 |
 |
 |
| |
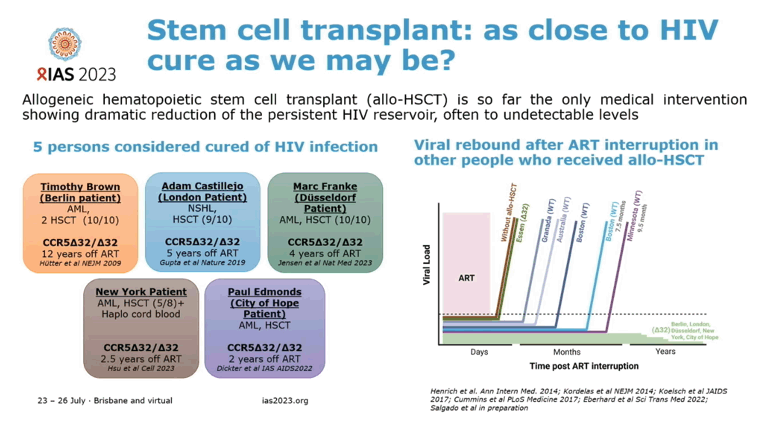
No Rebound 20 Months After ART Stops After Novel Stem Cell Transplant
|
| |
| |
IAS 2023, July 23-26, 2023. Brisbane
Mark Mascolini
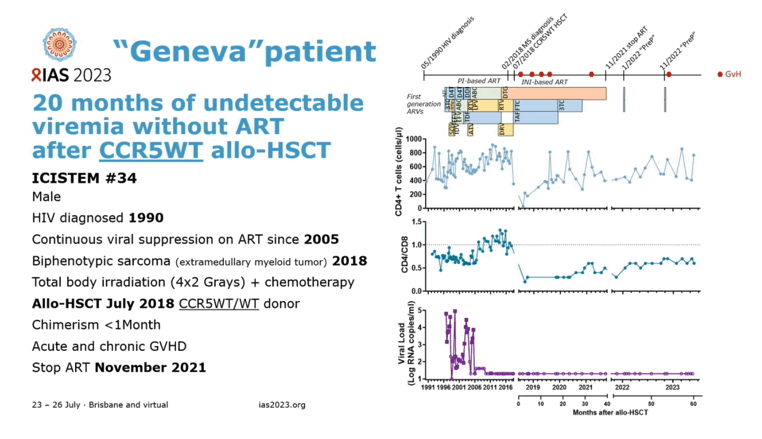
A Geneva man with HIV and sarcoma joined the handful of people who maintained apparent HIV clearance without antiretroviral therapy (ART) after allogeneic hematopoietic stem cell transplant (aHSCT) [1]. But the so-called Geneva patient differs from his aHSCT colleagues in an important way: His stem cell donor lacked the delta-32 deletion in the gene coding for HIV's CCR5 coreceptor. For the first time it appears that someone not homozygous for the delta-32 change (inheriting identical versions of a gene from each parent) can donate stem cells that may help rid a stem-cell transplant recipient's body of HIV for good.
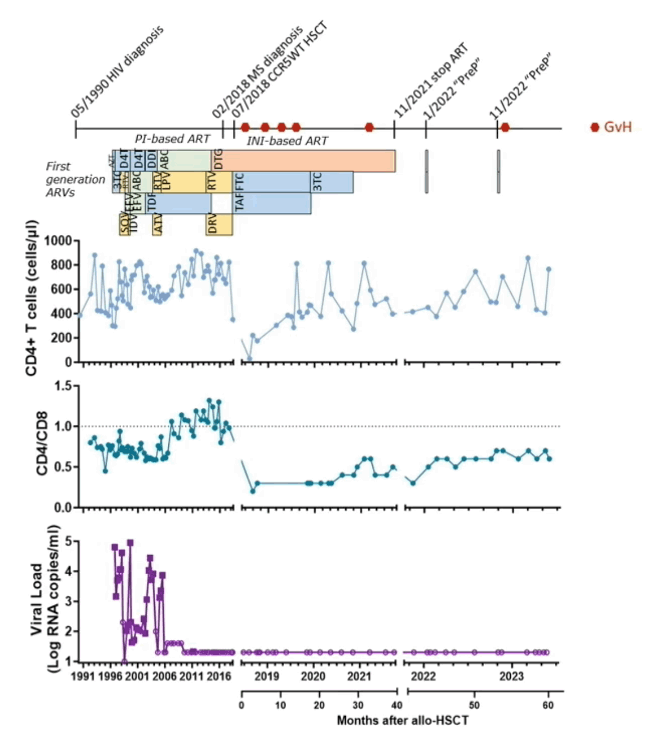
Diagnosed with HIV infection in 1990, the Geneva man maintained an undetectable viral load with ART since 2005. His physicians treated biphenotypic sarcoma diagnosed in 2018 with chemotherapy and total body irradiation followed by aHSCT in July 2018. The stem cells came from a person whose CCR5-encoding gene was wild-type, not the much rarer CCR5 delta-32 mutant that protects cells from HIV infection via the CCR5 coreceptor.
