 |
 |
 |
| |
JAK Inhibitor Ruxolitinib Reverses Immune Dysfunction and Depletes HIV Reservoir
|
| |
| |
IAS 2023, July 23-26, 2023. Brisbane
Mark Mascolini
Ruxolitinib, a JAK inhibitor sometimes identified as a tyrosine kinase inhibitor, appeared to reduce immune dysfunction in a phase 2a trial that enrolled 60 people with continuous HIV suppression on antiretroviral therapy (ART) [1]. The 12-week trial also yielded evidence of near clearance of the peripheral HIV reservoir in people who entered the trial with a deep viral reservoir.
Researchers pursue agents that can help pull the plug of otherwise impregnable HIV reservoirs. A group at Atlanta's Emory University, with colleagues from other centers, proposed that one ruxolitinib mechanism for reducing the reservoir is reversing the immune dysfunction that prevents HIV cure.
Ruxolitinib has won approval for treatment of myelofibrosis (bone marrow cancer), polycythemia vera (abnormally increased blood cells levels), and steroid-refractory graph-versus-host disease [2]. A group led by Emory's Christina Gavegnano studies ruxolitinib and other JAK inhibitors for HIV infection. They believe JAK inhibitors are good candidates for HIV control because the JAK STAT pathway largely drives chronic inflammation.
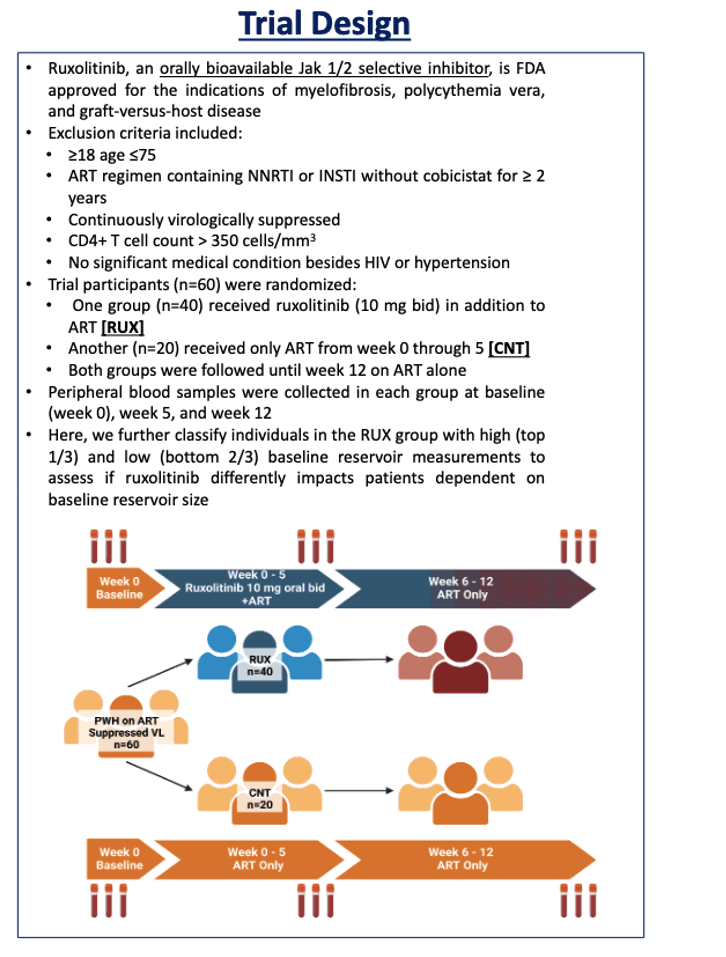
The Emory team randomized 40 people with HIV to ruxolitinib at an oral dose of 10 mg twice daily plus antiretrovirals for 5 weeks or to antiretroviral therapy alone for 5 weeks. From week 5 through 12 participants in both study arms took only antiretroviral therapy. Participants had to be between 18 and 75 years old with a CD4 count above 350 and continuous viral control with a combination built on a nonnucleoside or an integrase inhibitor without cobicistat. Researchers stratified participants receiving ruxolitinib into a high-reservoir group (top one third) and a low-reservoir group (bottom two thirds). They measured markers of interest in blood samples collected at weeks 0, 5, and 12.
