| |
Antiviral Activity of Single-Dose PRO 140, a CCR5 Monoclonal Antibody, in HIV-Infected Adults
|
| |
| |
"subjects were randomized 10:3 to receive a single iv infusion of PRO 140 or placebo in 1 of 3 dose cohorts and then followed up for 58 days. PRO 140 is a humanized IgG4κ form of the CCR5 MAb PA14.....Dose-dependent and highly significant reductions in HIV-1 RNA level were observed. Mean ± SD nadir HIV-1 RNA reductions of 0.39 +/- 0.20, 0.58 +/- 0.30 (p=.0002), and 1.83 +/- 0.41 (p<.0001) ) log10 copies/mL were observed for the placebo, 0.5-mg/kg, 2-mg/kg, and 5-mg/kg groups, respectively...individual nadir reductions were as great as 2.5 log10 copies/mL (99.7%) in both the 2-mg/kg and 5-mg/kg groups. In the 5-mg/kg group, individual HIV-1 RNA nadirs were observed on or between days 8 and 15.....PRO 140 was generally well tolerated. With the exception of 1 individual in the 0.5-mg/kg group....The present study provides proof of concept for PRO 140 as a potent antiretroviral agent with prolonged single-dose activity. The viral load reductions observed after treatment with single-dose PRO 140 are similar in magnitude to those reported during 10-14 days of treatment with small-molecule CCR5 antagonists"
The Journal of Infectious Diseases
Received 15 February 2008; accepted 5 May 2008; electronically published 4 September 2008.
Jeffrey M. Jacobson,1 Michael S. Saag,2 Melanie A. Thompson,3 Margaret A. Fischl,4 Ralph Liporace,5 Richard C. Reichman,6 Robert R. Redfield,10 Carl J. Fichtenbaum,11 Barry S. Zingman,7 Mahesh C. Patel,8 Jose D. Murga,9 Suzanne M. Pemrick,9 Paul D'Ambrosio,9 Marti Michael,9 Hans Kroger,9 Hieu Ly,9 Yakov Rotshteyn,9 Robert Buice,9 Stephen A. Morris,9 Joseph J. Stavola,9 Paul J. Maddon,9 Alton B. Kremer,9 and William C. Olson9
1Drexel University College of Medicine, Philadelphia, Pennsylvania; 2University of Alabama, Birmingham; 3AIDS Research Consortium of Atlanta, Atlanta, Georgia; 4University of Miami, Miami, Florida; 5Albany Medical Center, Albany, 6University of Rochester, Rochester, 7Montefiore Medical Center and the Einstein/Montefiore Center for AIDS Research and 8Jacobi Medical Center, Bronx, and 9Progenics Pharmaceuticals, Tarrytown, New York; 10University of Maryland, Baltimore, Maryland; 11University of Cincinnati, Cincinnati, Ohio
ABSTRACT
Background. The current goal of human immunodeficiency virus type 1 (HIV-1) therapy is to maximally suppress viral replication. Securing this goal requires new drugs and treatment classes. The chemokine receptor CCR5 provides an entry portal for HIV-1, and PRO 140 is a humanized monoclonal antibody that binds to CCR5 and potently inhibits CCR5-tropic (R5) HIV-1 in vitro.
Methods. A randomized, double-blind, placebo-controlled, dose-escalating study was conducted in 39 individuals with HIV-1 RNA levels >5000 copies/mL, CD4+ cell counts >250 cells/μL, no antiretroviral therapy for 3 months, and only R5 HIV-1 detectable. Cohorts were randomized 3:10 to receive placebo or doses of PRO 140 of 0.5, 2, or 5 mg/kg. Subjects were monitored for 58 days for safety, antiviral effects, and serum concentrations of PRO 140.
Results. PRO 140 was generally well tolerated and demonstrated potent, rapid, prolonged, and dose-dependent antiviral activity. Mean reductions in HIV-1 RNA level of 0.58 log10, 1.20 log10 (p=.0002) and 1.83 log10 (p<.0001) were observed for the 0.5-, 2-, and 5-mg/kg dose groups, respectively. Reductions in mean viral load of 10-fold were observed within 4 days and persisted for 2-3 weeks after treatment.
