| |
Add-on combination therapy with adefovir dipivoxil induces renal impairment in patients with lamivudine-refractory hepatitis B virus
|
| |
| |
Jnl of Viral Hepatitis Feb 2010
A. Tamori 1 , M. Enomoto 1 , S. Kobayashi 1 , S. Iwai 1 , H. Morikawa 1 , H. Sakaguchi 1 , D. Habu 2 , S. Shiomi 3 , Y. Imanishi 4 and N. Kawada 1
1 Department of Hepatology, Osaka City University Graduate School of Medicine, Osaka, Japan ; 2 Department of Medical Nutrition, Osaka City University Graduate School of Life Science, Osaka, Japan ; 3 Department of Nuclear Medicine, Osaka City University Graduate School of Medicine, Osaka, Japan ; and 4 Department of Metabolism, Endocrinology and Molecular Medicine, Osaka City University Graduate School of Medicine, Osaka, Japan
Correspondence to Akihiro Tamori, MD, Department of Hepatology, Osaka City University Graduate School of Medicine, 1-4-3 Asahimachi, Abeno-ku, Osaka 545-8585, Japan. E-mail: atamori@med.osaka-cu.ac.jp
Summary.
Combination therapy with adefovir dipivoxil (ADV) and lamivudine (LAM) is recommended for patients infected with LAM-refractory hepatitis B virus (HBV). However, the effects of such therapy on renal function and serum phosphorus levels have not been fully evaluated. Combination therapy with ADV and LAM was given to 37 patients infected with LAM-refractory HBV, including 17 with hepatic cirrhosis. Serum HBV DNA levels decreased to below 2.6 log10 copies/mL in 23 (62%) of 37 patients at 12 months, 25 (78%) of 32 patients at 24 months, and 16 (84%) of 19 patients at 36 months. Except for one cirrhotic patient, serum alanine aminotransferase levels were below 50 IU/L in all patients during combination therapy. Serum creatinine levels increased in 14 (38%) of 37 patients, and serum phosphate levels decreased to below 2.5 mg/mL in 6 (16%) of 37 patients during combination therapy. Patients who received combination therapy for 36 months or longer had a significantly incidence of elevated serum creatinine levels. Fanconi syndrome occurred in a 57-year-old woman with cirrhosis after ADV was added to LAM. Combination therapy with ADV and LAM can maintain biochemical remission in patients with LAM-refractory HBV. However, the dosing interval of ADV should be adjusted according to renal function and serum phosphate levels in patients receiving long-term treatment.
Introduction
Hepatitis B virus (HBV) is a widely prevalent pathogen that causes chronic hepatitis, hepatic cirrhosis, and hepatocellular carcinoma (HCC) [1]. Interferon and nucleos(t)ide analogues are used for antiviral therapy in patients infected with chronic hepatitis B infection [2,3]. Lamivudine (LAM) is the first nucleoside analogue approved for the treatment of HBV-infected liver disease. LAM suppresses HBV replication in most patients and improves hepatic inflammation [4]. However, more than 60% of patients with HBV who receive long-term LAM therapy become resistant to the drug within 4 years after starting treatment [5]. For LAM-resistant HBV, a switching-to adefovir dipivoxil (ADV) or entecavir (ETV) treatment is a choice of treatment [6,7]. However, in ADV monotherapy for LAM-refractory chronic hepatitis B, virological breakthrough due to development of ADV resistant mutations occurred in three (21%) of 14 patients within 18 months [8]. In another study, ETV-resistant mutations developed in 12 (8%) of 151 patients with LAM-refractory chronic hepatitis B who received 1 mg ETV once daily for 2 years [7]. In contrast, combination therapy with ADV and LAM decreased HBV DNA levels in patients with LAM-resistant HBV and maintained the effect without virological and biochemical breakthroughs for 3 years [8,9]. In one study performed in Japan, ADV-resistant mutations occurred in only 2 of 129 patients with LAM-refractory chronic hepatitis B who received ADV plus LAM for 2 years [10]. These studies concluded that combination therapy with ADV and LAM was the treatment of choice for patients with LAM-resistant HBV.
