| |
REPRIEVE Results published: Pitavastatin to Prevent Cardiovascular Disease in HIV Infection
|
| |
| |
Download the PDF here
July 23 2023
NEJM
Steven K. Grinspoon, M.D., Kathleen V. Fitch, M.S.N., Markella V. Zanni, M.D., Carl J. Fichtenbaum, M.D., Triin Umbleja, M.S., Judith A. Aberg, M.D., Edgar T. Overton, M.D., Carlos D. Malvestutto, M.D., M.P.H., Gerald S. Bloomfield, M.D., M.P.H., Judith S. Currier, M.D., Esteban Martinez, M.D., Ph.D., Jhoanna C. Roa, M.D., Marissa R. Diggs, B.A., Evelynne S. Fulda, B.A., Kayla Paradis, M.B.A., Stephen D. Wiviott, M.D., Borek Foldyna, M.D., Sara E. Looby, Ph.D., Patrice Desvigne Nickens, M.D.,
Beverly Alston Smith, M.D., Jorge Leon Cruz, M.S., Sara McCallum, M.P.H., Udo Hoffmann, M.D., M.P.H., Michael T. Lu, M.D., M.P.H., Heather J. Ribaudo, Ph.D., and Pamela S. Douglas, M.D., for the REPRIEVE Investigators*
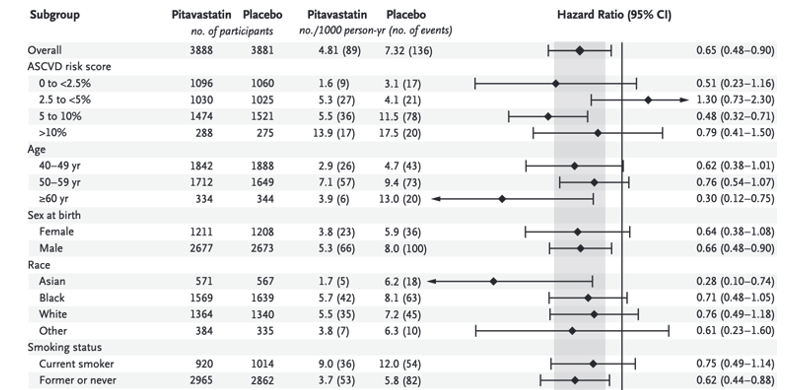
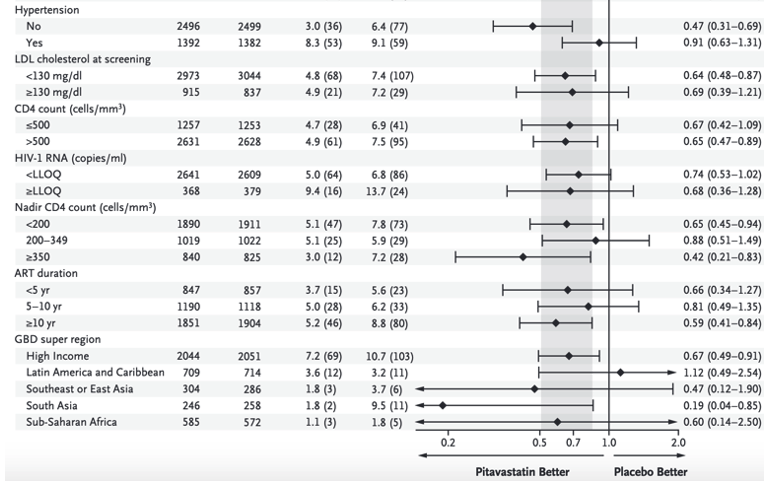
This observed reduction was larger than that predicted by the Cholesterol Treatment Trialists' Collaboration on the basis of the achieved reduction in LDL cholesterol levels.22 This finding suggests effects on cardiovascular risk beyond those associated with the lowering of LDL cholesterol alone. The degree of reduction is consistent with that observed in a study of rosuvastatin23 involving older persons (median age, 66 years) who did not have HIV infection but who had increased C-reactive protein levels without elevated LDL cholesterol levels. In REPRIEVE, the median screening LDL cholesterol level was 108 mg per deciliter, and the incidence of major adverse cardiovascular events was similar among participants in the pitavastatin group regardless of the screening LDL cholesterol level. In addition to the lowering of LDL cholesterol levels, statin therapy reduces measures of immune activation and inflammation in persons with HIV infection,9-12 which suggests potential mechanisms by which statins benefit this population beyond the effects of LDL-cholesterol lowering.
Abstract
Background
The risk of cardiovascular disease is increased among persons with human immunodeficiency virus (HIV) infection, so data regarding primary prevention strategies in this population are needed.
Methods
In this phase 3 trial, we randomly assigned 7769 participants with HIV infection with a low-to-moderate risk of cardiovascular disease who were receiving antiretroviral therapy to receive daily pitavastatin calcium (at a dose of 4 mg) or placebo. The primary outcome was the occurrence of a major adverse cardiovascular event, which was defined as a composite of cardiovascular death, myocardial infarction, hospitalization for unstable angina, stroke, transient ischemic attack, peripheral arterial ischemia, revascularization, or death from an undetermined cause.