Conclusions. This trial established clear proof of concept for PRO 140 as a potent antiretroviral agent with extended activity after a single dose.
Highly active antiretroviral therapy for HIV-1 infection has significantly improved the survival of infected individuals [1, 2]. However, such therapy is noncurative. Its primary goal, to maximally suppress viral replication, currently is unmet in many individuals because of the development of multidrug-resistant virus, the requirement of multiple active drugs, treatment toxicities, drug-drug interactions, food and fasting requirements, and suboptimal adherence. New treatment strategies are required to improve the life-long care of HIV-infected individuals.
The HIV-1 replicative cycle is initiated when the virus enters cells that express CD4 [3] and a coreceptor molecule, typically the chemokine receptor CCR5 [4-6] or CXCR4 [7]. CCR5 is the predominant coreceptor used for transmission and throughout the early to middle stages of disease. The percentage of subjects with viruses that can use CXCR4 increases with disease progression. An inactivating genetic deletion in CCR5 (_32) slows HIV-1 disease progression or protects against initial infection when present in 1 or 2 copies, respectively [8]. The _32 deletion has no obvious effect on health; however, homozygosity has been associated with increased risk for symptomatic disease after infection with West Nile virus and tickborne encephalitis virus [9, 10].
PRO 140 is a humanized CCR5 monoclonal antibody (MAb) that broadly and potently inhibits R5 HIV-1 at concentrations that do not antagonize the natural activity of CCR5 in vitro [11-13]. PRO 140 does not inhibit CXCR4-using viruses. Here, we report findings of the first study of PRO 140 in HIV-infected individuals. Subjects with early-stage HIV-1 infection were treated in dose-ascending cohorts with single intravenous (iv) infusions of PRO 140 or placebo and then followed up for antiviral effects, safety, and pharmacology. Potent, prolonged, dose-dependent, and highly significant antiviral activity was observed, with minimal toxicity.
Methods
Study design. A randomized, double-blind, placebo-controlled, dose-ascending phase 1b study was conducted to evaluate the tolerability, antiviral activity, and pharmacokinetics (PK) of single iv doses of PRO 140 in HIV-infected adults. Informed consent was obtained from the subjects, and the human experimentation guidelines of the US Department of Health and Human Services were followed. Subjects were >18 years of age with HIV-1 RNA levels 5000 copies/mL, only CCR5-tropic (R5) HIV-1 detectable, CD4+ cell counts >250 cells/μL with no documented nadir <200/μL, and no antiretroviral therapy for >3 months. Exclusion criteria included pregnancy, history of AIDS-defining illness, and acute or symptomatic viral hepatitis within 6 months. Eligible subjects were randomized 10:3 to receive a single iv infusion of PRO 140 or placebo in 1 of 3 dose cohorts and then followed up for 58 days. PRO 140 is a humanized IgG4κ form of the CCR5 MAb PA14 [14]. PRO 140 (10 mg/mL nominal concentration) and matched placebo were provided as sterile PBS (pH 7.2) and were administered within 30 min by means of an infusion pump. PRO 140 concentrations and dose levels are nominal values based on a theoretical ultraviolet spectroscopy extinction coefficient. Based on the experimentally determined extinction coefficient, the actual concentrations and doses of PRO 140 are 10% lower than those reported here.
Virological analyses. Plasma HIV-1 RNA levels were determined with the Cobas Amplicor HIV-1 Monitor Test (version 1.5; Roche Diagnostics). Samples with levels <400 copies/mL were reanalyzed with the ultrasensitive method. Coreceptor tropism was determined at screening and on days 1, 8, 29, and 59 (Trofile Assay; Monogram Biosciences) [15]. Viral susceptibility was determined on days 1 and 59 with the PhenoSense HIV Entry Assay. Results obtained with CCR5+ target cells were reported as fold change or relative EC50 values, defined as (EC50 of the test isolate)/(EC50 of the reference virus). EC50 is the concentration required for 50% inhibition and was determined by logistical fit of the data. Maximum percentages of inhibition were determined from the plateaus of the inhibition curves. The reference virus was HIV-192HT594. The HIV-1 subtype and genotypic susceptibility to protease and reverse-transcriptase inhibitors were assessed using GeneSeq (Monogram Biosciences).
