Armed Forces Health Surveillance Division, Defense Health Agency, U S. Department of Defense, Silver Spring, Maryland, USA., Silver Spring, MD, USA, 3Emory University School of Medicine, Atlanta, GA, USA, 4White River Junction Veterans Affairs Medical Center, White River Junction, VT, USA, 5VA Center for Data and Computational Sciences, Boston, MA, USA, 6Duke University, Durham, NC, USA, 7Stanford University, Los Angeles, CA, USA, 8US Department of Veterans Affairs, Washington, DC, USA, 9Case Western Reserve University School of Medicine, Cleveland, OH, USA
Background: The effectiveness of Smallpox vaccines against Mpox is unknown. In the global outbreak of Mpox, these vaccines are being used as prevention in individuals with high-risk of exposure to Mpox. There is an urgent need to determine their effectiveness.
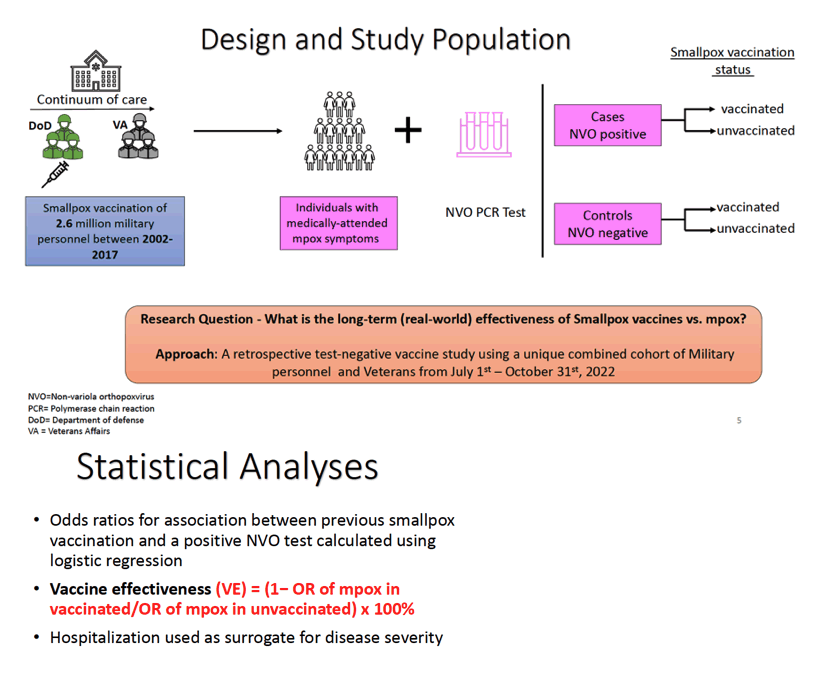
Methods: We conducted a retrospective, test-negative case-control study among US current and former military personnel. The goal of the study was to evaluate the vaccine effectiveness (VE) of smallpox vaccines against Mpox . Uni- and multivariate logistic regressions were used to calculate odds ratios (ORs) with 95% confidence interval (CI) for the association between positive Mpox testing and receipt of Smallpox vaccine. Vaccine effectiveness (VE)
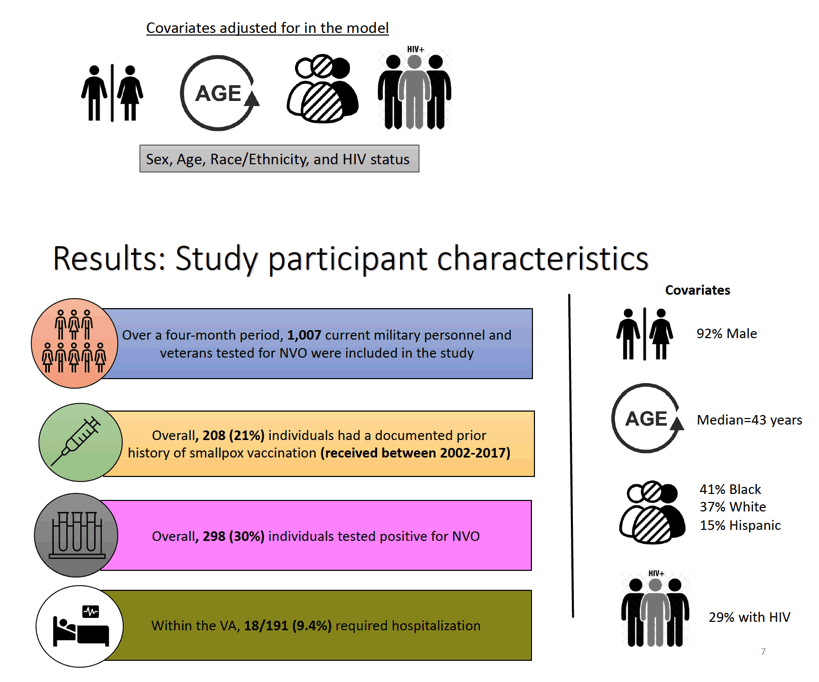
was estimated as (1- OR of Mpox in vaccinated/OR of Mpox in unvaccinated) x 100%. Models for adjusted analyses included covariates for potential confounders of the association between vaccination status and testing positive for Mpox. The adjustment covariates included age, race, sex, and HIV status. Among individuals tested in the VA system, severity of disease was defined by admission to the hospital for Mpox. This information was not available for individuals outside of a VA facility. Analyses were conducted using Stata17 and SAS9.4 software.
