| |
Vaccine Effectiveness of JYNNEOS against Mpox Disease in the United States
|
| |
| |
Download the PDF here
May 2023
Our findings have implications for public health and clinical practice. Evidence from immunogenicity studies provided the basis for implementing intradermal vaccination as a strategy to increase vaccine supply, because intradermal administration requires one fifth the dosage of subcutaneous administration.7,26 We found that two doses of vaccine, including those administered subcutaneously, intradermally, or heterologously, provided the highest protection against mpox disease.26 Furthermore, our finding that vaccine effectiveness was higher among persons who received two doses, rather than one dose, highlights the importance of following the approved dosing schedule. Recent data suggest that only 57.6% of first-dose vaccine recipients who were eligible to receive a second dose did so.27
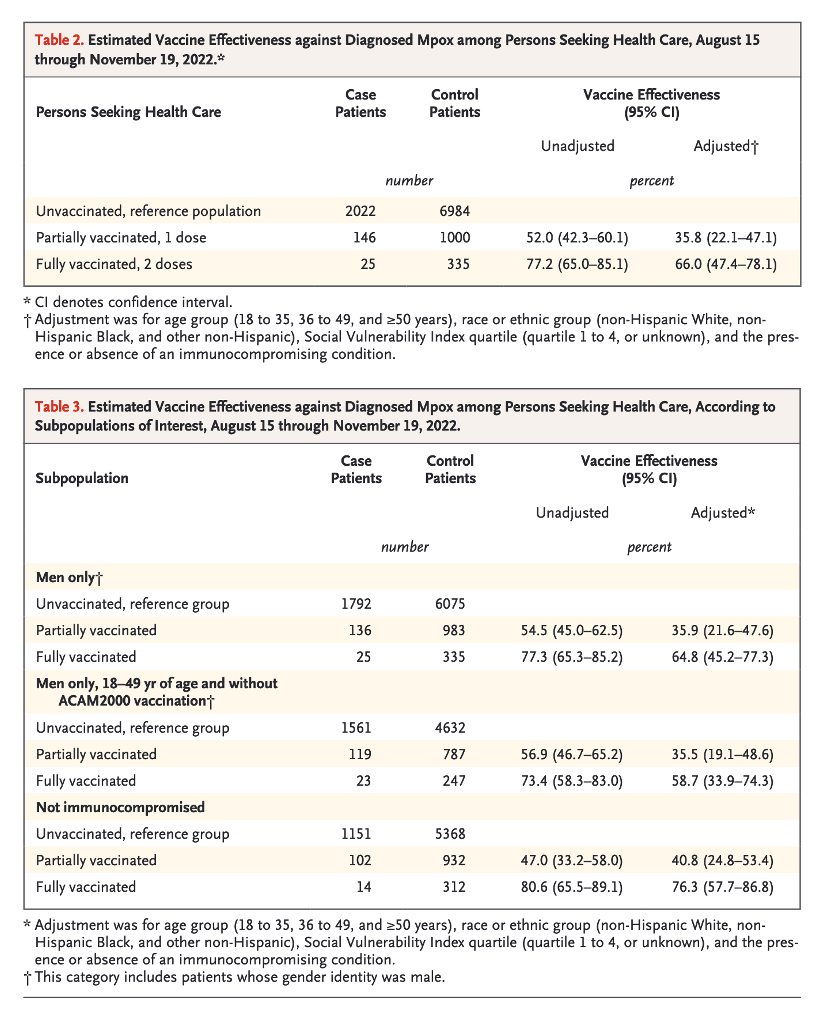
In this study using nationwide EHR data, patients with mpox were less likely to be vaccinated with JYNNEOS vaccine than control patients; the estimated adjusted vaccine effectiveness was 66.0% (95% CI, 47.4 to 78.1) in fully vaccinated persons and 35.8% (95% CI, 22.1 to 47.1) in partially vaccinated persons. These results suggest that both one and two doses of the vaccine provide protection against mpox disease.
Among men 18 to 49 years of age without a history of ACAM2000 vaccination, adjusted vaccine effectiveness was 58.7% (95% CI, 33.9 to 74.3) in fully vaccinated persons and 35.5% (95% CI, 19.1 to 48.6) in those who were partially vaccinated. Persons 50 years of age or older, who were excluded from this subpopulation analysis, might have had a previous smallpox vaccination19 or might have been at lower risk for mpox disease than younger men22; however, the point estimates for this subpopulation were similar to the overall point estimates.