Safety evaluations. Evaluations included physical examinations, 12-lead electrocardiograms, determination of vital signs, and reporting of concomitant medications and adverse events. Clinical laboratory tests included serum chemistry, hematology, and urinalysis.
Bioanalytical and PK methods. Serum concentrations of PRO 140 and of antibodies to PRO 140 were measured using validated ELISAs. The assay for PRO 140 had a lower limit of quantification (80 ng/mL) and used a mouse anti-idiotype MAb (Progenics Pharmaceuticals) for capture and a mouse anti-IgG4 Fc antibody linked to horseradish peroxidase (The Binding Site) for detection. PK metrics were estimated after noncompartmental analysis using WinNonlin (version 4.1; Pharsight). Anti-PRO 140 antibodies were detected by capture onto PRO 140-coated microtiter plates and then by sequential addition of biotinylated PRO 140, streptavidin-conjugated horseradish peroxidase, and substrate. Serum samples were tested at 1:10 dilutions initially. Positive samples were serially diluted for titer analysis.
Lymphocyte and RANTES analyses. CCR5+ and CD4+ lymphocytes were measured by flow cytometry. CCR5+ lymphocytes were analyzed using phycoerythrin (PE)-labeled PRO 140, CTC5 (R&D Systems), and isotype-control antibodies. PRO 140 blocks the binding of PRO 140-PE but not CTC5-PE. PRO 140-PE and CTC5-PE assess the level of coating and depletion of CCR5+ lymphocytes, respectively. Pretreatment CCR5+ lymphocyte counts were based on CTC5-PE data. Because PRO 140 potentiates the binding of CTC5-PE to CCR5 in vitro, CTC5-PE data were obtained only to assess the potential depletion of CCR5+ cells after treatment. RANTES was measured in platelet-depleted plasma using a validated ELISA.
Statistical and pharmacodynamic analyses. Statistical analyses were performed on log10-transformed HIV-1 RNA data. Changes were calculated relative to baseline (predose day 1). Treatment and placebo groups were compared using an analysis of variance model. If the overall F test was found to have statistically significant results, each treatment group was compared with the placebo group, using pairwise 2-sided t tests. WinNonlin (version 4.1) was used to fit antiviral and PK data to the following Emax model:
where E is the change in HIV-1 RNA level, Emax is the maximum predicted change in HIV-1 RNA level, AUC is the area under the concentration-time curve, and AUC50 is the AUC required to achieve 50% of Emax. Goodness of fit was assessed from the correlation, residuals, and SEs. AUC from time 0 to infinity (AUC∞) values for individual subjects were used in the model.
Results
Subject characteristics and disposition. The study was conducted at 10 centers in the United States. Baseline characteristics for the 39 treated subjects are summarized in table 1 and were similar for the different treatment groups. The study included 31 male and 8 female subjects, and their median age, CD4+ cell count, and plasma HIV-1 RNA level at baseline were 40.3 years, 484 cells/μL, and 26,900 copies/mL, respectively. Fifteen subjects reported prior use of antiretroviral therapy, and baseline genotypic resistance to existing drugs was limited to single-class resistance in 1-2 subjects per group. All HIV-1 isolates were subtype B. Seven individuals were coinfected with hepatitis C virus. The mean infusion time was 36 min, and all subjects completed the 58-day follow-up period.
