| |
Susceptibility to 3BNC117 and 10-1074 in
ART suppressed chronically infected persons
|
| |
| |
Download the PDF here
CROI-2023: Susceptibility Screening to bNAbs Teropavimab (GS-5423) and Zinlirvimab (GS-2872) in ART-Suppressed Participants - (02/23/23)
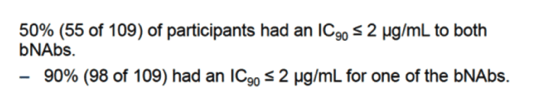
LENACAPAVIR WITH bNAbs GS-5423 AND GS-2872 DOSED EVERY 6 MONTHS IN PEOPLE WITH HIV
(ABSTRACT 193)
Joseph J. Eron
University of North Carolina at Chapel Hill, Chapel Hill, NC, United States
https://www.croiwebcasts.org/console/player/51834?mediaType=slideVideo&
CROI: LENACAPAVIR WITH bNAbs GS-5423 AND GS-2872 DOSED EVERY 6 MONTHS IN PEOPLE WITH HIV - (03/22/23)
Pablo TEBAS 1 , Kenn LYNN1 , Livio AZZONI 2 , Giorgio COCCHELLA 1 , Emmanouil PAPASAVVAS 2 , Matthew FAIR2 , Brijesh KARANAM2 , Paridhima SHARMA 2 , Jacqueline D
REEVES 3 , Christos J PETROPOULOS 3 , Linden LALLEY-CHARECZKO 4 , Jay R. KOSTMAN5 , Willian SHORT1 , Karam MOUNZER4 , Luis J. MONTANER 1
Abstract
Objective:
The aim of this study was to assess the susceptibility of HIV to two HIV monoclonal antibodies (bnAbs), 3BNC117 and 10-1074, in individuals with chronically suppressed HIV infection.
Design:
The susceptibility of bnAbs was determined using the PhenoSense mAb Assay, which is a cell-based infectivity assay designed to assess the susceptibility of luciferase-reporter pseudovirions. This assay is the only CLIA/CAP compliant screening test specifically developed for evaluating bnAb susceptibility in people with HIV infection.
Method:
The susceptibility of luciferase-reporter pseudovirions, derived from HIV-1 envelope proteins obtained from PBMCs of 61 ART-suppressed individuals, to 3BNC117 and 10-1074 bnAbs was assessed using the PhenoSense mAb assay. Susceptibility was defined as an IC90 of <2.0 μg/ml and 1.5 μg/ml for 3BNC117 and 10-1074, respectively.
Results:
About half of the individuals who were chronically infected and virologically suppressed were found to harbor virus with reduced susceptibility to one or both of the tested bnAbs.
The baseline viruses of 24% of the participants exhibited an IC90 greater than 2 μg/mL against 3BNC17 and the baseline viruses of 30% of the participants exhibited an IC90 greater than 1.5 μg/mL against 10-1074. Seven individuals (13%) were resistant to both. Paired IC90 values were independent of one another, e.g.: viruses resistant to 3BNC117 were neither more nor less likely to be resistant to 10-1074 (Figure 2B).
Conclusions:
The reduced combined susceptibility of 3BNC117 and 10-1074 highlights a potential limitation of using only two bnAbs for PREP or treatment. Further studies are needed to define and validate the clinical correlates of bnAb susceptibility.
Given the high rate of preexisting resistance, the expense in manufacturing, and the variability of the pharmacokinetics of the different products both at the individual and the product level, accurate estimates of susceptibility to bnAbs versus clinical response are needed as well as alternative methods of delivery of multiple of these molecules (mRNA, DNA, multispecificity[16] , AAV [17], etc.) to make these therapeutic interventions sustainable, particularly when compared to use of long- acting, low molecular weight antiretroviral agents.
|
|
| |
| |
|
|
|