| |
Thiazolidinedione Treatment Decreases Bone Mineral Density in
Type 2 Diabetic Men
|
| |
| |
From Jules Levin: as far as I can find rosiglitazone has been shown to result in bone loss in men & women. In the pioglitazone study postmenopausal women had bone loss & fractures on piolglitazone but men did not. This was a large pioglitazone I think phase III or 4 study.
Diabetes Care 30:1574-1576, 2007
DOI: 10.2337/dc06-2606
Subhashini Yaturu, MD1,2, Barbara Bryant, BS1 and Sushil K. Jain, PHD3
1 Department of Endocrinology, Overton Brooks VA Medical Center, Shreveport, Louisiana
2 Department of Endocrinology, Louisiana State University Health Sciences Center, Shreveport, Louisiana
3 Department of Pediatrics, Louisiana State University Health Sciences Center, Shreveport, Louisiana
INTRODUCTION
Case-control studies of patients with fractures have found that subjects with diabetes have at least a twofold higher risk of fracture than subjects without diabetes (1), with an increased risk of hip, humerus, and foot fractures in elderly diabetic subjects (2-4). Risk factors that contribute to increased fracture in diabetic subjects include number of falls (5,6), insulin use (7,8), functional disability (9-11), diabetes duration (7,12), and poor vision (7). Recent studies report that older women and African Americans have higher incidence of osteoporotic fractures (13). In addition, lower bone strength (bone mineral density [BMD]) might be expected to increase risk for the development of osteoporosis and fracture.
Type 2 diabetic patients are widely prescribed drugs called thiazolidinediones, which increase insulin sensitivity via activation of peroxisome proliferator-activated receptor (PPAR)- receptors. However, it is not known whether thiazolidinedione use has any effect on bone mass and thereby increases risk of fracture in type 2 diabetic patients. In animal studies, thiazolidinedione treatment was associated with bone loss in a mouse model (14,15), which was explained by possible imbalance in bone from increased apoptotic death of osteogenic cells and diminished bone formation (15), and also in ovariectomized rats (16). Other investigators did not find bone loss in troglitazone-treated mice (17). There is limited information on the effect of thiazolidinediones on BMD in humans. A recent study has suggested that thiazolidinedione use may cause bone loss in older women with type 2 diabetes (18). In the present study on 160 men with type 2 diabetes, we examined BMD in patients on rosiglitazone treatment compared with matched men with type 2 diabetes not on rosiglitazone treatment and found that rosiglitazone treatment increases bone loss in men with type 2 diabetes.
RESEARCH DESIGN AND METHODS-
After institutional review board approval, we retrieved the clinical data for the previous 4 years of 354 subjects with diabetes and compared with BMD data of age- and BMI-matched nondiabetic subjects. Among 354 subjects with diabetes, we had follow-up BMD data for 32 men with type 2 diabetes receiving rosiglitazone (considered the study group) and 128 men with type 2 diabetes not receiving any thiazolidinedione (considered the control group). Clinical data included are history of smoking, alcohol consumption, hypertension, coronary artery disease, stroke, current medications, and duration of diabetes and family history of diabetes and osteoporosis. Other details collected include height, weight, BMI, abdominal girth, hip-to-waist measurements and ratio, and blood pressure. The dose of rosiglitazone was 4 mg twice a day. We excluded the data of the subjects with chronic renal insufficiency, hyperparathyroidism, and Cushing's syndrome and of those who were on glucocorticoids and gonadotropin-releasing hormone analogs.
BMD measurements were performed using dual-energy X-ray absorptiometry (DEXA) (Lunar Prodigy) at the lumbar spine: anteroposterior (AP) and lateral (L1-4) and the proximal femur (total hip, femoral neck, and trochanter) and 33% radius. The precision of DEXA scans is 1-1.8%. Annualized absolute change in BMD was calculated as the difference between the results of the baseline and follow-up hip scans divided by the time between scans in years. Annualized percentage of change in BMD was calculated by dividing the annualized absolute change by the baseline BMD. We compared the BMD data of subjects with diabetes and on rosiglitazone with those of subjects with diabetes and not on rosiglitazone using unpaired t test. A P value <0.05 on two-tailed testing was considered significant.