Antiviral effects. Dose-dependent and highly significant reductions in HIV-1 RNA level were observed. Mean ± SD nadir HIV-1 RNA reductions of 0.39 +/- 0.20, 0.58 +/- 0.30 (p=.0002), and 1.83 +/- 0.41 (p<.0001) ) log10 copies/mL were observed for the placebo, 0.5-mg/kg, 2-mg/kg, and 5-mg/kg groups, respectively (table 2). Individual nadir reductions were as great as 2.5 log10 copies/mL (99.7%) in both the 2-mg/kg and 5-mg/kg groups. In the 5-mg/kg group, individual HIV-1 RNA nadirs were observed on or between days 8 and 15. In univariate linear regression analyses with the Bonferroni correction for multiple comparisons, no significant correlation was observed between the maximum viral load reduction and baseline HIV-1 RNA level, CD4+ cell count, or CCR5+ cell count.
Antiviral effects exhibited a rapid onset and extended duration (figure 1). At 5 mg/kg, mean viral load reductions of 1 log10 copies/mL were first observed on day 5 and persisted until 2-3 weeks after treatment. Viral load reductions were statistically significant at p<.0001 from day 5 to 15 for the 5-mg/kg group and at p<.01 from day 8 to 15 for the 2-mg/kg group. On day 10, the mean ± SD log10 declines in viral load were .13 +/- 0.24, 0.37 +/- 0.54, 1.04 +/- 0.45 and 1.70 +/- 0.49 (p=.0001) copies/mL for the placebo and ascending PRO 140 dose groups, respectively. For the placebo group, there was no significant change from baseline in HIV-1 RNA level at any time point (figure 1). Viral loads returned to baseline by day 29 in all groups.
Antiviral effects exhibited a rapid onset and extended duration (figure 1). At 5 mg/kg, mean viral load reductions of 1 log10 copies/mL were first observed on day 5 and persisted until 2-3 weeks after treatment. Viral load reductions were statistically significant at p<.0001 from day 5 to 15 for the 5-mg/kg group and at p<.01 from day 8 to 15 for the 2-mg/kg group. On day 10, the mean ± SD log10 declines in viral load were .13 +/- 0.24, 0.37 +/- 0.54, 1.04 +/- 0.45 and 1.70 +/- 0.49 (p=.0001) copies/mL for the placebo and ascending PRO 140 dose groups, respectively. For the placebo group, there was no significant change from baseline in HIV-1 RNA level at any time point (figure 1). Viral loads returned to baseline by day 29 in all groups.
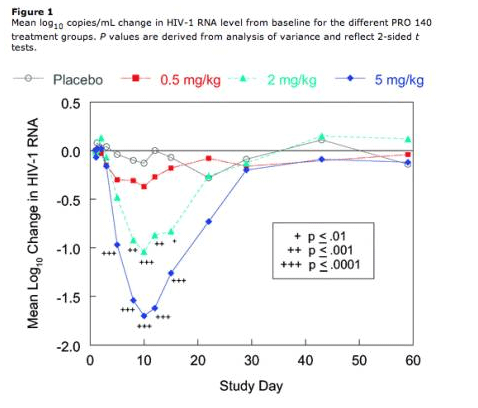
Antiviral response was defined as a >1.0 log10 copies/mL reduction in HIV-1 RNA level at any time point after treatment. The antiviral response rate was 100% at 5 mg/kg (p<.0001), 60% at 2 mg/kg (p=.011), and 10% at 0.5 mg/kg. No subject receiving placebo experienced a >1.0 log10 copies/mL reduction in HIV-1 RNA level. The duration of response also increased with dose (p=.0059). At 5 mg/kg, individual durations of response varied 2-fold, as viral load reductions of >1.0 log10 copies/mL were observed through day 10 at minimum (1 subject) and day 22 at maximum (4 subjects). Viral load was reduced to <400 copies/mL in 4 subjects receiving 5 mg/kg and in 1 subject receiving 2 mg/kg. The lowest documented HIV-1 RNA level in the present study was 61 copies/mL.
