| |
Antisense Reduced LPa by 35% to 72%
|
| |
| |
Download the PDF here
Download the PDF here
Download the PDF here
3 publications
Lipoprotein(a) Reduction in Persons with Cardiovascular Disease - antisense oligonucleotide AKCEA-APO(a)-LRx
Effect of Pelacarsen on Lipoprotein(a) Cholesterol and Corrected Low-Density Lipoprotein Cholesterol
Lipoprotein(a) Cholesterol Masquerading as Low-Density Lipoprotein Cholesterol: Catch Me if You Can
-----------------------------
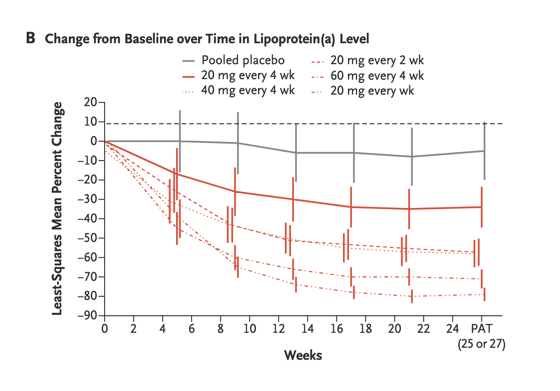
At 6 months of exposure (25 or 27 weeks), dose-dependent mean percent reductions in lipoprotein(a) from baseline were noted in all the APO(a)-LRx groups, with decreases of 35% at a dose of 20 mg every 4 weeks, 56% at 40 mg every 4 weeks, 58% at 20 mg every 2 weeks, 72% at 60 mg every 4 weeks, and 80% at 20 mg every week, as compared with 6% for the pooled placebo group (P value range for the comparison with placebo, 0.003 to <0.001) (Figure 1A).
Lipoprotein(a) Reduction in Persons with Cardiovascular Disease
Abstract
Background
Lipoprotein(a) levels are genetically determined and, when elevated, are a risk factor for cardiovascular disease and aortic stenosis. There are no approved pharmacologic therapies to lower lipoprotein(a) levels.
Methods
We conducted a randomized, double-blind, placebo-controlled, dose-ranging trial involving 286 patients with established cardiovascular disease and screening lipoprotein(a) levels of at least 60 mg per deciliter (150 nmol per liter). Patients received the hepatocyte-directed antisense oligonucleotide AKCEA-APO(a)-LRx, referred to here as APO(a)-LRx (20, 40, or 60 mg every 4 weeks; 20 mg every 2 weeks; or 20 mg every week), or saline placebo subcutaneously for 6 to 12 months. The lipoprotein(a) level was measured with an isoform-independent assay. The primary end point was the percent change in lipoprotein(a) level from baseline to month 6 of exposure (week 25 in the groups that received monthly doses and week 27 in the groups that received more frequent doses).
Results
The median baseline lipoprotein(a) levels in the six groups ranged from 204.5 to 246.6 nmol per liter. Administration of APO(a)-LRx resulted in dose-dependent decreases in lipoprotein(a) levels, with mean percent decreases of 35% at a dose of 20 mg every 4 weeks, 56% at 40 mg every 4 weeks, 58% at 20 mg every 2 weeks, 72% at 60 mg every 4 weeks, and 80% at 20 mg every week, as compared with 6% with placebo (P values for the comparison with placebo ranged from 0.003 to <0.001). There were no significant differences between any APO(a)-LRx dose and placebo with respect to platelet counts, liver and renal measures, or influenza-like symptoms. The most common adverse events were injection-site reactions.
Conclusions
APO(a)-LRx reduced lipoprotein(a) levels in a dose-dependent manner in patients who had elevated lipoprotein(a) levels and established cardiovascular disease. (Funded by Akcea Therapeutics; ClinicalTrials.gov number, NCT03070782. opens in new tab.)
Lipoprotein(a) is composed of a low-density lipoprotein (LDL)-like moiety bound covalently to apolipoprotein(a).1,2Lipoprotein(a) potentially contributes to cardiovascular disease through proatherogenic effects of its LDL-like moiety, proinflammatory effects of its oxidized phospholipid content, and prothrombotic effects through its inactive, plasminogen-like protease domain on apolipoprotein(a). Mechanistic, epidemiologic, and genetic evidence reported over the past 20 years provides support for the idea that elevated plasma lipoprotein(a) is an independent genetic risk factor for cardiovascular disease and calcific aortic-valve stenosis.3,4 In contrast, genetically determined low levels of lipoprotein(a) (<30 mg per liter [<75 nmol per liter]) are associated with a decreased risk of cardiovascular disease but not of other non-cardiovascular disease adverse phenotypes.5
There are currently no approved pharmacologic therapies that specifically target lipoprotein(a). Antisense oligonucleotides (ASOs) inhibit the production of apolipoprotein(a) in the hepatocyte, the source of approximately 99% of plasma lipoprotein(a).6 Preclinical proof-of-concept studies have established that ASOs targeting hepatic LPA messenger RNA (mRNA) specifically reduce plasma levels of lipoprotein(a).7,8 Subsequent phase 1 and 2 studies of a non-hepatocyte-targeted, second-generation ASO showed lowering of lipoprotein(a) levels in healthy participants who had elevated lipoprotein(a), as well as in patients with established cardiovascular disease and elevated plasma levels of lipoprotein(a).9,10 Advances in directing ASOs to hepatocytes by conjugation with a triantennary N-acetylgalactosamine (GalNAc3) moiety, a high-affinity ligand for the asialoglycoprotein receptor on the surface of hepatocytes, have resulted in large increases (by a factor of 15 to 30) in their potency,10 with implications for improvements in the side-effect profile and safety of ASOs.11,12 AKCEA-APO(a)-LRx - here referred to as APO(a)-LRx and previously called IONIS-APO(a)-LRx - is a GalNAc3-conjugated 2'-methoxyethyl chimeric second-generation ASO drug targeted to LPA mRNA. In a phase 2a trial, APO(a)-LRx was shown to result in a dose-dependent reduction of 66 to 92% in circulating lipoprotein(a) in participants with elevated lipoprotein(a) levels.10 The long half-life of APO(a)-LRx(approximately 1 month) that was observed in that trial prompted us to consider longer dosing intervals in the trial we report here.

|
|
| |
| |
|
|
|