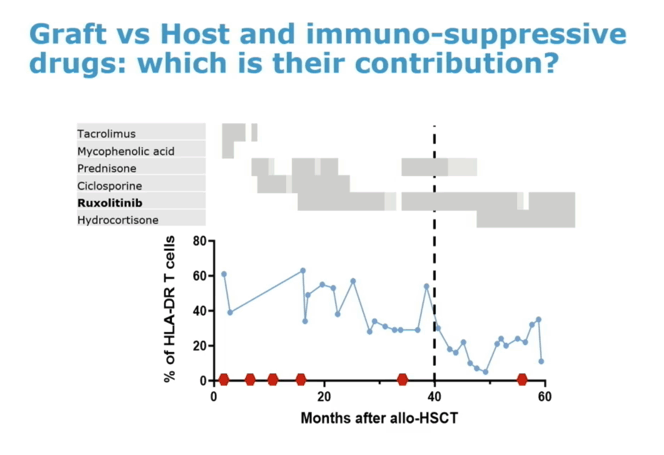
Then the Geneva man endured bouts of graft-versus-host disease, the first arising soon after aHSCT and treated with corticosteroids and immunosuppressant calcineurin inhibitors. Graft-versus-host disease recurred 8 months after aHSCT, which his clinicians addressed with a course of the JAK inhibitor ruxolitinib. After treatment ended, graft-versus-host disease relapsed and ruxolitinib had to be restarted. ART stopped completely in November 2021.
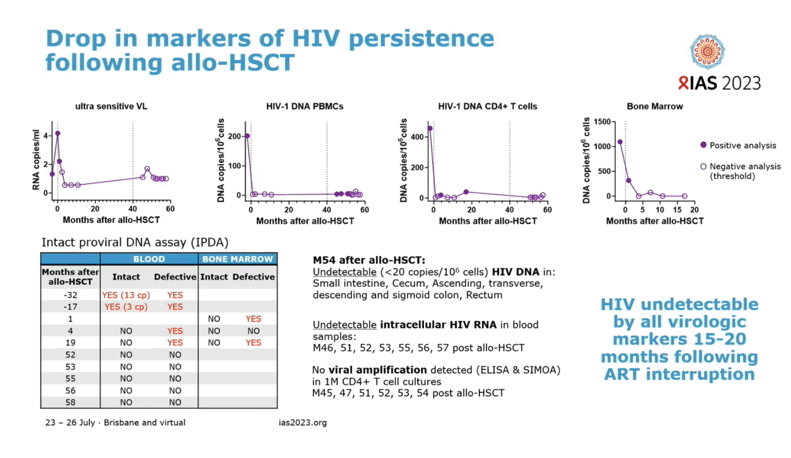
Before aHSCT an ultrasensitive HIV RNA assay detected 4 copies per milliliter in blood, and HIV DNA could be detected at 457 copies per million CD4 cells in blood and at 1096 copies per million CD4 cells in bone marrow. After aHSCT a standard viral load assay could not spot HIV RNA in the circulation or in blood cells, and RNA also became undetectable with an ultrasensitive assay. Proviral DNA, the viral genome stitched into chromosomes of infected cells, could be detected sporadically at low levels (4 to 40 copies per million cells) after aHSCT. Further analysis indicated this was probably defective, not intact, HIV DNA. Fifty-four months after aHSCT, an assay that can measure as few as 20 HIV DNA copies did not detect DNA in samples from the small intestine, cecum, or ascending, transverse, descending, or sigmoid colon.