An important limitation of previous studies of combination therapy with ADV and LAM is the lack of adequate safety data. Monotherapy with LAM is given to more than 30 000 patients with HBV-related chronic liver disease in Japan. LAM has not been reported to induce serious adverse effects, except for the emergence of LAM-resistant HBV. On the other hand, nephrotoxicity is the dose-limiting adverse effect in the use of ADV. In phase III, randomized, controlled studies, there were no increases from baseline of 0.5 mg/dL or greater in the serum creatinine level and no confirmed instances of serum phosphate levels below 2.0 mg/dL during 48 weeks of monotherapy with 10 mg ADV [11]. However, the renal safeness of combination therapy with ADV and LAM in long-term use is not enough to be evaluated. In particular, there are few reports about decrease of serum phosphate during the combination therapy. In our hospital, an open-label study of long-term add-on treatment with ADV in patients with LAM-refractory HBV has been in progress since 2003.
In the present study, we investigated the incidence of serum creatinine increase and hypophosphorus in patients with HBV-related chronic liver diseases during long-term ADV and LAM combination therapy. In addition, clinical characteristics of patients in whom mild renal impairment was observed were evaluated, since early detection of adverse event is important.
Results
Baseline characteristics of patients with LAM-refractory HBV
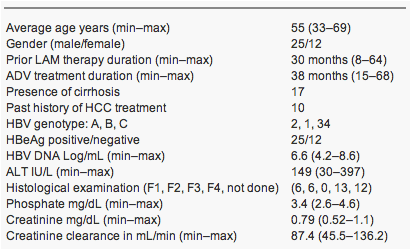
The HBV genotype was A in 2 patients, B in 1, and C in 34 (Table 1). At the start of add-on treatment with ADV, HBeAg was positive in 25 of the 37 patients. On analysis of the LAM resistant motif, M204I mutations were detected in 12 patients, and M204V mutations were detected in 12. In 11 patients, both mutated motifs of HBV were detected. In one of the patients with both mutations, an additional mutation (L80I) was found. LAM-resistant motifs were not examined in the other two patients. ADV-resistant mutations (A181V/T or N236T) were not detected before the start of ADV add-on treatment in any patient with LAM-resistant HBV.
Virological response to combination therapy
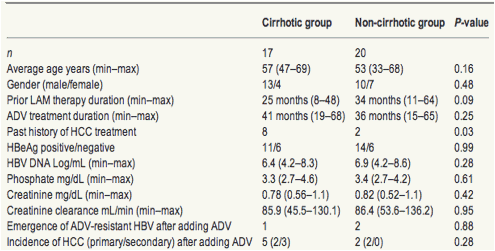
HBV DNA decreased to below 2.6 log10 copies/mL in 23 (62%) of 37 patients with LAM-refractory HBV at 12 months, 25 (78%) of 32 patients at 24 months, 16 (84%) of 19 patients at 36 months, and 8 (80%) of 10 patients at 48 months (Fig. 1). In three patients with HBeAg, the HBV DNA level did not decrease to below 4 log10 copies/mL during more than 30 months of combination therapy. In two of these patients, who did not have cirrhosis, A181T mutations were detected 18 months after the start of ADV add-on therapy. A181V/T or N236T mutation was not detected in the other patient, who had cirrhosis and genotype C (Table 2). Combination therapy reduced the HBV DNA level to below 2.6 log10 copies/mL in 10 (59%) of 17 patients with hepatic cirrhosis at 12 months, and in 12 (80%) of 15 patients with hepatic cirrhosis at 24 months. Among the 10 patients who received curative treatment for HCC before add-on treatment with ADV, combination therapy reduced the HBV DNA level to below 2.6 log10 copies/mL in 6 (60%) of 10 patients at 12 months and 5 (83%) of 6 patients at 24 months.