Results
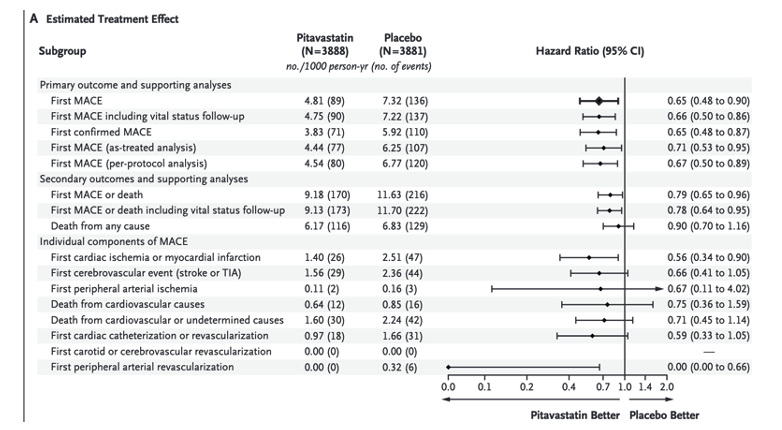
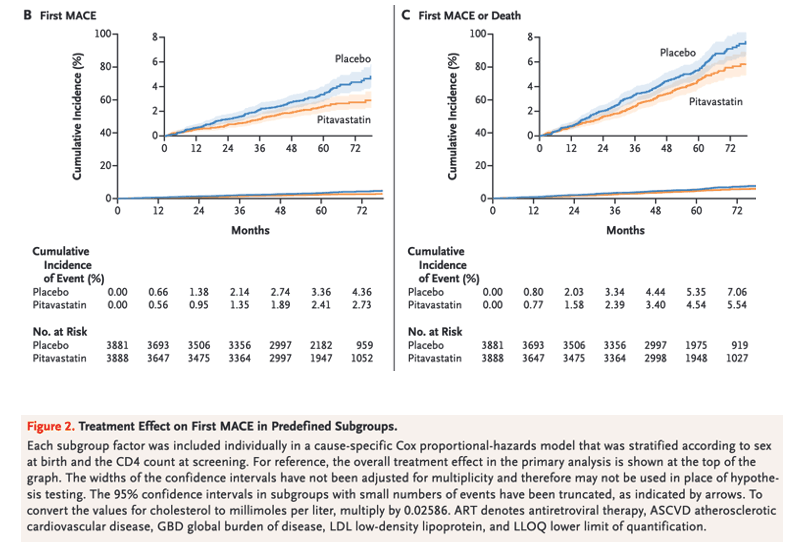
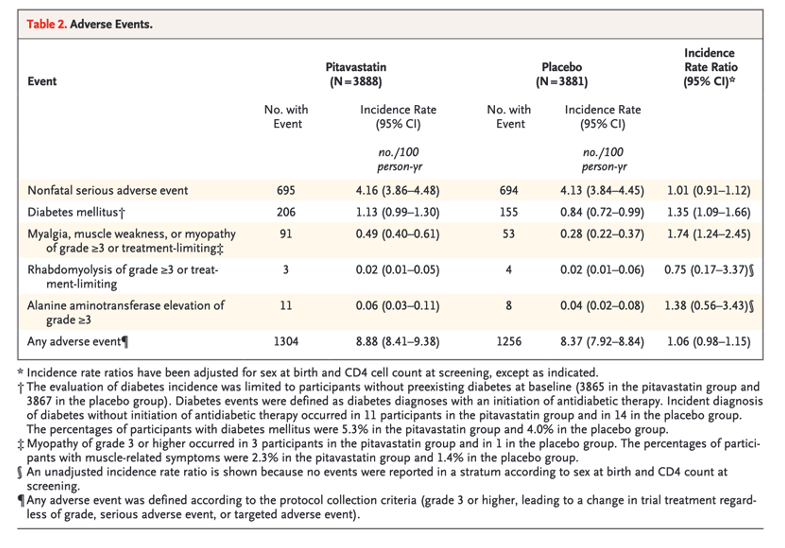
The median age of the participants was 50 years (interquartile range, 45 to 55); the median CD4 count was 621 cells per cubic millimeter (interquartile range, 448 to 827), and the HIV RNA value was below quantification in 5250 of 5997 participants (87.5%) with available data. The trial was stopped early for efficacy after a median follow-up of 5.1 years (interquartile range, 4.3 to 5.9). The incidence of a major adverse cardiovascular event was 4.81 per 1000 person-years in the pitavastatin group and 7.32 per 1000 person-years in the placebo group (hazard ratio, 0.65; 95% confidence interval [CI], 0.48 to 0.90; P=0.002). Muscle-related symptoms occurred in 91 participants (2.3%) in the pitavastatin group and in 53 (1.4%) in the placebo group; diabetes mellitus occurred in 206 participants (5.3%) and in 155 (4.0%), respectively.
Conclusions
Participants with HIV infection who received pitavastatin had a lower risk of a major adverse cardiovascular event than those who received placebo over a median follow-up of 5.1 years. (Funded by the National Institutes of Health and others; REPRIEVE ClinicalTrials.gov number, NCT02344290. opens in new tab.)

|
|
| |
| |
|
|
|