| |
Treating hepatitis D with bulevirtide - Real-world experience from 114 patients
|
| |
| |
Download the PDF here
March 15, 2023
we herein report data from five patients with initially decompensated liver disease who showed good virologic and biochemical response rates. This is the very first - even though anecdotical - evidence that bulevirtide may also safely be given to patients with decompensated cirrhosis.
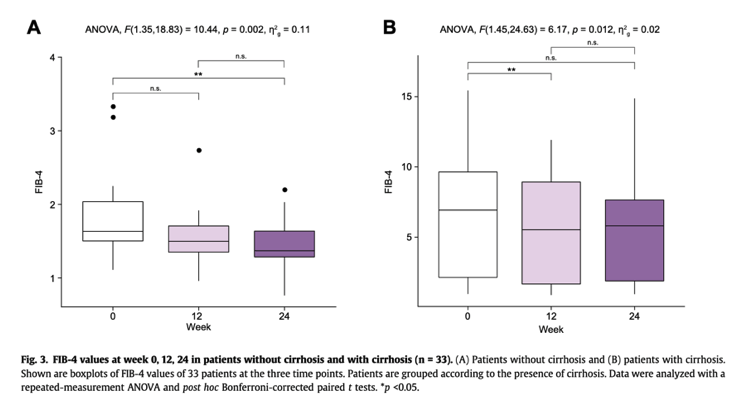
Bulevirtide is the only approved treatment for hepatitis D. Herein, we report the first data from the largest multicenter real-world cohort of patients with hepatitis D treated with the approved dose of 2 mg bulevirtide without additional PEG-IFNa. Our main findings were that (i) more than 50% of patients indeed achieve a virologic response (at least 2 log HDV RNA decline) with less than 10% of patients not achieving an HDV RNA drop of at least 1 log after 24 weeks; (ii) an improvement of biochemical hepatitis activity as measured by ALT values was seen regardless of virologic response status, and (iii) treatment was safe and well tolerated and prolonged treatment was associated with better clinical surrogate parameters of cirrhosis and portal hypertension.
Highlights
• 114 patients were treated with bulevirtide under real-world conditions, including 50 with signs of significant portal hypertension.
• A virologic response was observed in 87/114 patients, with 25 patients achieving HDV-RNA negativity.
• ALT levels improved regardless of virologic response status.
• In a small subgroup of patients with decompensated liver disease, bulevirtide treatment appeared to be safe.
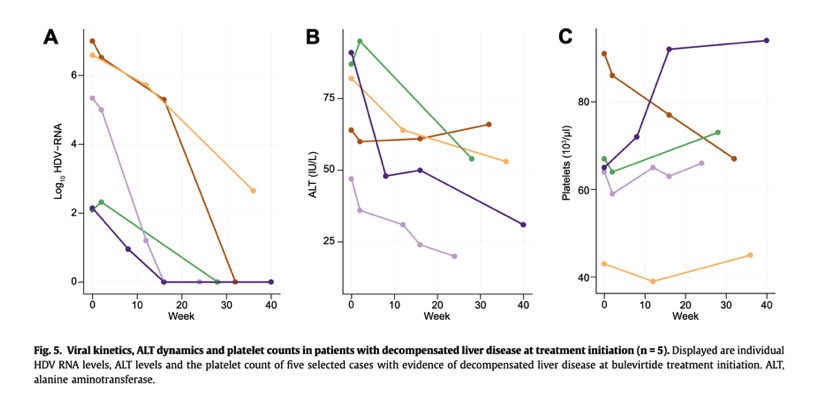
Fig. 5. Viral kinetics, ALT dynamics and platelet counts in patients with decompensated liver disease at treatment initiation (n = 5).
Displayed are individual HDV RNA levels, ALT levels and the platelet count of five selected cases with evidence of decompensated liver disease at bulevirtide treatment initiation. ALT, alanine aminotransferase.
There was evidence of decompensated liver disease at treatment initiation in five patients. Four cases were classified as Child-Pugh B and one case as Child-Pugh C. These patients are of special interest as clinical trials excluded patients with decompensated cirrhosis. As shown in Fig. 5, all five patients showed a virologic response. All but one patient showed decreasing ALT levels and rising platelet counts. One patient with refractory ascites (shown in purple in Fig. 5) experienced a temporary improvement in ascites.