Table 2 Comparison of clinical characteristics and events between the cirrhotic and non-cirrhotic group during combination therapy
Biochemical and serological responses to combination therapy
Serum ALT levels decreased to below 50 IU/L in 26 (70%) of 37 patients with LAM-resistant HBV at 6 months, 27 (73%) of 37 patients at 12 months, 26 (81.2%) of 32 patients at 24 months, 17 (89.4%) of 19 patients at 36 months, and 9 (90%) of 10 patients at 48 months. Except for one patient with hepatic cirrhosis, serum ALT levels fell to below 50 IU/L in all patients who received ADV and LAM combination therapy (Fig. 1). HBeAg became undetectable in 6 (24%) of 25 patients at 12 months, 10 (48%) of 21 patients at 24 months, and 5 (38%) of 13 patients at 36 months.
Incidence of HCC
In 2 of 10 cirrhotic patients who received curative treatment for HCC, secondary HCC appeared during combination therapy with ADV and LAM (Table 2). One patient with HCC recurrence continuously had a serum HBV DNA level of more than 4 log10 copies/mL. In 4 (14.8%) of 27 patients with LAM-refractory HBV, primary HCC appeared after adding ADV to LAM treatment. Two of the four patients in whom primary HCC developed had hepatic cirrhosis at the start of add-on treatment with ADV. In one patient with cirrhosis, the serum HBV DNA level exceeded 4 log10 copies/mL at the time of diagnosis of HCC. In the other three patients, serum HBV DNA levels remained below 2.6 log10 copies/mL on the occurrence of HCC. One patient died of advanced HCC 33 months after the start of combination therapy with ADV and LAM.
Renal impairment and hypophosphataemia during combination therapy
Serum creatinine levels gradually increased after the start of add-on treatment with 10 mg of ADV in 14 (38%) of 37 patients. In patients who received combination therapy for longer than 36 months or longer, the incidence of elevated serum creatinine levels increased significantly (Table 3). Serum creatinine did not increase in three patients whose HBV DNA level remained above 4 log10 copies/mL during more than 30 months of combination therapy. Except for these patients, there were no differences in clinical course between patients with creatinine increase and patients without it. In four patients (11%) whose serum creatinine levels increased to above 1.4 mg/dL, the dosing interval of ADV was adjusted to every 2 days. After this adjustment, serum creatinine levels decreased without reactivation of HBV replication. Two of these patients had progression to hepatic cirrhosis at the beginning of add-on treatment with ADV. The other patient without cirrhosis had hypertension as a complication. Creatinine clearance in this patient had decreased to 53.6 min/mL at the beginning of add-on treatment with ADV. The left patient did not have other complications.
Table 3 Comparison of clinical characteristics between patients with and those without an increase in serum creatinine levels
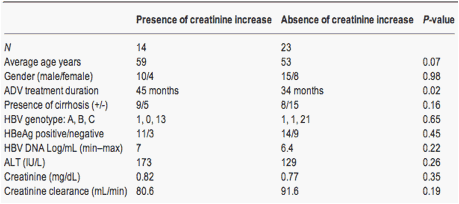
Serum phosphate levels decreased to below 2.5 mg/mL in 6 (16%) of 37 patients. Serum creatinine levels increased in all six of these patients. No other variables correlated with decreased serum phosphate levels.