RESULTS AND DISCUSSION-
The BMD data at both AP spine and hip in subjects with diabetes were similar to those of subjects with no diabetes when matched for age and BMI (data not given here). Among men with type 2 diabetes, 160 subjects had a follow-up DEXA with a mean interval of 16 months, including 128 subjects with diabetes and not receiving rosiglitazone (control group) and 32 subjects with diabetes and receiving rosiglitazone (study group). The BMD and clinical characteristics of subjects with type 2 diabetes are shown in Table 1. There was no significant difference in the baseline BMD data at both AP spine and hip in subjects with type 2 diabetes with or without rosiglitazone. Since all of the follow-up DEXA data were not collected at exact similar intervals, annualized percentage changes were calculated. Annualized absolute percentage changes in both groups were compared, and the results are shown in Table 1.
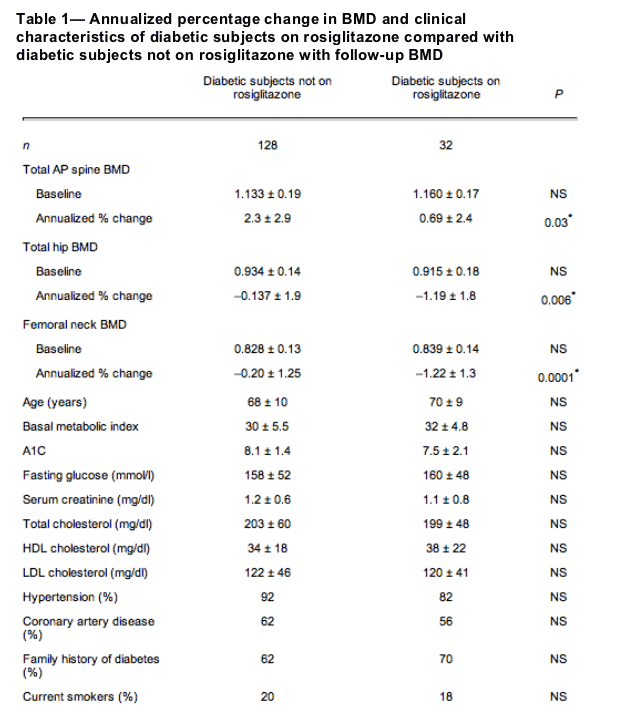
BMD in subjects with type 2 diabetes was similar to that in nondiabetic subjects. This finding is consistent with that of a previous study (19). A novel finding in our report is the adverse effect of rosiglitazone, a thiazolidinedione, on skeletal health in humans. There was a significant difference in the BMD, with loss of bone at both the spine and hip in type 2 diabetic subjects on rosiglitazone (Table 1). This study confirms the results of previous animal studies and shows the possible adverse effect of rosiglitazone on skeletal health in men.
Osteoblasts and adipocytes are derived from a common multipotential mesenchymal stem cell progenitor (18-20). PPAR- is essential for normal adipocyte differentiation and proliferation as well as fatty acid uptake and storage. The thiazolidinediones rosiglitazone and pioglitazone are commonly used to increase insulin sensitivity for the treatment of type 2 diabetes (21). It has been shown that activation of PPAR-2 with rosiglitazone stimulates adipogenesis and inhibits osteoblastogenesis (22). This may be a potential mechanism for the loss of BMD observed in hips of rosiglitazone-treated diabetic men.
This study is retrospective. Limitations include that there were no bone turnover markers for comparison and that the data groups are not large enough to perform regression analyses for confounding factors such as age, duration of diabetes, use of insulin, association of hypertension, and other factors such as smoking and alcoholism. Nevertheless, this study shows a significant increase in bone loss both at total hip and femoral neck areas in type 2 diabetic men on rosiglitazone treatment. This suggests that thiazolidinedione treatment is a risk factor and can contribute to excess incidence of fractures in diabetes.
Journal of Clinical Endocrinology & Metabolism, doi:10.1210/jc.2006-2646
The Peroxisome Proliferator-Activated Receptor- Agonist Rosiglitazone Decreases Bone Formation and Bone Mineral Density in Healthy Postmenopausal Women: A Randomized, Controlled Trial
Andrew Grey, Mark Bolland, Greg Gamble, Diana Wattie, Anne Horne, James Davidson and Ian R. Reid
Department of Medicine (A.G., M.B., G.G., D.W., A.H., I.R.R.), University of Auckland, and LabPlus (J.D.), Auckland City Hospital, 1020 Auckland, New Zealand
Address all correspondence and requests for reprints to: Dr. Andrew Grey, Department of Medicine, University of Auckland, Private Bag 92019, 1020 Auckland, New Zealand. E-mail: a.grey@auckland.ac.nz.
