 |
 |
 |
| |
The DoxyPEP Debate: To DoxyPEP or not to DoxyPEP?
|
| |
| |


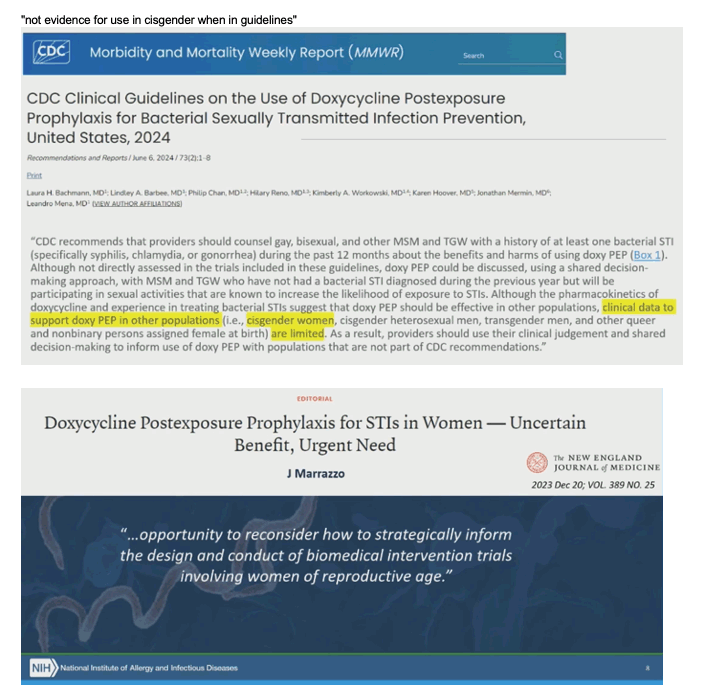
JAMA March 2024, J Marrazzo. HIV Preexposure Prophylaxis With Emtricitabine and Tenofovir Disoproxil Fumarate Among Cisgender Women
None of the 498 women with consistently daily adherence acquired HIV. Only 1 of the 658 women with consistently high adherence acquired HIV (incidence rate, 0.13/100 person-years [95% CI, 0.02-0.92]).
A total of 12 incident HIV infections were observed among the 2955 participants with adherence data, compared with 20 diagnoses among 3341 participants without adherence data. None of the 498 participants with consistently daily adherence by group-based trajectory acquired HIV (Figure 4); because there were no cases of infection, an IRR could not be calculated for this group. Only 1 of the 658 participants with consistently high adherence (4-6 doses/week) acquired HIV
NEJM Editorial. December 20, 2023, J Marrazzo - Doxycycline Postexposure Prophylaxis for STIs in Women — Uncertain Benefit, Urgent Need
In 2012, the Food and Drug Administration approved Truvada (emtricitabine–tenofovir disoproxil fumarate) as HIV PrEP for adults at increased risk for HIV infection. Shortly after, the DISCOVER trial involving cisgender men who have sex with men and transgender women who have sex with men showed that Descovy (emtricitabine–tenofovir alafenamide), a drug with a more favorable safety profile, was noninferior to emtricitabine–tenofovir disoproxil fumarate.9 To this day, daily oral PrEP with emtricitabine–tenofovir alafenamide is not recommended for cisgender women because no trial data are available. The dapivirine vaginal ring is effective and acceptable for HIV PrEP,10 but it is not available in the United States. This situation has informed a perception that studies of STI and HIV interventions in women too often follow a "one size should fit all" approach, yet the biology of transmission and infection have some important differences.
8+ Year Pooled Analysis: Adherence and HIV Incidence in >6000 Women on F/TDF for PrEP - adherence matters
No new HIV infections were observed in women with high, steady adherence


"we have to consider how to use this medication judiciously especially considering antimicrobial resistance"
"there are ways to do DoxyPEP so we can be more judicious about its use"
"I find it difficult to believe we cannot come to recommend DoxyPEP given we have such robust evidence especially in the MSM & transgender women population" - "its about equity in these groups". "There is no way we cannot recommend it in these groups.
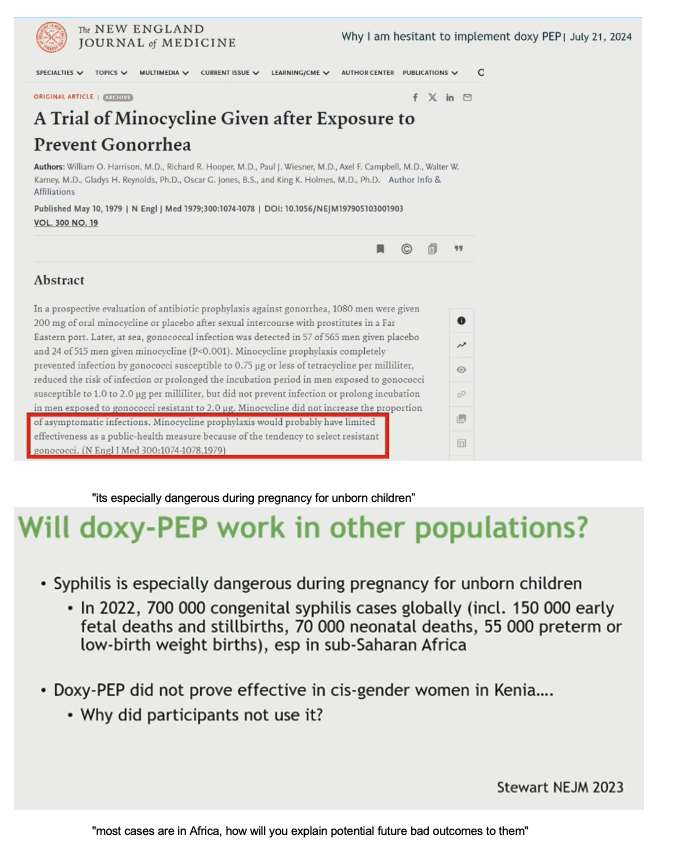
"We expect it will wrk in cisgender women, it builds up in the cervix. I believe it failed in Kenya due to lack of adherence".
"CDC Guidelines discussed using it in MSM & transgender women with a history of bacterial STIs. We need a little more data in cisgender women, in my practice I use it on a case by case basis in cisgender heterosexual women, in my opinion we should all be doing that". If you have a cisgender heterosexual women in your practice, potentially a sex worker, transactional sex, someone at high risk for bacterial STIs, it makes sense currently I think to prescribe it to that group. In terms of public health it makes sense to use it especially for MSM & transgender women".