Case presentation
In December 2005, ADV was added to LAM therapy in a 57-year-old woman infected with YIDD-mutated HBV. She was given a clinical diagnosis of hepatic cirrhosis with no other complications, including HCC. Before combination therapy, HBeAg was negative, and the HBV DNA level was 6.5 log10 copies/mL. Creatinine and phosphate levels in serum were 0.56 and 3.8 mg/dL, respectively. The creatinine clearance was 56.7 mL/min. Nine months after starting combination therapy, severe lumbago developed. At 14 months, oedema of the legs and joint pain of the feet occurred. The serum alkaline phosphatase level increased to 800 IU/L, and she was admitted to our hospital. The serum HBV DNA level had decreased to less than 2.6 log10 copies/mL (Fig. 2). The serum phosphate level had decreased to 1.9 mg/dL, and the serum creatinine level was 0.88 mg/dL. Bone scintigraphy showed multiple-hot spots (Fig. 3). Urinalysis revealed glucosuria, proteinuria and generalized aminoaciduria. In addition, the serum fibroblast growth factor 23 level had decreased to 3 pg/mL (normal range, 10Ð50 pg/mL). On the basis of these results, the patient was given a clinical diagnosis of osteomalacia due to secondary (drug-induced) Fanconi syndrome. The dosing interval of ADV was adjusted to once every 3 days, with replenishment of phosphate. After 1 month, the serum phosphate level increased dramatically to the normal range. The serum HBV DNA level has been maintained below 1.7 log10 copies/mL for 12 months with a combination of LAM daily and ADV every 3 days. In November 2008, the abnormal spots in bone improved without the continuous replenishment of phosphate.
Discussion
Our study showed that the addition of ADV to LAM therapy decreased serum HBV DNA levels and improved elevated ALT levels in patients with LAM-refractory HBV. Combination therapy with ADV and LAM continued to be effective for more than 3 years in 19 patients. In 2 (5%) of 37 patients with LAM-resistant HBV, ADV-resistant mutants (A181T) were detected 18 months after the start of combination therapy. The effects of ADV add-on therapy in the present study were consistent with the results of previous studies [9,10]. In particular, our data showed that the antiviral effects of combination therapy in patients with hepatic cirrhosis and those who received treatment for HCC were not inferior to the effect in patients with chronic hepatitis. Three patients have received ADV add-on combination therapy for more than 5 years without elevation of HBV DNA levels. The latest examinations showed that ALT has remained below 40 IU/mL in 36 patients, including three infected with AVD-resistant HBV, during combination therapy. Although the sample size was small, our results suggest that combination therapy suppressed LAM-refractory HBV DNA levels in patients with cirrhosis or HCC and consistently improved elevated ALT levels, even after the emergence of A181T mutants.
Renal impairment is one of the most important adverse effects of ADV. The dosing interval of ADV should therefore be adjusted according to the creatinine clearance of patients. However, guidelines for dosage adjustment in patients given ADV plus LAM are lacking. In the present study, we evaluated the safety of treatment with ADV 10 mg daily added to LAM. Creatinine clearance was above 50 mL/min in all except one patient. In four (11%) patients, including one with a low creatinine clearance, the serum creatinine level increased to more than 1.4 mg/dL. After the interval between doses of ADV was adjusted to every 2 days, serum creatinine levels improved, with no increase in HBV DNA levels. A long-term study safety and efficacy study of ADV monotherapy showed that the serum creatinine level increased by at least 0.5 mg/dL as compared with the baseline value in 5 (8%) of 65 patients at 240 months [18]. In previous studies of ADV plus LAM combination therapy, daily treatment with ADV was shifted to every 2 days in 4 (3%) of 132 patients or 10 (7%) of 145 patients because the serum creatinine level rose by more than 0.5 mg/dL [9,10]. To evaluate slight alterations in renal function, we defined elevation of the serum creatinine level as a 30% increase from the baseline value. Elevations of serum creatinine were detected in 14 (38%) patients. The incidence of elevated serum creatinine levels was significantly higher in patients who received ADV plus LAM combination therapy for 36 months or longer. These results suggested that patients who receive long-term combination therapy are at risk for renal impairment. In the present study, 19 of 37 patients, including 17 with cirrhosis and 2 without cirrhosis who had received treatment for HCC, had a high risk of HCC. Computed tomography with contrast medium was repeatedly performed to detect the onset or recurrence of HCC. In addition to ADV, contrast medium might have contributed to renal impairment.