Background & Aims
Bulevirtide is a first-in-class entry inhibitor of hepatitis B surface antigen. In July 2020, bulevirtide was conditionally approved for the treatment of hepatitis D, the most severe form of viral hepatitis, which frequently causes end-stage liver disease and hepatocellular carcinoma. Herein, we report the first data from a large multicenter real-world cohort of patients with hepatitis D treated with bulevirtide at a daily dose of 2 mg without additional interferon.
Methods
In a joint effort with 16 hepatological centers, we collected anonymized retrospective data from patients treated with bulevirtide for chronic hepatitis D.
Results
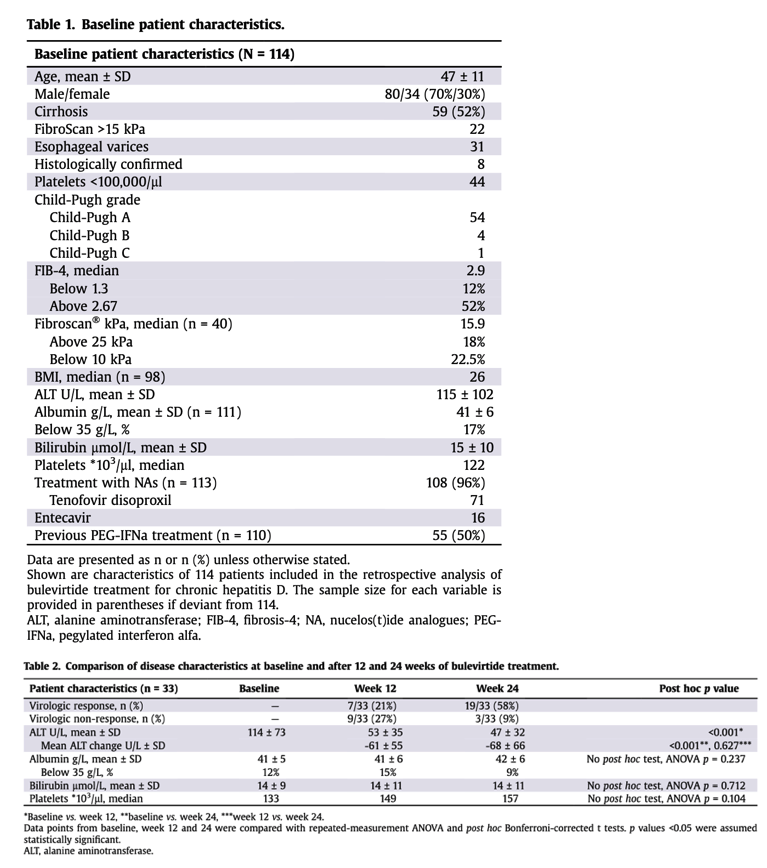
Our analysis is based on data from 114 patients, including 59 (52%) with cirrhosis, receiving a total of 4,289 weeks of bulevirtide treatment. A virologic response defined as an HDV RNA decline of at least 2 log or undetectable HDV RNA was observed in 87/114 (76%) cases with a mean time to virologic response of 23 weeks.
In 11 cases, a virologic breakthrough (>1 log-increase in HDV RNA after virologic response) was observed.
After 24 weeks of treatment, 19/33 patients (58%) had a virologic response, while three patients (9%) did not achieve a 1 log HDV RNA decline.
No patient lost hepatitis B surface antigen. Alanine aminotransferase levels improved even in patients not achieving a virologic response, including five patients who had decompensated cirrhosis at the start of treatment. Treatment was well tolerated and there were no reports of drug-related serious adverse events.
Conclusions
In conclusion, we confirm the safety and efficacy of bulevirtide monotherapy in a large real-world cohort of patients with hepatitis D treated in Germany. Future studies need to explore the long-term benefits and optimal duration of bulevirtide treatment.
Impact and implications
Clinical trials proved the efficacy of bulevirtide for chronic hepatitis D and led to conditional approval by the European Medical Agency. Now it is of great interest to investigate the effects of bulevirtide treatment in a real-world setting. In this work, we included data from 114 patients with chronic hepatitis D who were treated with bulevirtide at 16 German centers. A virologic response was seen in 87/114 cases. After 24 weeks of treatment, only a small proportion of patients did not respond to treatment. At the same time, signs of liver inflammation improved. This observation was independent from changes in hepatitis D viral load. The treatment was generally well tolerated. In the future, it will be of interest to investigate the long-term effects of this new treatment.
|
|
| |
| |
|
|
|