| |
A formula for the end [of HIV], Paradigm Shift by Pharma
|
| |
| |
AbbVie | phase 1: ABBV-1882 is a combination of AbbVie's budigalimab, an anti-PD1 mAb and ABBV-382, an anti-a4b7 mAb, being investigated as a functional cure for HIV.
ViiV Healthcare | phase 2: N6LS, ViiV's broadly neutralizing antibody (bNAb), is being tested in the HIV cure space.
Rockefeller University, Gilead Sciences | phase 2: Two investigational broadly neutralizing antibodies (bNAbs) - GS-5423 and GS-2872 - developed by Rockefeller University and licensed by Gilead are being evaluated for use in HIV cure strategies.
Gilead Sciences | phase 2: Lefitolimod, a toll-like receptor 9 (TLR-9) agonist being investigated as part of a functional cure for HIV by Aarhus University Hospital in collaboration with Gilead.
Gilead Sciences | phase 1b: Elipovimab is a first-in-class effector-enhanced broadly neutralizing HIV-1 antibody (bNAb) being tested towards the goal of reducing or eliminating the HIV reservoir in patients.
-------------------------------------
Now, over 40 years later, the FDA has approved more than 60 drugs in the buzzing HIV treatment and prevention space. With combination drug treatments started early, people living with HIV can not only survive but can potentially remain undetectable and unable to transmit HIV - living near-normal lifespans. Despite three isolated cases of individuals cured of HIV while undergoing invasive treatments for cancer, a scalable cure has yet to be found.
But the voices of those living with HIV are still ever-present and many engaged in the fight have united in an ambitious goal: ending the HIV/AIDS epidemic. The target year is 2030. In the U.S., there are two national plans in place to help stick to that timeline. Globally, the Joint United Nations Program on HIV/AIDS (UNAIDS) has set the same 2030 deadline.
There are many parameters being used to define the epidemic's 'end' and just as many plans to get there. But few, if any, federal or global initiatives expressly specify the need for a cure.
Pharma, however, has its own plan. Within the industry, optimism is high regarding the feasibility of ending the HIV epidemic. This race to the end has become a delicate - and at times, conflicting - balancing act between the hunt for continued innovation in a crowded treatment space and the ongoing, collaborative search for a cure. Ultimately, pharma's ability to weigh these pursuits could pay off exponentially for both the industry and people living with HIV.
The succinctness of the call to 'end HIV by 2030!' makes it highly effective as a rallying cry, but confusing as a quantitative goal. Ultimately, the objective is the end of HIV/AIDS as an epidemiologically-defined term, using certain criteria to measure progress. In the U.S., the HIV National Strategic Plan uses viral suppression as the main indicator of success. Specifically, the U.S. goal is for 86% of people living with HIV to be virally suppressed on antiretroviral treatment by 2030.
If no other factors were considered, epidemiologically, HIV could end without a cure. Given how lucrative the HIV treatment space has been for pharma companies (the industry's top-selling treatment, Gilead's single-tablet blockbuster, Biktarvy, is predicted to achieve sales of around $11 billion by 2025) it may seem contradictory that a company would pursue something - in this case, a cure - that could, in theory, make treatments obsolete.
But taking into consideration the vast amount of human lives affected, the myriad variables that come into play in the complicated HIV treatment landscape, and the commitment to end the epidemic by 2030, many pharma companies active in the HIV market have made the hunt for a cure a dedicated part of their epidemic-ending plans.
Pharma's approach to ending the epidemic has evolved over years of observing the patient experience, and that evolution has brought the industry closer to a cure
Davinderpreet Singh Mangat, Informa Pharma Intelligence senior analyst with a focus on infectious disease research says a change in treatment paradigms is already underway.
"There will likely be a continued shift towards developing long-acting regimens and that will become the new normal in 5-10 years' time," says Mangat.
"The need for lifelong treatment of HIV with antiretroviral therapy poses multiple challenges for people living with HIV (PLWH)," says Brian Plummer, a spokesperson for Gilead Sciences.