Safety. PRO 140 was generally well tolerated. With the exception of 1 individual in the 0.5-mg/kg group, all subjects receiving PRO 140 or placebo reported at least 1 adverse event. The most frequently reported adverse events were headache, in 12 subjects (31%); lymphadenopathy, in 11 subjects (28%); and diarrhea and fatigue, in 8 subjects (21%) each. No dose-proportional trend in the frequency of adverse events was observed. The majority of events were judged by the investigator to be either not related or unlikely to be related to study medication. Shortly after infusion, 1 subject in the 5-mg/kg group experienced self-limiting symptoms (headache, fever, aches, nausea, and emesis) similar to those associated with iv infusion of immunoglobulins [16]. No drug-related serious adverse events or dose-limiting toxicity was reported. There was no clinically relevant drug-related effect on heart rate-corrected QT (QTc) intervals or other electrocardiographic parameters. There were no remarkable findings in laboratory analyses for liver function, renal function, hematology, serum electrolytes, or other parameters.
PK results. Mean serum concentrations of PRO 140 are illustrated in figure 2A. Peak concentrations typically were observed 30-60 min after infusion and averaged 13, 61, and 173 _g/mL for the 0.5-, 2-, and 5-mg/kg groups, respectively. The mean time to last observation (tlast) was 6.1, 18.2, and 18.9 days for these 3 dose groups, and the corresponding AUC∞ values were 11.1, 74.3, and 278 mg x day/L. These values are a <6% extrapolation of the AUC from time 0 to tlast. Mean terminal half-lives were 3.9 and 3.5 days at the 2 highest doses. A smaller mean value (1.5 days) was observed in the low-dose group, perhaps because of insufficient data from the terminal phase. Clearance decreased with increasing dose, and respective mean values were 4.1, 3.0, and 1.6 L/day for the 3 ascending dose groups. The volume of distribution averaged 8.4, 13.8, and 7.1 L for the ascending dose groups. Tests for anti-PRO 140 antibodies were negative, with the exception of a single low-titer (1:40) result on day 59 for a subject receiving 5 mg/kg. This subject experienced no meaningful adverse events during drug administration. The antibodies had no obvious effect on PK metrics or antiviral response. The relationship between antiviral response and PRO 140 exposure was examined using an Emax model (figure 2B). At saturation, the model predicted a mean ± SD log10 decline in HIV-1 RNA level of 2.14 ± 0.22 for single-dose PRO 140. The exposure required to achieve 50% of this reduction was 43.6 ± 15.6 mg x day/L.
Coreceptor tropism and viral susceptibility to PRO 140. One hundred one individuals were screened, and coreceptor tropism data were obtained for 95, of whom 81 (85%) exhibited R5-only tropism and 14 (15%) had dual/mixed tropism. R5-only tropism results were observed in all treated subjects at all time points, with 2 exceptions: 1 of 9 subjects receiving placebo (11%) had an R5-only tropism result at screening and dual/mixed tropism results thereafter, and 1 of 30 subjects receiving PRO 140 (3.3%; 0.5-mg/kg group) had a dual/mixed tropism result on day 8 and R5-only results at all other time points, including the end of the study. In vitro studies have been initiated to characterize the pre- and posttreatment viruses of this subject; these findings will be presented elsewhere.
All pretreatment viruses were susceptible to PRO 140. The mean ± SD relative EC50 values were 1.9 +/- 0.6, 1.7 +/- 0.4, 2.6 +/- 1.1, and 1.9 +/- 0.8 for the placebo and ascending PRO 140 dose groups, respectively. Baseline and end-of-study relative EC50 values were constant to within a factor of 2 for all subjects. Maximum percentage inhibitions (mean ± SD) of 98.9% +/- 1.5% and 99.0% +/- 1.0% were observed at baseline and the end of the study, respectively. Overall, the in vitro analyses revealed no change in viral susceptibility after treatment.
Lymphocyte and chemokine analyses. Baseline CD4+ cell counts ranged from 269 to 853 cells/μL (table 1). At 5 mg/kg, there was a trend toward an increased CD4+ cell count compared with baseline. The mean changes were +129 (p=.055), +96, and +83 cells/μL on days 8, 15, and 22, respectively, and less than +10 cells/μL thereafter. The mean change in CD4+ cell count ranged from _24 to +26 cells/μL in the placebo group. No significant change in CD4+ cell count was observed for the 0.5- and 2-mg/kg groups.