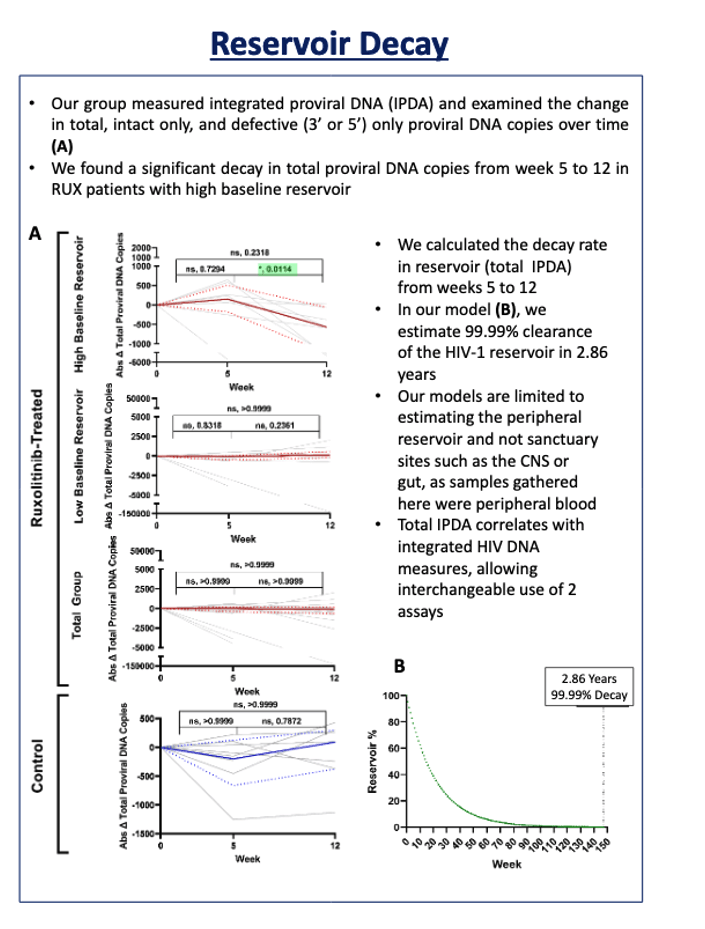
Total proviral DNA in blood samples fell significantly from week 5 to 12 in people taking ruxolitinib in the high pretreatment reservoir group (P = 0.0114) but not in the low-reservoir group. The researchers' statistical model estimated 99.99% clearance of the peripheral HIV reservoir in 2.86 years.
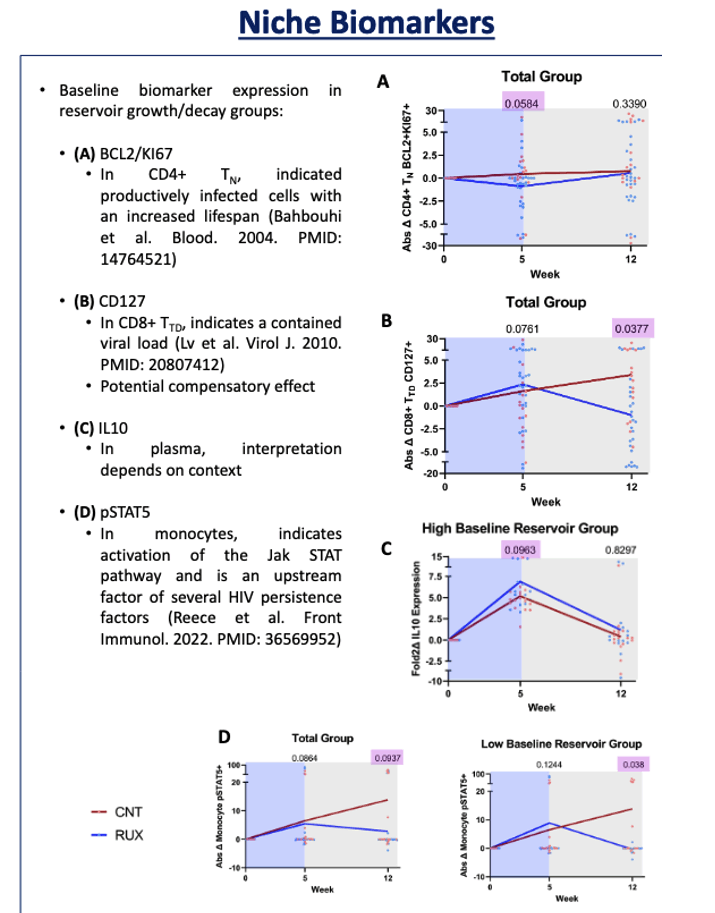
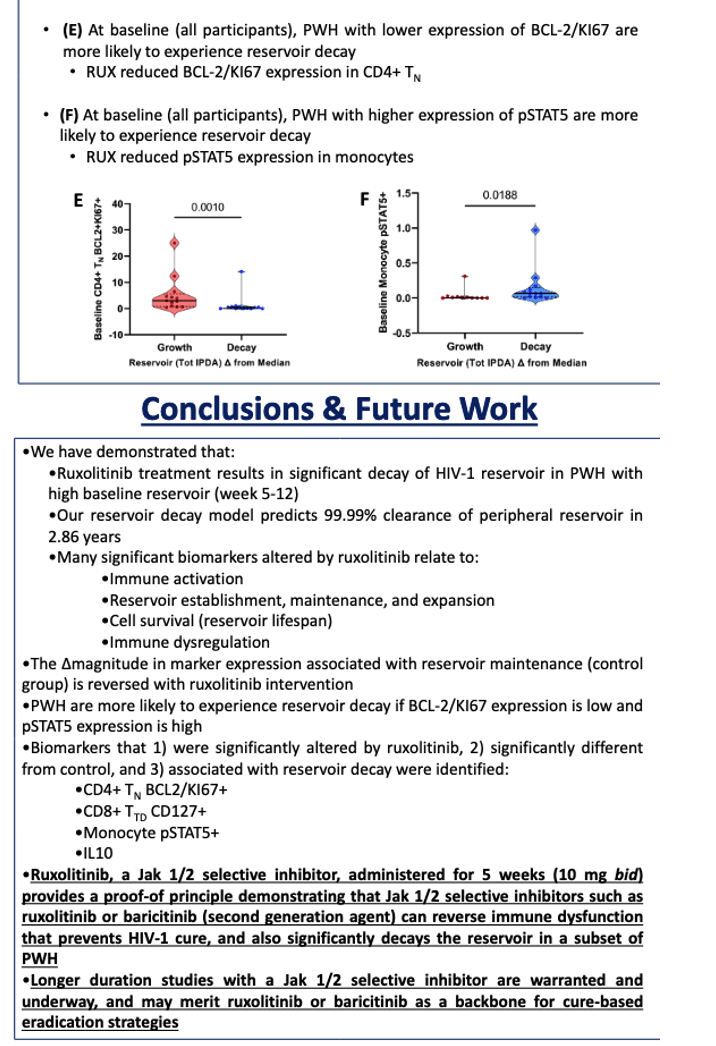
Four biomarkers were significantly altered by ruxolitinib, had levels significantly different from control levels, and were associated with reservoir decay: CD4 TN BCL2/Kl67, CD8 TTD CD127, monocyte pSTAT5, and IL-10. IL-10 emerged as a correlate of reservoir decay only in the high baseline reservoir group, pSTAT5 in the total and low baseline reservoir groups, and the other two markers in the total group. Other studies show that these four markers are associated with immune activation, reservoir establishment, maintenance, and expansion, cell survival (reservoir lifespan), and immune dysregulation.
A case study presented at IAS 2023 and reviewed separately by NATAP documented no viral rebound for 18 months without antiretrovirals after pretreatment with ruxolitinib followed by allogeneic hematopoietic stem cell transplantation with wild-type CCR5 donor cells to treat a biphenotypic sarcoma [3].
The Emory group proposed that longer studies of ruxolitinib or a second-generation agent, baricitinib, could position one of these JAK inhibitors as a backbone for HIV cure therapy.
References
1. Reece MD, Zhang Z, Pereira-Ribeiro S, et al. Ruxolitinib-mediated HIV-1 reservoir decay in A5336 phase 2a trial. IAS 2023, July 23-26, 2023. Brisbane.
2. National Library of Medicine. StatPearls. Ruxolitinib. https://www.ncbi.nlm.nih.gov/books/NBK570600/
3. Saez-Cirion A, et al. Absence of viral rebound for 18 months without antiretrovirals after allogeneic hematopoietic stem cell transplantation with wild-type CCR5 donor cells to treat a biphenotypic sarcoma. IAS 2023, July 23-26, 2023. Brisbane.



|
| |
|
 |
 |
|
|