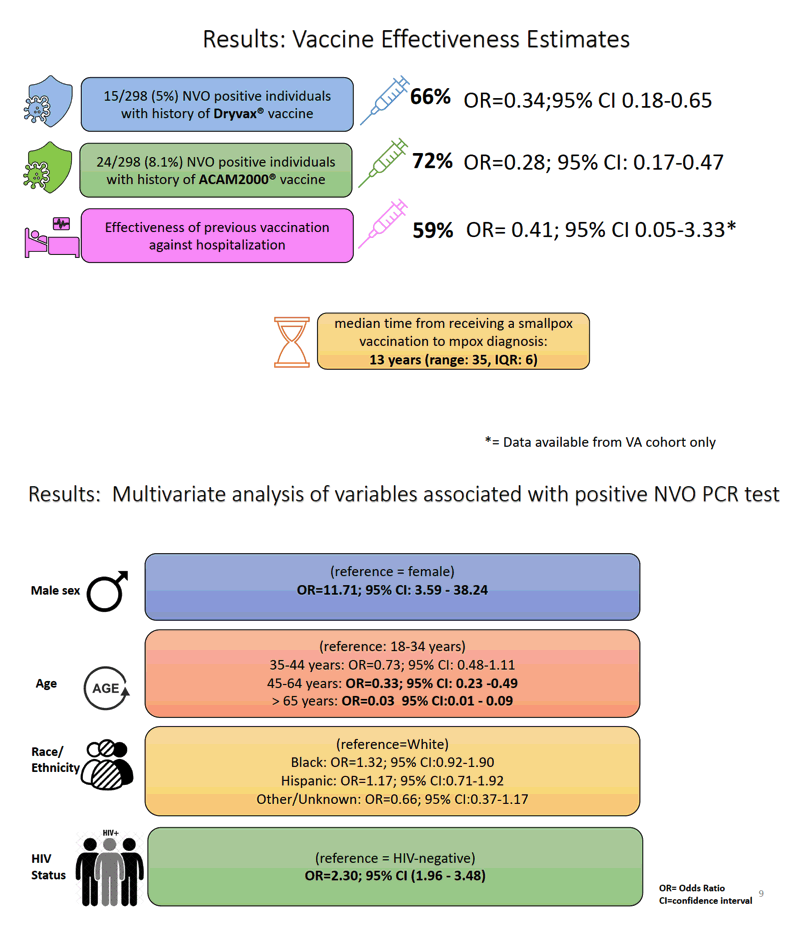
Results: The analysis included 1007 military personnel and Veterans who were tested for Orthopoxvirus between July 1-October 31, 2022, 208 (21%) of whom had a documented Smallpox vaccination. Among the 298 (30%) individuals who tested positive for Orthopoxvirus, 9.4% required hospitalization, 29% had HIV, 24 (8.3%) were previously vaccinated with ACAM2000 ("2nd-generation" Smallpox vaccine) and 15 (5%) were previously vaccinated with Dryvax ("1st-zzgeneration" Smallpox vaccine). The median time from receipt of Smallpox vaccination to Mpox diagnosis was 12-years (range:35, IQR:6.5). Individuals with prior ACAM2000 (OR=0.28; 95%CI: 0.17-0.47) or Dryvax (OR=0.36; 95%CI 0.18-0.73) vaccination were less likely to test positive for Orthopoxvirus compared to unvaccinated individuals. The estimated VE rate was 72% for ACAM2000 and 64% for Dryvax . Among those who tested positive for orthopoxvirus within the VA, 14/183 (7.7%) required hospitalization; Of those who tested positive, 126 (43.4%) were people with HIV (OR: 2.30, 95% CI:1.63 - 3.25).
Conclusion: Vaccination with Smallpox vaccines reduces the likelihood of testing positive for Orthopoxvirus. Our study provides the largest evaluation of the effectiveness of earlier smallpox vaccines against Mpox, supporting their usefulness in containing the global outbreak.



|
| |
|
 |
 |
|
|