PLWH often struggle with treatment fatigue or pill burden. Side effects from lifelong antiretroviral therapy (ART) regimens can include weight gain, muscle pain, nausea and insomnia. Living with HIV also means confronting numerous social and psychological challenges.
Despite the long-standing buzz surrounding the hunt for a cure, quantifying that interest - especially from the pharma industry - is challenging.
Leaders in the pharma space have backed their commitments to a cure with large amounts of funding initiatives and philanthropic endeavors.
The next major breakthroughs will be about providing options that allow people to go even longer between their HIV treatments, like medicines administered twice or even once a year," says Smith.{Kim at ViiV]
These treatment innovations have also helped nudge pharma closer to a cure. Functional cures - a scenario where treatments trigger long periods of viral remission in the absence of ART - could advance progress a step further than current long-acting treatments, potentially allowing people to go years between taking medicine.
Additionally, while the scientific advances made in the HIV space have fortunately led to substantial improvements in the life expectancy of people living with HIV, this too comes with challenges and a new set of considerations.
"The corollary of a longer lifespan is that PLWH are now faced with an increased risk of developing comorbidities and chronic diseases associated with aging, in addition to chronic HIV. Chronic comorbidities may also contribute to a greater pill burden and additional complications, which may negatively impact adherence to HIV therapy," says Plummer. "Achieving a cure would have a dramatic impact on the challenges associated with lifelong treatment and the social stigma associated with HIV and is key to accomplishing the shared goal of ending the HIV epidemic."
But lacking immediate prospects for a cure, the pharma industry has looked to address unmet patient needs with innovative, more convenient treatment solutions.
According to Kimberly Smith, M.D., MPH, head of R&D for ViiV Healthcare, "For the past decade, it felt like we had reached a plateau in how we treated HIV with once-daily oral HIV medicine. Once-daily oral medicine has been and remains the bedrock of our approach to treatment, but for many individuals, it can be challenging and to continue our progress towards ending the epidemic, we must look for innovative options beyond daily medicine."
https://www.pharmamanufacturing.com/development/clinical-trials/article/11288765/hiv-an-ending-in-the-balance

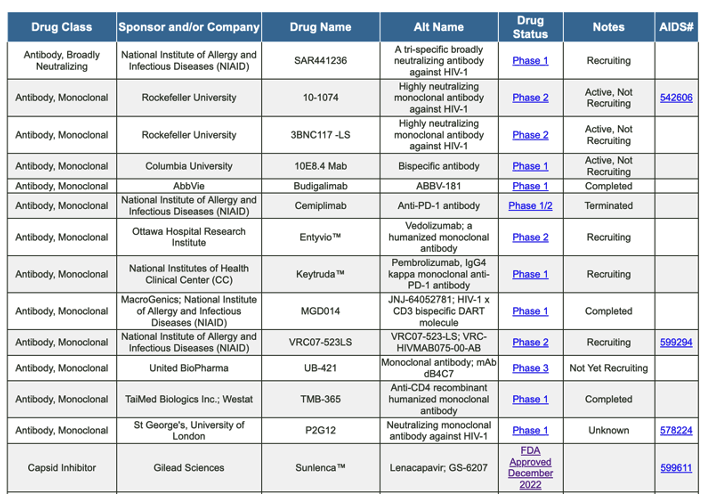
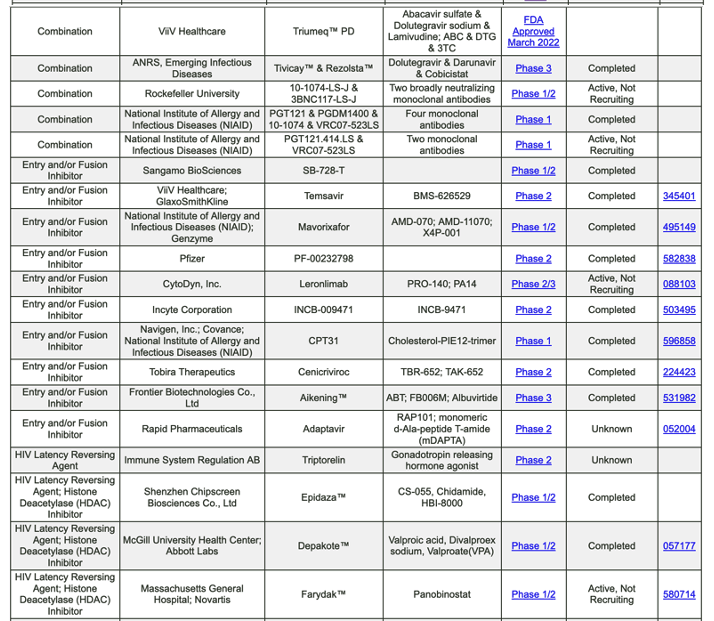
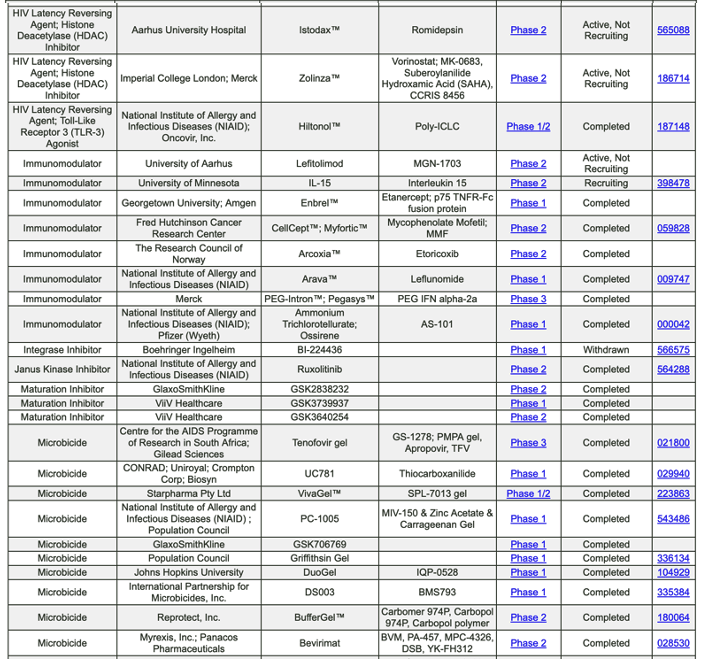
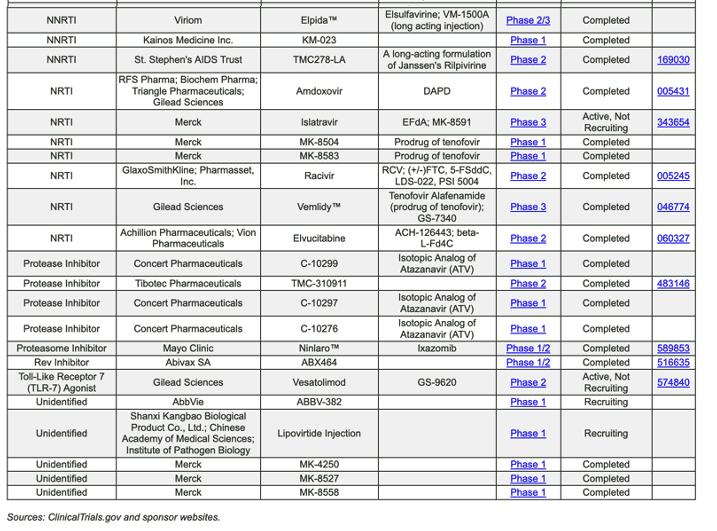
https://chemdb.niaid.nih.gov/DrugDevelopmentHIV.aspx

|
|
| |
| |
|
|
|