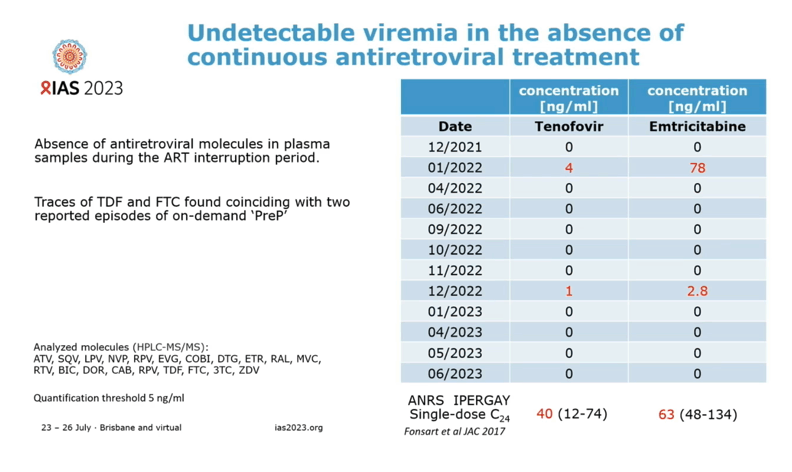
In lab studies researchers could not amplify virus from stimulated CD4 cells collected from the Geneva man after ART stopped. But these cells remained susceptible to infection with HIV in lab dishes. Antiretrovirals could not be detected in blood plasma after ART stopped, except for traces of tenofovir and emtricitabine when this man used them for preexposure prophylaxis (PrEP) 2 and 12 months after stopping ART to treat HIV infection.
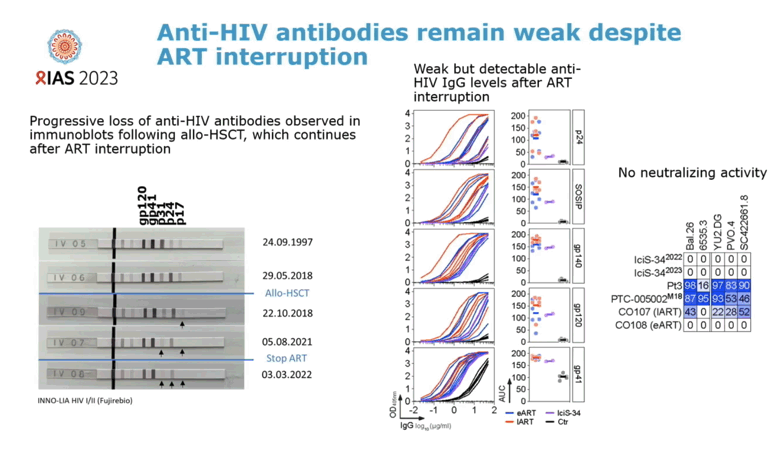
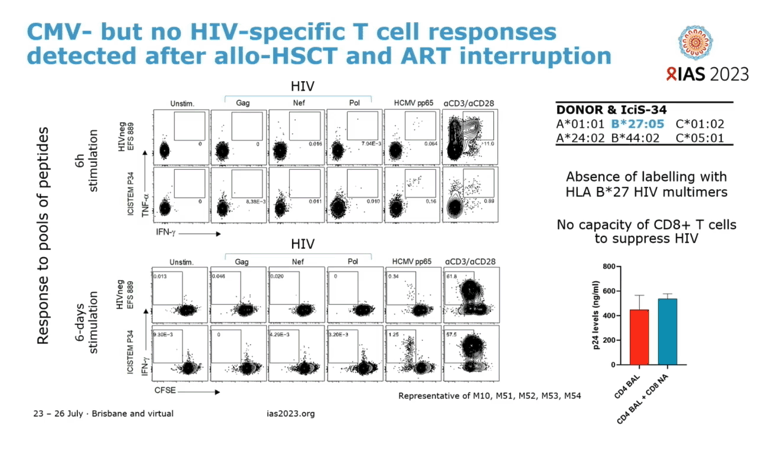
After aHSCT, immunoblots traced progressive loss of anti-HIV antibodies. Anti-HIV IgG remained weakly detectable after ART stopped. Cytomegalovirus-specific T-cell responses could be detected after aHSCT and ART interruption, but HIV-specific T-cell responses could not.
