| |
EuroHIV-HepMtg2024: Acute Hepatitis B Infections after Reducing ART among Vaccinated Patients Living with HIV: HBV "PrEP-effect" of TDF/FTC?
|
| |
| |
European Meeting on HIV & Hepatitis 2024 | Treatment Strategies & Antiiral Drug Resistance
Barcelona, Spain | 22 - 24 May 2024
Download the PDF
Todesco E1, Valantin M2, Akhavan S1, Wirden M1, Passe-Coutrin P2, SouliE C1, Abdi B1, Cocherie T1, Calvez V1, Tubiana R2, Palich R2, Martinez V2, Marcelin A1, Seang S2
1Sorbonne UniversitE, INSERM, Institut Pierre Louis d'EpidEmiologie et de SantE Publique (iPLESP), Assistance Publique-Hopitaux de Paris (AP-HP), Hopital PitiE-Salpêtrière, Service de Virologie, Paris, France, 2Sorbonne UniversitE, INSERM, Institut Pierre Louis d'EpidEmiologie et de SantE Publique (iPLESP), Assistance Publique-Hopitaux de Paris (AP-HP), Hopital PitiE-Salpêtrière, Infectious Diseases department, Paris, France
ABSTRACT
Background: In order to improve quality of life and lessen ART burden and toxicity, reduced ART approaches have become a significant part of HIV therapeutic guidelines. In such strategies, one of the options is to remove one drug that may have a dual anti-HBV/HIV activity such as Tenofovir disoproxil (TDF) and Emtricitabine (FTC). We report here some clinical cases of acute hepatitis B (HBV) infections after withdrawal of TDF/ FTC among HBV-vaccinated patients living with HIV.
Materiel and Methods: This descriptive case-report study included patients living with HIV and followed in Infectious Diseases Department of PitiE-Salpètrière hospital (Paris, France). Clinical and socio-demographic data were obtained from patient's medical records. HBV serologies were performed by Alinity® (Abbott, Chicago, IL, USA) and viral loads by Cobas 6800® devices (Roche Diagnostics, Bâle, Switzerland).
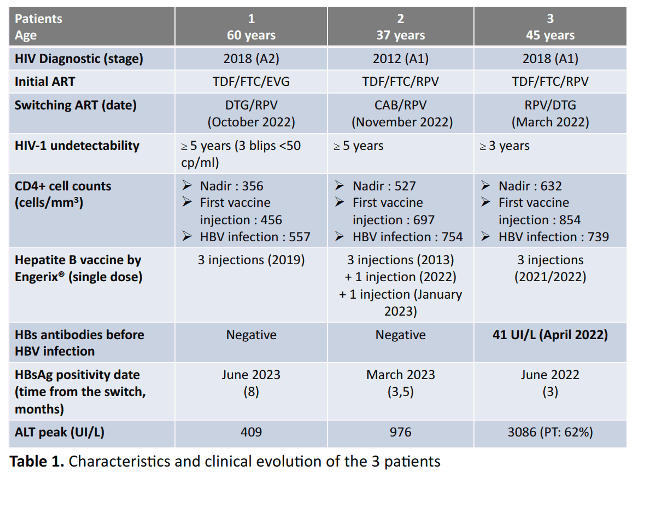
Results: Three male patients living with HIV, HBV-vaccinated and virologically suppressed were switched from a TDF/FTC-based regimen
(+Rilpivirine or Elvitegravir) to a TDF-free dual therapy (Rilpivirine+Dolutegravir or Cabotegravir). After 3, 3.5 and 8 months respectively, they suffered an acute HBV infection (positive HBs antigen, anti-HBc IgM and HBV viral loads above 6 log10IU/ml) that required hospitalization for one of them (Prothrombin rate: 62%). FTC/TDF 200/245mg/d or Entecavir 1mg/d were introduced in order to treat the HBV infection, with a dramatic decrease of HBV viral loads for the three patients and a positive clinical evolution.
At the time of HBV diagnosis, HIV stages of infection were A1 (n=2) or A2, CD4 cell counts were 754, 739 and 557/mm3 and ALT titers were 917, 3086 and 409 UI/L, respectively. Nadir of CD4 cell counts were 527, 632 and 356/mm3.
HBs antigen and anti-HBc antibodies were in all 3 cases negative before the ART switch. All of them had received HBV vaccine before the ART switch. One patient displayed an anti-HBs antibodies (HBsAb) titer of 41UI/L a few months before acute infection but HBV sequencing did not reveal any immune escape substitution. For the two other patients, no protective HBsAb were observed before and up to 7 and 9 months after the HBV infection (<10UI/L). Risk factors for HBV infection were condomless sex, and use of IV drugs for two of the patients.
Conclusions: Humoral response to HBV vaccine is known to be impaired among patients living with HIV and should be checked after the schedule of vaccination, even for patients with high CD4 cell counts. Moreover, cellular immune responses need to be further investigated in people living with HIV. Indeed, these three cases of acute HBV infection addressed several questions about the protective correlate for HBV among people living with HIV and also about the HBV "PrEP-effect" of TDF/FTC. Withdrawal of an active anti-HBV drug should be considered with caution in case of risky behavior towards sexually transmitted infections and absence or low immune response to HBV vaccine.
|
|
| |
| |
|
|
|