Context: Thiazolidinediones, which are peroxisome proliferator-activated receptor- agonists, are widely prescribed to patients with disorders characterized by insulin resistance. Preclinical studies suggest that peroxisome proliferator-activated receptor- signaling negatively regulates bone formation and bone density. Human data on the skeletal effects of thiazolidinediones are currently available only from observational studies.
Objective: The objective of the study was to determine whether rosiglitazone, a thiazolidinedione, inhibits bone formation.
Design: The study was a 14-wk randomized, double-blind, placebo-controlled trial.
Setting: The study was conducted in the general community.
Patients: Fifty healthy, postmenopausal women participated in the study.
Intervention: Intervention was rosiglitazone 8 mg/d.
Main Outcome Measures: The primary end point was biochemical markers of bone formation, and secondary end points were a bone resorption marker and bone mineral density.
Results: The osteoblast markers procollagen type I N-terminal propeptide and osteocalcin declined by 13% (P < 0.005 vs. placebo) and 10% (P = 0.04 vs. placebo), respectively, in the rosiglitazone group. These changes were evident by 4 wk and persisted for the duration of the study. There was no change in the serum ß-C-terminal telopeptide of type I collagen, a marker of bone resorption (P = 0.9 vs. placebo). Total hip bone density fell in the rosiglitazone group (mean change from baseline rosiglitazone -1.9%, placebo -0.2%; between-group difference 1.7%, 95% confidence interval 0.6-2.7, P < 0.01); lumbar spine bone density fell significantly from baseline values in the rosiglitazone group (P = 0.02 vs. baseline) but was not significantly different between groups (mean change from baseline rosiglitazone -1.2%, placebo -0.2%; between-group difference 1.0%, 95% confidence interval -0.2-2.3, P = 0.13).
Conclusions: Short-term therapy with rosiglitazone exerts detrimental skeletal effects by inhibiting bone formation. Skeletal end points should be included in future long-term studies of thiazolidinedione use.
Introduction
THIAZOLIDINEDIONES ARE insulin-sensitizing agents that are widely prescribed in the management of a variety of clinical conditions characterized by insulin resistance (1, 2). They are agonists of the peroxisome proliferator-activated receptor (PPAR) family of nuclear transcription factors, in particular the PPAR- isoform (1). In patients with type 2 diabetes mellitus, their use is associated with significant improvements in glycemic control and serum lipoprotein profile, although their ability to reduce the incidence of vascular events is uncertain (3). At present, thiazolidinediones account for 21% of oral antihyperglycemic drugs used in the United States and 5% in Europe (4). It is estimated that 2 million Americans were prescribed rosiglitazone last year (personal communication, Westun, C., GlaxoSmithKline, New Zealand).
PPAR- is expressed in a number of tissues (1), raising the possibility that drugs that interact with it may induce clinical effects other than insulin sensitization. Prominent among the tissues in which PPAR- is expressed is bone. In skeletal tissue, PPAR- acts as a molecular switch that regulates the fate of pluripotent mesenchymal stem cells, which have the ability to differentiate into adipocytes or osteoblasts. In vitro, PPAR- agonists promote adipocyte differentiation in preference to osteoblast differentiation (5, 6, 7, 8). There are conflicting reports of the effects of PPAR- activation on osteoclastogenesis (9, 10). Haploinsufficiency of the PPAR- gene in mice induces a high bone density phenotype characterized by increased rates of osteoblastic bone formation (11, 12), whereas treatment of rodents with PPAR- agonists induces bone loss characterized by deficient osteoblast function (12, 13, 14). Data from human studies of the skeletal actions of thiazolidinediones are currently available only from an observational study, which reported that female, but not male, diabetic thiazolidinedione users experience accelerated bone loss, compared with nonthiazolidinedione users (15).
Patients with type 2 diabetes may be at increased risk of fragility fractures (16, 17, 18, 19, 20, 21, 22). Because PPAR- agonists are increasingly frequently used to treat this disease, it is important to determine whether these drugs have adverse effects on the human skeleton. We undertook a randomized, placebo-controlled trial to test the hypothesis that treatment with rosiglitazone would cause adverse skeletal effects in healthy postmenopausal women. The primary objective was to determine the effect of rosiglitazone 8 mg daily on biochemical markers of bone formation over a 14-wk period. Secondary end points were change in markers of bone resorption and bone mineral density.