CCR5+ cell counts showed considerable intersubject variation. Predose values ranged from 65 to 736 cells/μL, with a median of 296 cells/μL. There was no depletion of CCR5+ cells after treatment, and end-of-study values ranged from 64 to 1244 cells/μL, with a median of 348 cells/μL. Significant coating of the PRO 140 epitope on CCR5+ lymphocytes was observed after treatment (figure 3). Coating was significant (p<.05) for 2-4 weeks for all PRO 140 treatment groups; no significant CCR5 coating was observed for the placebo group (p>.18 at all time points). RANTES concentrations varied from 2.5 to 24.2 ng/mL at baseline and were unaffected by treatment (p>.18 for all dose levels and time points).
Discussion
The present study provides proof of concept for PRO 140 as a potent antiretroviral agent with prolonged single-dose activity. The viral load reductions observed after treatment with single-dose PRO 140 are similar in magnitude to those reported during 10-14 days of treatment with small-molecule CCR5 antagonists. In addition, the single-dose efficacy of PRO 140 compares favorably with that of other HIV-inhibitory MAbs tested at similar and higher doses. The efficacy and tolerability findings of this study support further development of PRO 140 as a long-acting therapy for HIV-1 infection.
CCR5 represents the first HIV-1 target to be explored with both MAb and small-molecule drugs in similarly designed studies of short-term monotherapy. The small-molecule CCR5 antagonists were administered daily or twice daily (BID) for 10-14 days [17-19]. Although mean maximum reductions were not reported for all of the small-molecule CCR5 antagonists, the 1.70 log10 copies/mL reduction for PRO 140 at 5 mg/kg on day 10 can be compared with the 1.60, 1.62, and 1.66 log10 copies/mL reductions observed on days 11, 14, and 12 for maraviroc (300 mg BID), vicriviroc (50 mg BID), and aplaviroc (600 mg BID) [17, 20, 21]. The similar antiviral effects suggest that PRO 140 and the small molecules were similarly effective in penetrating the major sites of infection to block viral replication.
There are fundamental differences in how PRO 140 and small-molecule CCR5 antagonists recognize CCR5 and inhibit R5 HIV-1. Although PRO 140 binds to hydrophilic extracellular regions on CCR5 and likely inhibits HIV-1 via competitive mechanisms, small-molecule CCR5 antagonists bind to a hydrophobic cavity and inhibit via allosteric mechanisms [14, 21-23]. Despite the fact that the small-molecule CCR5 drugs are potent chemokine antagonists, antiviral concentrations of PRO 140 did not block the natural activity of CCR5 in vitro, although antagonism was observed at higher concentrations [14]. PRO 140 inhibited viruses that are resistant to small-molecule CCR5 antagonists [24, 25], and PRO 140 exhibited potent antiviral synergy in combination with small-molecule CCR5 antagonists in vitro [11]. In many respects (e.g., distinct binding sites on the target, allosteric vs. competitive inhibition, synergistic activity, and limited viral cross-resistance), the distinctions between PRO 140 and small-molecule CCR5 antagonists parallel the distinctions between nucleoside-analogue and nonnucleoside reverse-transcriptase inhibitors.
PRO 140 bound to CCR5 without depleting CCR5+ cells from the circulation. CCR5 coating was maximal for 1 week at 0.5 mg/kg and for 2 weeks at the higher doses. These kinetics were broadly consistent with the timing of antiviral effects. However, the differences in the extent of CCR5 coating at 2 and 5 mg/kg were less pronounced than the differences in the antiviral effects observed at these doses. This observation may reflect differences in the sensitivities of the 2 measurements.
There was a trend toward increased CD4+ cell counts in the 5-mg/kg group for 1-3 weeks after treatment. This finding may reflect redistribution of these cells from tissues into the periphery, which often accompanies potent antiviral suppression. However, regardless of the mechanism, the trend is encouraging, given that CD4+ cells are an established surrogate marker for monitoring the course of HIV-1 therapy.
