 |
 |
 |
| |
Tesamorelin reduces visceral tissue and liver fat in INSTI-treated persons with HIV
|
| |
| |
Tesamorelin Trims Visceral and Liver Fat in People Taking an Integrase Inhibitor
IDWeek 2023, October 11-15, 2023, Boston

Mark Mascolini
Tesamorelin, licensed to treat visceral adiposity in people with HIV, lowered visceral and liver fat levels in HIV-positive people taking an integrase inhibitor (INSTI) regimen, according to results of a 52-week randomized placebo-controlled trial [1].
Researchers from Massachusetts General Hospital noted that fat composition has not been closely examined in people who gain weight during integrase inhibitor therapy, but experts recognize visceral adipose tissue (VAT) buildup as a contributor to weight gain during antiretroviral therapy. And excess VAT is linked to comorbidities like metabolic-associated steatotic liver disease (MASLD).
A placebo-controlled trial found that tesamorelin, a growth hormone-releasing hormone analog, cut VAT 15% in 26 weeks in antiretroviral-treated people with excess VAT [2]. But that 2008 study took place before the arrival of INSTIs, so the impact of tesamorelin on VAT in people taking these antiretrovirals remained unknown.
The Massachusetts General team leveraged data from a phase 2 double-blind trial in people with HIV and nonalcoholic fatty liver disease (NAFLD) randomized to 2 mg of tesamorelin daily for 52 weeks or placebo [3]. Analysis of people taking an integrase inhibitor during the trial yielded 30 people randomized to tesamorelin and 31 randomized to placebo. Participants were 18 to 70 years old, had an hepatic fat fraction of 5% or greater, took a stable antiretroviral regimen for at least 3 months, and took stable metabolic medications for at least 3 months. The researchers compared body composition and hepatic fat fraction measures at baseline and 12 months by magnetic resonance imaging/spectroscopy (MRI/MRS).
Baseline demographic, HIV-related, and body-composition variables were similar in people taking an INSTI combination in the original trial and in those taking a non-INSTI regimen: average age 54 and 51, proportion of men 74.4% and 86.4%, HIV duration 17 and 16 years, CD4 count 754 and 785, weight 92.4 and 96.5 kg, body mass index 31.1 and 32.1 kg/m2, waist circumference 110 and 111 cm, VAT 235 and 251 cm2, and hepatic fat fraction 13.5% and 14.4%.