Discussion
This study demonstrates that short-term therapy with rosiglitazone, a commonly prescribed PPAR- agonist, inhibits bone formation and accelerates bone loss in healthy postmenopausal women. These data are consistent with those from in vitro and animal studies demonstrating that PPAR- signaling negatively regulates osteoblast function (bone formation) and bone mass (7, 8, 11, 13, 14). The pattern of alteration of bone remodeling that we observed in response to rosiglitazone is similar to that seen after the initiation of glucocorticoid therapy (26). The uncoupling of bone formation from resorption by glucocorticoids is accompanied by early and rapid bone loss and an increased risk of fragility fractures (27). Our data suggest that rosiglitazone may also promote rapid bone loss; longer-term studies are needed to determine whether the rate of loss we observed is sustained. Because patients with type 2 diabetes may have an increased risk of fragility fractures (16, 17, 18, 19, 20), the possibility that one of the therapies commonly used to treat the disease may be increasing that risk is a cause for concern. The increasing use of thiazolidinediones in other clinical conditions characterized by insulin resistance (28, 29), including impaired glucose tolerance (30), is a further reason to fully characterize their long-term skeletal effects. We therefore suggest that skeletal safety end points should be added to existing and planned randomized trials of PPAR- agonists so that the skeletal effects of thiazolidinediones can be studied over a longer period.
Although preclinical studies have consistently reported that rosiglitazone impairs osteoblast function (13, 14, 31, 32), conflicting in vitro data exist as to whether PPAR- signaling affects osteoclastogenesis (7, 9, 10). Our data suggest that PPAR- agonists do not influence bone resorption in vivo, a finding consistent with those of in vivo studies in rodents (11, 13, 14). The limited preclinical data that are available on the skeletal effects of pioglitazone, the other commonly prescribed thiazolidinedione, suggest that it has comparable actions with those of rosiglitazone (33, 34). Whether there is a class effect of thiazolidinediones on skeletal homeostasis is uncertain, with recent preclinical studies of new compounds reporting both adverse (35) and neutral (36) effects in rodent models.
Currently there are few data available on the skeletal actions of thiazolidinediones in humans. Uncontrolled studies of Japanese subjects with type 2 diabetes treated with troglitazone, a PPAR- agonist no longer in clinical use, reported significant reductions in markers of both bone formation and resorption after 1 month, but values returned to baseline by 1 yr (37, 38). More recently an analysis of the small number (n = 69) of diabetic subjects taking thiazolidinediones (pioglitazone, troglitazone, and rosiglitazone) in the Health, Aging, and Body Composition observational study reported accelerated bone loss in over 4 yr in women but not men (15). After our manuscript was submitted, Kahn et al. (39) reported a higher incidence of fractures, detected as adverse events, in female diabetic subjects randomized to receive rosiglitazone, compared with those randomized to receive either metformin or glyburide, during a 4 yr study of glycemic durability of oral monotherapies. Our findings provide rigorous evidence for a detrimental effect of PPAR- agonists on the postmenopausal female skeleton. Whether there is a gender difference in the skeletal response to thiazolidinediones can be determined only by a randomized, controlled trial in men.
The mechanism(s) by which rosiglitazone alters bone remodeling likely involves direct effects on osteoblast development and function, but the possibility of indirect skeletal actions also exists. Adipose tissue is a target for PPAR- agonists, and some adipokines influence bone cell function. Thiazolidinediones may decrease circulating levels of leptin (38), the peripheral actions of which include osteoblast anabolism (40). The insulin-sensitizing actions of PPAR- agonists lower circulating levels of insulin (1) and therefore are likely to reduce levels of the cosecreted pancreatic ß-cell peptide amylin, each of which is anabolic to osteoblasts (41, 42).
A theoretical limitation of our study is the possibility that dual-energy x-ray absorptiometry may underestimate bone density in rosiglitazone-treated subjects because of changes in bone marrow adiposity. We think this unlikely because, although it is not known whether rosiglitazone increases marrow fat in humans, 30-fold higher doses (by body weight) of drug than those used in our study do not increase marrow fat in rodents (13), and dual-energy x-ray absorptiometry accurately measures bone density in vivo, as corroborated by histomorphometry and/or microcomputed tomography, when marrow adiposity is known to be either increased (31, 36) or absent (43).