Among persons without immunocompromising conditions, vaccine effectiveness was 76.3% (95% CI, 57.7 to 86.8) in those who were fully vaccinated and 40.8% (95% CI, 24.8 to 53.4) in those who were partially vaccinated; these percentages were higher than the overall point estimate, although confidence intervals overlapped. Persons with immunocompromising conditions might mount a less effective immune response after vaccinations,23 such that vaccine effectiveness is lower in this group than among persons without such conditions. Persons with immunocompromising conditions compose a large proportion of those with mpox and may be more likely to have severe outcomes.3,4,22,24 We could not estimate vaccine effectiveness among the immunocompromised population because vaccine coverage was low (1.3% were fully vaccinated). Additional research is needed to evaluate vaccine effectiveness among persons with immunocompromising conditions, and these persons may choose to take additional precautions to reduce the risk of or prevent mpox infection.25
Our findings are consistent with those of the few studies examining the effectiveness of JYNNEOS (MVA) vaccine against mpox disease. A study that examined vaccine performance with the use of surveillance data from 43 U.S. jurisdictions showed that the incidence of mpox disease was 7.4 times and 9.6 times as high among unvaccinated men who were 18 to 49 years of age and who were eligible for JYNNEOS vaccination as among those who received a first or second vaccine dose at least 14 days earlier, respectively; it is notable that this study relied on data from separate surveillance systems, which required several assumptions, and was unable to account for potential confounding factors.11 A cohort study from Israel showed that MVA vaccine was associated with an 86% decrease in the incidence of mpox disease among men receiving HIV PrEP or men with HIV who had a diagnosis of one or more STIs; however, the study examined only a single, subcutaneously administered dose, and the cohort included only 21 persons with mpox, factors that resulted in imprecise estimates.12
Abstract
Background
In the United States, more than 30,000 cases of mpox (formerly known as monkeypox) had occurred as of March 1, 2023, in an outbreak disproportionately affecting transgender persons and gay, bisexual, and other men who have sex with men. In 2019, the JYNNEOS vaccine was approved for subcutaneous administration (0.5 ml per dose) to prevent mpox infection. On August 9, 2022, an emergency use authorization was issued for intradermal administration (0.1 ml per dose); however, real-world effectiveness data are limited for either route.
Methods
We conducted a case–control study based on data from Cosmos, a nationwide Epic electronic health record (EHR) database, to assess the effectiveness of JYNNEOS vaccination in preventing medically attended mpox disease among adults. Case patients had an mpox diagnosis code or positive orthopoxvirus or mpox virus laboratory result, and control patients had an incident diagnosis of human immunodeficiency virus (HIV) infection or a new or refill order for preexposure prophylaxis against HIV infection between August 15, 2022, and November 19, 2022. Odds ratios and 95% confidence intervals were estimated from conditional logistic-regression models, adjusted for confounders; vaccine effectiveness was calculated as (1−odds ratio for vaccination in case patients vs. controls)×100.
Results
Among 2193 case patients and 8319 control patients, 25 case patients and 335 control patients received two doses (full vaccination), among whom the estimated adjusted vaccine effectiveness was 66.0% (95% confidence interval [CI], 47.4 to 78.1), and 146 case patients and 1000 control patients received one dose (partial vaccination), among whom the estimated adjusted vaccine effectiveness was 35.8% (95% CI, 22.1 to 47.1).
Conclusions
In this study using nationwide EHR data, patients with mpox were less likely to have received one or two doses of JYNNEOS vaccine than control patients. The findings suggest that JYNNEOS vaccine was effective in preventing mpox disease, and a two-dose series appeared to provide better protection. (Funded by the Centers for Disease Control and Prevention and Epic Research.)
In 2019, the Food and Drug Administration (FDA) approved JYNNEOS vaccine (modified vaccinia Ankara [MVA] vaccine, Bavarian Nordic) administered subcutaneously in a two-dose series (0.5 ml per dose, 4 weeks apart) for prevention of smallpox and mpox.5 On August 9, 2022, in order to increase the supply of vaccine available for use, an emergency use authorization (EUA) was issued for intradermal administration of the vaccine in a two-dose series (0.1 ml per dose, 4 weeks apart).6,7 Vaccination is available for persons with known or presumed exposure to a person with mpox (offered as postexposure prophylaxis) or with factors potentially increasing the likelihood of having been exposed (previously referred to as expanded postexposure prophylaxis).8,9 Vaccination may also be offered to persons at high risk for exposure to mpox virus (MPXV) or who might benefit from vaccination (PrEP for mpox), including gay, bisexual, or other men who have sex with men; transgender persons; nonbinary or gender nonconforming persons; persons with HIV; persons eligible for PrEP against HIV; or persons with a recent (past 6 months) diagnosis of HIV or other nationally reportable STI.8,9
The effectiveness of JYNNEOS vaccine against mpox disease has been inferred from animal and immunogenicity studies for both the subcutaneous10 and intradermal7 administration routes; however, real-world effectiveness data about mpox PrEP are limited.11,12 We analyzed a large, nationwide electronic health record (EHR) database in the United States to estimate the effectiveness of partial (one dose) and full (two doses) vaccination with JYNNEOS as PrEP against mpox disease overall, among select subpopulations, and according to route of administration.
|
|
| |
| |
|
|
|