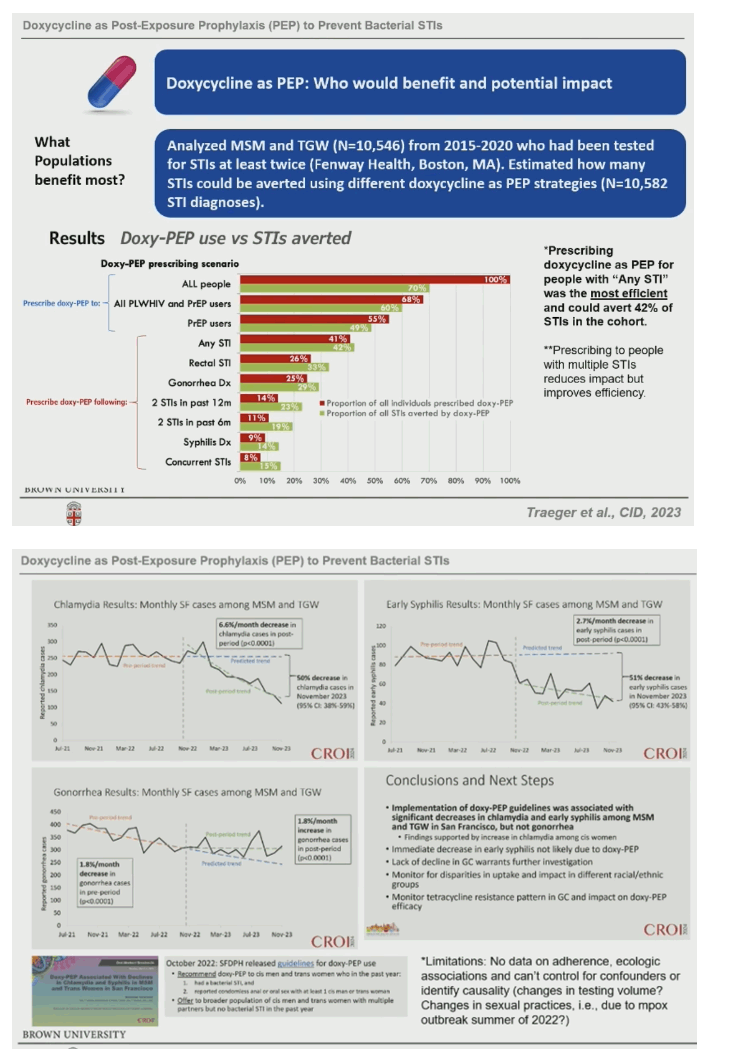
Chan: "there are bridging populations (bisexual men) between MSM/transgender women & cisgender women, so even if DoxyPEP reduced incidence of bacterial STIs only in MSM I would expect there would be a reduction of bacterial STIs in heterosexual cisgender women. Regarding microbiome we should stop eating at McDonald's if you are concerned about harming the microbiome, its difficult NOT to remmed DoxyPEP to my patients based on a theoretical risk of the microbiome, so many other medications we take on a regular basis affect the microbiome, I dont think its fair withhold DoxyPEP from MSM or transgender women based on the theoretical risk of harm to the microbiome. I DO believe we need to be judicious in its use, it not black & white, yes or no, about how we use it, it need a framework around how to use it, I like what CDC Guidelines said to consider it in MSM & transgender women wo have had a bacterial infection in the past 12 months, focus on populations at high risk for bacterial STIs. Regarding resistance, there is very little data, resistance is rare to non existent in syphillis & chlamydia, we are certainly concerned about it in genorrhea, micro plasma genitelium, in community acquired MRSA (Doxy is one of my go to drugs for that). Its something to watch & observe, as a physician & public health person I dont feel comfortable not recommending when its efficacious especially within historically marginalized groups, we need to recommend & put a framework & guidelines around it but I dont feel comfortable not recommending it in those groups".
Henry De Vries:
"It works for syphilis". This Australian recommendation is a good group to recommend it you. I highly doubt it will affect other groups like Can suggests for cisgender heterosexual women, bridging populations".




|
| |
|
 |
 |
|
|