Limitations of the present study are its short duration and the healthy volunteer study population. An advantage of studying healthy subjects is that it allows an assessment of the effects of rosiglitazone on bone metabolism, independent of any confounding introduced by improvements in the metabolic control of diabetes mellitus (44). Furthermore, because the indications for use of thiazolidinediones are expanding to include individuals with insulin resistance (30), assessment of their bone effects outside the context of a specific pathological state is appropriate. The short duration of the study was necessary because the hypothesis was one of harm, and the study participants were normal volunteers who would not be expected to benefit from the intervention. We believe that the 14-wk study duration was adequate to provide proof of principle that thiazolidinediones impact adversely on the human skeleton, without exposing the subjects to a significant risk of long-term skeletal harm. Longer-term studies are clearly necessary, but these would best be undertaken in study populations that might reasonably be expected to derive some benefit from thiazolidinedione use, such as people with type 2 diabetes and/or impaired glucose tolerance.
Journal of Clinical Endocrinology & Metabolism, doi:10.1210/jc.2005-2226
Thiazolidinedione Use and Bone Loss in Older Diabetic Adults
Ann V. Schwartz, Deborah E. Sellmeyer, Eric Vittinghoff, Lisa Palermo, Beata Lecka-Czernik, Kenneth R. Feingold, Elsa S. Strotmeyer, Helaine E. Resnick, Laura Carbone, Brock A. Beamer, Seok Won Park, Nancy E. Lane, Tamara B. Harris, Steven R. Cummings for the Health, Aging and Body Composition (Health ABC) Study
Department of Epidemiology and Biostatistics (A.V.S., E.V., L.P.) and Division of Endocrinology (D.E.S., K.R.F.), Department of Medicine, University of California, San Francisco, San Francisco, California 94107-1762; Department of Geriatrics (B.L.-C.), Reynolds Institute on Aging, University of Arkansas for Medical Sciences, Little Rock, Arkansas 72202; Department of Epidemiology (E.S.S., S.W.P.), Graduate School of Public Health, University of Pittsburgh, Pittsburgh, Pennsylvania 15260; Department of Epidemiology (H.E.R.), MedStar Research Institute, Hyattsville, Maryland 20785; Division of Rheumatology (L.C.), Department of Medicine, University of Tennessee, Memphis, Tennessee 38163; Department of Medicine (B.A.B.), Johns Hopkins University, Baltimore, Maryland 21205; Department of Medicine (S.W.P.), Pochon CHA University, Seoul 135-081 Korea; Department of Medicine (N.E.L.), University of California, Davis, Sacramento, California 95616; Laboratory of Epidemiology, Demography, and Biometry (T.B.H.), National Institute on Aging, Bethesda, Maryland 20892; and Research Institute (S.R.C.), California Pacific Medical Center, San Francisco, California 94120
Context: Activation of peroxisome proliferator-activated receptor- by thiazolidinediones (TZDs) results in lower bone mass in mice.
Objective: The objective of the study was to determine whether TZD use is associated with changes in bone mineral density (BMD) in older adults with type 2 diabetes.
Design: We analyzed 4-yr follow-up data from the Health, Aging, and Body Composition observational study.
Setting: The study was conducted in a general community.
Patients: White and black, physically able men and women, aged 70-79 yr at baseline with diabetes defined by self-report, use of hypoglycemic medication, elevated fasting glucose (126 mg/dl), or elevated 2-h glucose tolerance test (200 mg/dl) participated in the study.
Main Outcome Measures: Whole-body, lumbar spine (derived from whole body), and hip BMD were measured by dual-energy x-ray absorptiometry at 2-yr intervals.
Results: Of 666 diabetic participants, 69 reported TZD use at an annual visit, including troglitazone (n = 22), pioglitazone (n = 30), and/or rosiglitazone (n = 31). Those with TZD use had higher baseline hemoglobin A1c and less weight loss over 4 yr but similar baseline BMD and weight than others with diabetes. In repeated-measures models adjusted for potential confounders associated with TZD use and BMD, each year of TZD use was associated with greater bone loss at the whole body [additional loss of -0.61% per year; 95% confidence interval (CI) -1.02, -0.21% per year], lumbar spine (-1.23% per year; 95% CI -2.06, -0.40% per year), and trochanter (-0.65% per year; 95% CI -1.18, -0.12% per year) in women, but not men, with diabetes.
Conclusion: These observational results suggest that TZDs may cause bone loss in older women. These results need to be tested in a randomized trial.
|
|
| |
| |
|
|
|