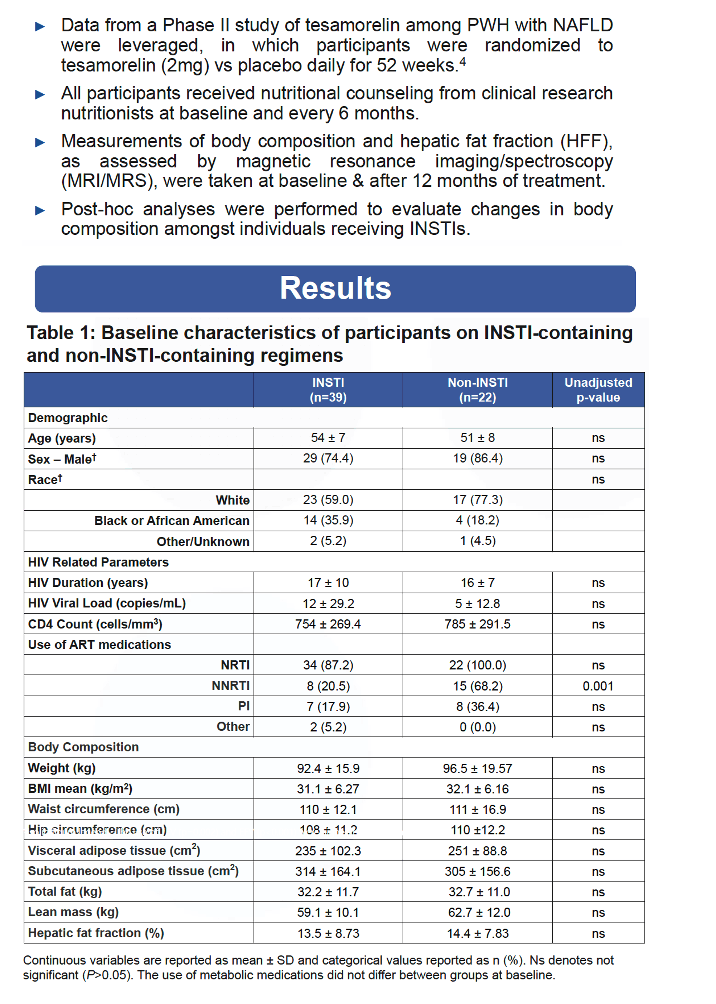
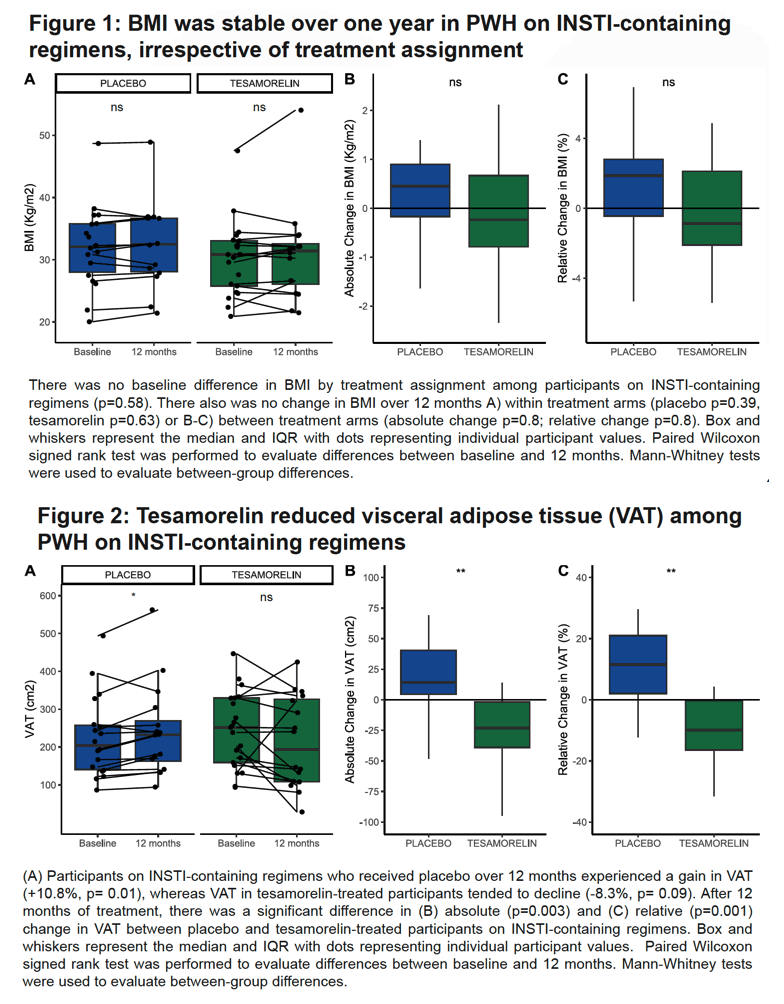
At baseline BMI in people taking an INSTI regimen did not differ by assignment to tesamorelin or placebo. And BMI did not change significantly through 12 months either within or between treatment arms. INSTI takers randomized to placebo had a significant 12-month increase in VAT (+10.8%, P = 0.01), whereas VAT tended to fall in the tesamorelin group (-8.3%, P = 0.09). At 12 months the placebo and tesamorelin groups differed significantly in both absolute (P = 0.003) and relative (P = 0.001) VAT change.
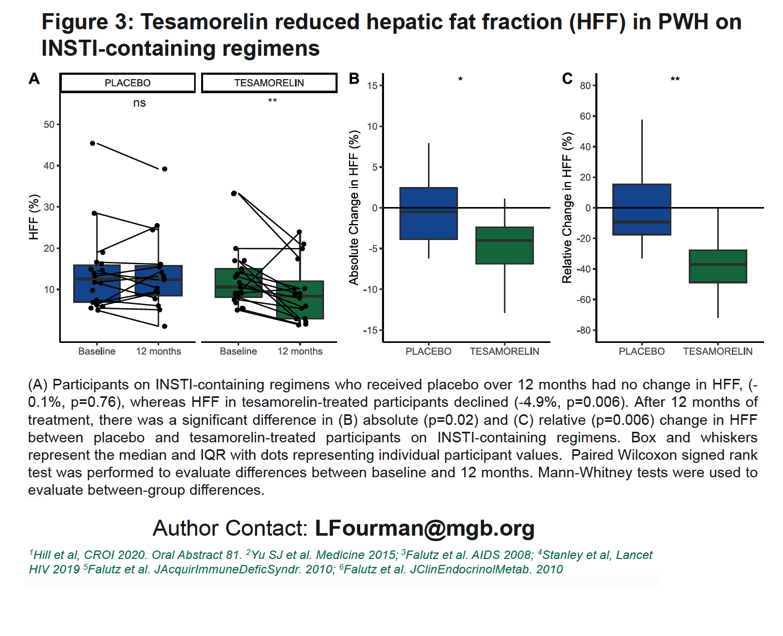
Hepatic fat fraction did not change over 12 months in INSTI takers assigned to placebo (-0.1%, P = 0.76), while that fat measure dropped significantly in people taking tesamorelin (-4.9%, P = 0.006). Again, at 12 months both absolute (P = 0.02) and relative (P = 0.006) change in hepatic fat fraction differed significantly between the tesamorelin and placebo groups.
The researchers concluded that tesamorelin lowered both visceral and liver fat over 12 months in people taking an INSTI-based regimen. In contrast, the INSTI-treated group getting placebo had VAT gains through 12 months, even though their BMI did not change.
References
1. McLaughlin T, Stanley TL, Fourman LT, Grinspoon S. Tesamorelin reduces visceral tissue and liver fat in INSTI-treated persons with HIV. IDWeek 2023, October 11-15, 2023, Boston.
2. Falutz J, Mamputu JC, Potvin D, et al. Effects of tesamorelin (TH9507), a growth hormone-releasing factor analog, in human immunodeficiency virus-infected patients with excess abdominal fat: a pooled analysis of two multicenter, double-blind placebo-controlled phase 3 trials with safety extension data. J Clin Endocrinol Metab. 2010;95:4291-4304. doi: 10.1210/jc.2010-0490. https://academic.oup.com/jcem/article/95/9/4291/2835394
3. Stanley TL, Fourman LT, Feldpausch MN, et al. Effects of tesamorelin on non-alcoholic fatty liver disease in HIV: a randomised, double-blind, multicentre trial. Lancet HIV. 2019;6:e821-e830. doi: 10.1016/S2352-3018(19)30338-8. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6981288/

|
| |
|
 |
 |
|
|