The clearance of PRO 140 was similar to that reported for CCR5MAb004, a human IgG4 MAb to CCR5 [26, 27]. However, both MAbs were cleared more rapidly than total IgG4 [28, 29]. The PK metrics of PRO 140 are consistent with a saturable, antigen-mediated clearance pathway. CCR5 internalizes constitutively in vitro [30], and internalizing antigens can accelerate the clearance of cognate MAbs in vivo [31, 32]. Although there was no obvious correlation between PRO 140 clearance and baseline CCR5+ cell count as measured by flow cytometry, this assay measures circulating CCR5+ cells and not the potentially greater reservoir of tissue-resident CCR5+ cells. The antiviral effects of PRO 140 were also independent of baseline HIV-1 RNA level and circulating CD4+ cell count.
At present, no MAbs are approved for HIV-1 therapy. Modest antiviral effects were observed for MAbs that bind the HIV-1 envelope glycoproteins, but greater promise was reported for MAbs to host cell receptors (reviewed in [33]). Single iv doses of CCR5MAb004 and the CD4 MAb TNX-355 have been administered to HIV-infected individuals as monotherapy or functional monotherapy [26, 27, 34]. Neither mean maximum reductions in HIV-1 RNA nor day 10 data were reported for CCR5MAb004 or TNX-355; however, mean viral load reductions on days 8, 15, 22, and 29 are available for these MAbs and PRO 140. The mean viral load reduction on day 8 for PRO 140 at 5 mg/kg (1.54 log10 copies/mL) compares favorably with the largest mean reductions reported at any fixed time point for CCR5MAb004 (1.0 log10 copies/mL on day 15) and TNX-355 (1.3 log10 copies/mL on day 15) at doses as high as 40 mg/kg and 25 mg/kg, respectively. The largest mean viral load reductions for CCR5 MAb004 and TNX-355 were observed at intermediate doses rather than the highest doses tested. In the present study, both the magnitude and the duration of antiviral response increased with PRO 140 dose, and modeling suggests that larger mean reductions may be attainable. In addition, higher doses potentially could support monthly iv dosing with PRO 140.
The potency of PRO 140 supports evaluation of self-administered subcutaneous (sc) formulations; sc MAbs include adalimumab (Humira; Abbott Laboratories), omalizumab (Xolair; Genentech and Novartis), and efalizumab (Raptiva; Genentech). These MAbs are administered weekly to monthly either long term or on a 12-week cycle at doses as high as 375 mg. PRO 140 administered sc has shown favorable tolerability and bioavailability in preclinical studies (data not shown), and sc dosing every 1 or 2 weeks has the potential to provide drug exposure adequate to significantly suppress viral replication.
Toxicities have been reported for all existing HIV-1 drugs and are among the leading causes for switching, discontinuing, and nonadherence to therapy [35]. Hepatotoxicity, postural hypotension, QTc interval prolongation, and possible malignancy have been reported for small-molecule CCR5 antagonists [36-39]. No such safety signals were observed for PRO 140 either preclinically or in the present study. Given that MAbs do not passively diffuse across cellular membranes, PRO 140 may offer an improved or at minimum a nonoverlapping tolerability profile with existing HIV-1 drugs. Favorable tolerability, infrequent dosing, and potent antiviral activity could enhance adherence to therapy.
Recent clinical trials of entry, integrase, and newer protease inhibitors have demonstrated that complete viral suppression (<50 copies/mL) is possible for many patients with multidrug-resistant virus [20, 40-42]. However, incomplete viral suppression was noted in a significant proportion of treated subjects within 24-48 weeks of initiating treatment. Outcomes were markedly improved when 2 or more active agents were present, and current guidelines recommend that subjects remain on a failing regimen until 2 or more active agents are available [43]. New agents provide a means to enhance the care of treatment-experienced individuals.
In summary, this initial study of PRO 140 in HIV-infected individuals provides clear proof of concept for this new agent, and the study demonstrates that potent inhibition of HIV-1 is feasible with a CCR5 MAb. In light of the extended antiviral activity and favorable tolerability exhibited in the present study and the limited potential for drug-drug or food interactions, PRO 140 could offer a new approach to HIV-1 therapy.
|
|
| |
| |
|
|
|