 |
 |
 |
| |
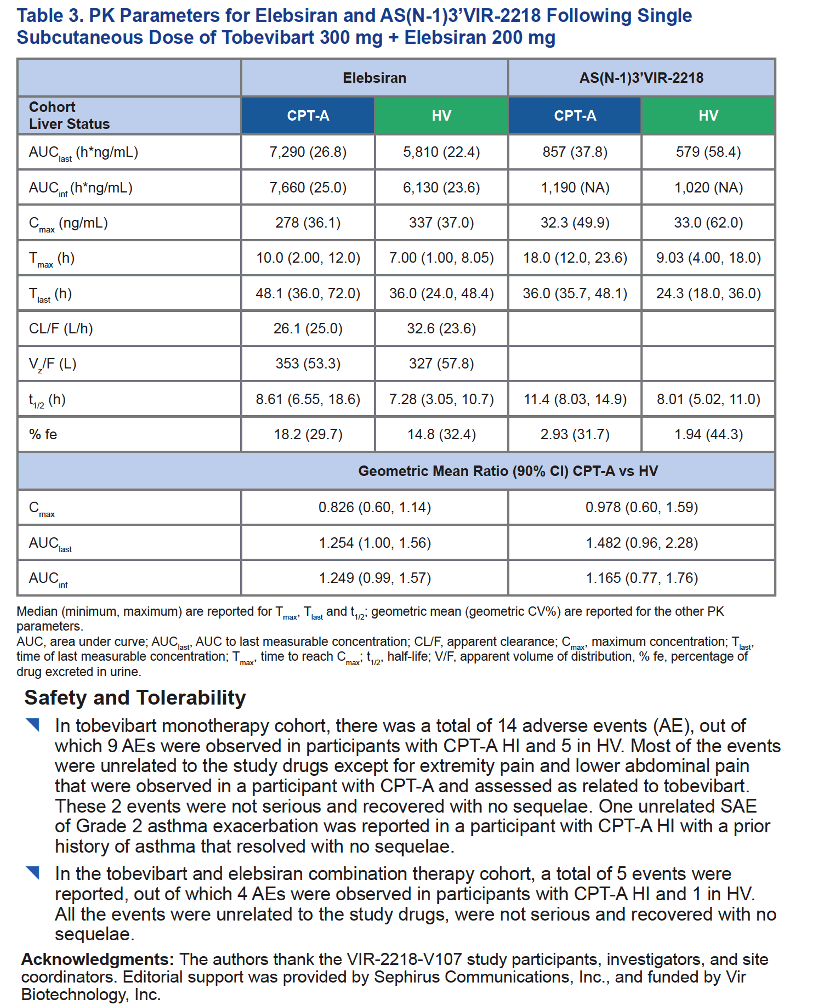
Pharmacokinetics and Safety of Single Dose of the Monoclonal Antibody Tobevibart (VIR-3434) Administered as Monotherapy or in Combination with the Small Interfering RNA Elebsiran (VIR-2218) in Cirrhotic Participants with Mild Hepatic Impairment
|
| |
| |
EASL (European Association for the Study of the Liver), 5-8 June, 2024, Milan, Italy
Li Wang, Sneha V. Gupta, Michael A. Chattergoon, Asma El-zailik, Cheng Huang, George Hristopoulos, Maribel Reyes
Vir Biotechnology, Inc., San Francisco, CA



|
| |
|
 |
 |
|
|