 |
 |
 |
| |
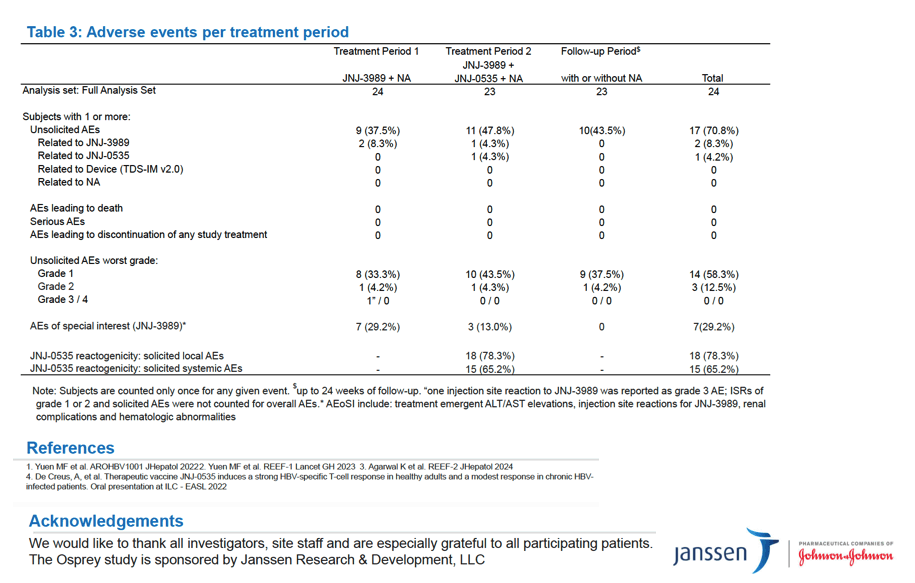
Efficacy, safety, tolerability, and immunogenicity of JNJ-0535 following a reduction of viral antigen levels through administration of siRNA JNJ-3989 in patients with chronic HBeAg negative hepatitis B - interim data of the OSPREY study
|
| |
| |
EASL 2024 June 5-8 Milan Italy
Late Breaker poster
Stefan Bourgeois1, Maria Buti2, Edward J. Gane3, Kosh Agarwal4, Ewa Janczewska5; Yao-Chun (Holden) Hsu6, Maurizia Brunetto7, Pietro Lampertico8, Joaquin Cabezas9, Gloria Kim10,
Leen Slaets11, Adam Bakala11, Joris Vandenbossche11, Simon Verheijden11, Bart Fevery11, An De Creus11, Michael Biermer11, Dessislava Dimitrova12


|
| |
|
 |
 |
|
|