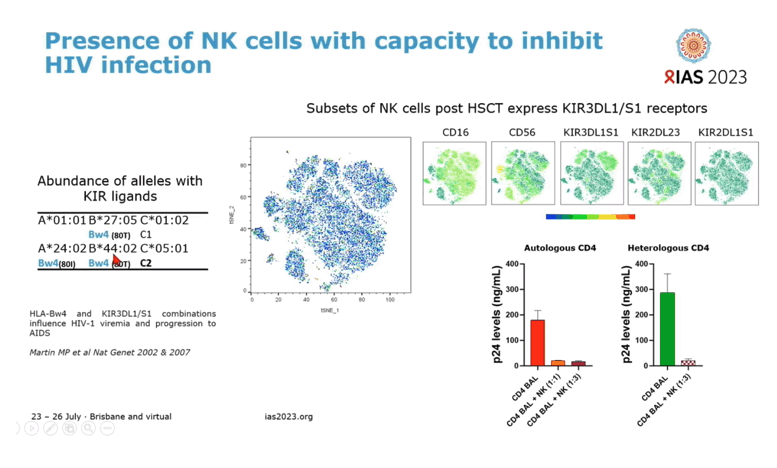
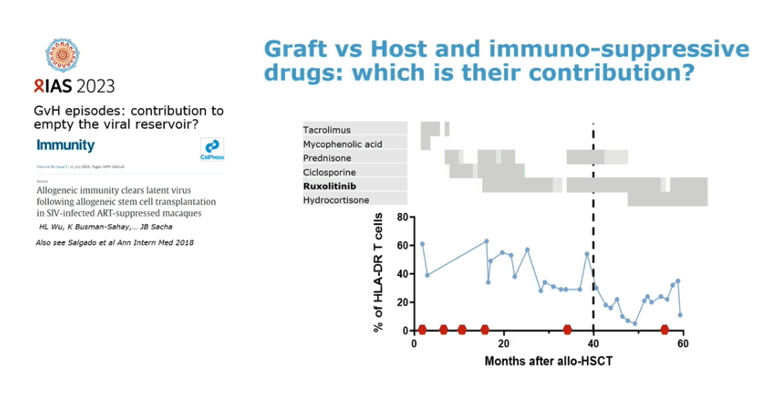
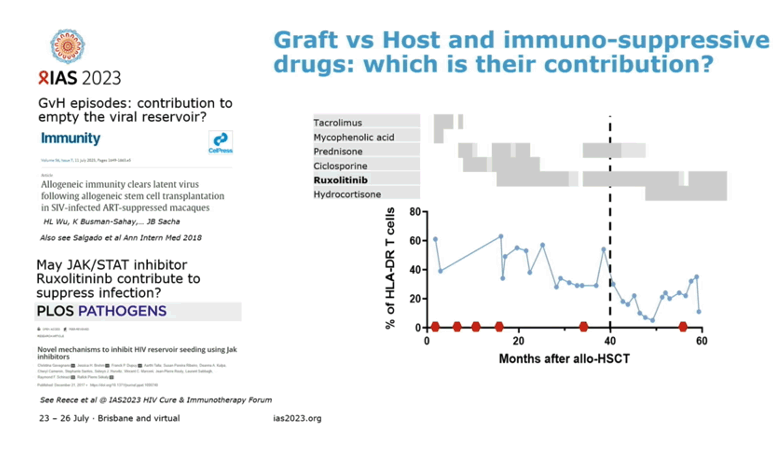
Collaborators from the Pasteur Institute in Paris, Geneva University Hospitals, and other centers speculated that immunosuppressive drugs given to prepare a person for aHSCT, graft-versus-host disease after the transplant [2], and the JAK inhibitor ruxolitinib [3] may all contribute to suppression of HIV without ART in this individual. Whatever the contributors, this case suggests that stem cells from a donor with common wild-type CCR5 (without the protective delta-32 deletion) can lead to HIV remission after aHSCT. The researchers cautioned, though, that they cannot exclude the possibility that HIV may still lurk in "anatomical or cellular sanctuaries" in this Geneva man.
In another study presented at IAS 2023 and reviewed separately by NATAP, ruxolitinib appeared to promote clearance of the peripheral blood HIV reservoir in people with long-term viral suppression [4].
References
1. Sáez-Cirión A, Mamez A-C, Avettand-Fenoel V, et al. Absence of viral rebound for 18 months without antiretrovirals after allogeneic hematopoietic stem cell transplantation with wild-type CCR5 donor cells to treat a biphenotypic sarcoma. IAS 2023, July 23-26, 2023. Brisbane.
2. Wu HL, Busman-Sahay K, Weber WC, et al. Allogeneic immunity clears latent virus following allogeneic stem cell transplantation in SIV-infected ART-suppressed macaques. Immunity. 2023;56:1649-1663.e5. doi: 10.1016/j.immuni.2023.04.019. https://pubmed.ncbi.nlm.nih.gov/37236188/
3. Gavegnano C, Brehm JH, Franck P Dupuy FP, et al. Novel mechanisms to inhibit HIV reservoir seeding using Jak inhibitors. PLoS Pathog. 2017;13:e1006740. doi: 10.1371/journal.ppat.1006740.
https://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1006740
4. Reece MD, Zhang Z, Pereira-Ribeiro S, et al. Ruxolitinib-mediated HIV-1 reservoir decay in A5336 phase 2a trial. IAS 2023, July 23-26, 2023. Brisbane.




|
| |
|
 |
 |
|
|