 |
 |
 |
| |
Cabotegravir/rilpivirine based long acting
therapy with insufficient drug levels in routine drug monitoring
|
| |
| |
Two Thirds of RPV Troughs Below Target With CAB + RPV in Berlin Study
EACS 2023, October 18-21, 2023, Warsaw
Mark Mascolini
Two thirds of rilpivirine (RPV) trough concentrations lay below the target level in a Berlin study of 49 people getting cabotegravir (CAB) + RPV injections every 2 months [1]. Eleven people (22%) had CAB levels below the target trough.
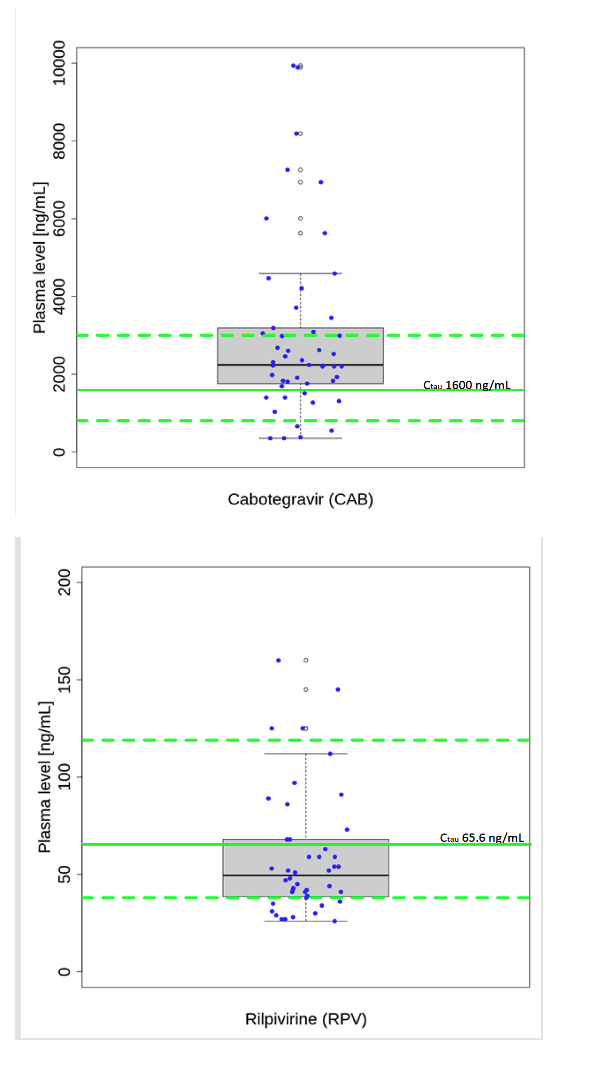
Workers at Berlin's Medical Center for Infectious Diseases measured CAB and RPV levels 8 weeks after 49 people started the long-acting injectables at standard doses of 600 mg for CAB and 900 mg for RPV every 2 months. According to hiv-druginteractions.org [2], target troughs should be approximately 1600 ng/mL (95% range 800 to 3000 ng/mL) for CAB and 65.6 ng/mL (95% range 38 to 119 ng/mL) for RPV.
Thirty-two of 49 RPV measurements (65%) by LC/MS/MS lay below the target trough. No RPV could be detected in plasma of 5 people (below 25 ng/mL). Eleven of 49 CAB troughs (22%) lay below the target concentration, including 7 people with low RPV troughs (below 65.6 ng/mL). Fifteen viral load measures done at the same time as drug level monitoring found 4 loads above 50 copies.
One person with CAB and RPV troughs above target (1910 and 91 ng/mL) had a viral load of 9000 copies. Resistance testing detected the integrase mutations L74I, S119P, G140AG, and Q148R plus the reverse transcriptase mutation E138K. This person with HIV-1 subtype A6 had the L74I integrase mutation before starting CAB + RPV and should not have begun a CAB regimen.
Another person with HIV-1 subtype A1 and with an adequate CAB trough but low RPV trough (45 ng/mL) had a viral load below 50 copies but a newly emerging Y181C reverse transcriptase mutation, "which appears to be evolving resistance to RPV compared to the pre-test in 2022."
Two other people with low RPV concentrations (subtype B and CRF01_AE) had viral loads detectable at 82 and 360 copies but no detectable resistance mutations in plasma.
The Berlin researchers "strongly recommend checking drug levels" when giving long-acting 2-drug antiretroviral therapies to avoid low drug levels, emergence of resistant HIV, and ultimately functional monotherapy. They say further research must clarify whether current drug concentration thresholds for CAB and RPV are correct, whether standard measurement methods are sensitive enough for new long-acting regimens, and "how much [drug concentration] margin is available before failure."
References
1. Ehret R, Krudewagen B, Obermeier M. Cabotegravir/rilpivirine based long acting therapy with insufficient drug levels in routine drug monitoring. EACS 2023, October 18-21, 2023, Warsaw. Abstract eP.B1.037.
2. www.hiv-druginteractions.org/prescribing_resources/hiv-pk-cabotegravir-im
www.hiv-druginteractions.org/prescribing_resources/hiv-pk-rilpivirine-im


|
| |
|
 |
 |
|
|