Some drugs have been reported to induce renal proximal tubulopathy in association with decreased reabsorption of phosphate. Serum phosphate concentrations were not enough to be evaluated in patients given ADV and LAM combination therapy. In our study, the serum phosphate level decreased to below 2.5 mg/mL in 6 (16.2%) of 37 patients during combination therapy. Serum creatinine levels increased in all six of these patients. It was suggested that decreased phosphate levels were accompanied by increased creatinine levels. In particular, Fanconi syndrome developed in one patient in whom the serum phosphate level decreased to 1.9 mg/dL. To our knowledge, this is the first case of combination therapy-related Fanconi syndrome to be reported. Tenofovir disoproxil fumarate (TDF), an anti- HIV drug, was approved for the treatment of patients with HBV in the United States [19,20]. This is an acyclic nucleotide analogue with a molecular structure related to that of ADV. Recent study showed that 300 mg of TDF treatment had superior antiviral effect to patients with chronic hepatitis B compared to 10 mg of ADV treatment. The serious clinical adverse event related to TDF did not occurred during 48 weeks of the administration [21].
However, Fanconi syndrome was reported to have developed in a 45-year-old cirrhotic woman coinfected with HIV and HCV during treatment with TDF [22]. To quantify the risk of Fanconi syndrome, renal proximal tubulopathy should be assessed in large numbers of patients with HBV during nucleotide therapy, including a combination of ADV and LAM.
In conclusion, our study showed that combination therapy with ADV and LAM effectively suppressed HBV replication and maintained biochemical remission in patients who have chronic liver disease associated with LAM-refractory HBV. However, it is important to closely monitor renal function and serum phosphate levels in patients with cirrhosis, as well as those who receive long-term antiviral therapy. Renal impairment improved without increased HBV replication after adjusting the dosing interval of ADV.
Methods
Patients
The study group comprised 37 consecutive Japanese patients with LAM-refractory HBV who received a combination of 100 mg of LAM plus 10 mg of ADV daily for more than 1 year in Osaka City University Hospital between September 2002 and November 2008 (Table 1). All patients had a 1.5-log10 copies/mL or greater increase in the serum HBV DNA level during LAM treatment. No patient had a history of treatment with other nucleoside analogues, such as ETV and famciclovir. Patients were excluded if they had antibodies to hepatitis C virus or human immunodeficiency virus (HIV). Serum creatinine levels were under 1.2 mg/dL in all patients, and creatinine clearance was over 50 mL/min in all patients except one, who had a value of 45.5 mL/min. Before adding ADV to LAM, 10 patients received curative treatment for HCC. Liver biopsy was performed in 25 patients. Hepatic cirrhosis was histologically diagnosed in 13 patients and clinically diagnosed in 4 patients with oesophageal varices. All patients gave written informed consent to undergo viral sequencing and to participate in this study.
Table 1 Patients' characteristics at the start of adding adefovir to lamivudine
Analysis of serological markers for HBV
Hepatitis B surface antigen (HBsAg), hepatitis B e antigen (HBeAg), and antibodies to HBeAg (anti-HBe) in patient sera were tested by enzyme immunoassay, radioimmunoassay, or both, using commercially available kits (Dainabott, Tokyo, Japan).
Analysis of DNA markers for HBV
Genotypes of HBV were identified by enzyme-linked immunosorbent assay with monoclonal antibodies to type-specific epitopes in the preS2-region (Institute of Immunology, Tokyo, Japan), as described elsewhere [12]. HBV DNA was measured by transcription-mediated amplification (TMA) with a hybridization protection assay (Chugai Diagnostics, Tokyo, Japan) [13]. The detection range of the TMA assay was between 3.7 and 8.7 log10 copies/mL of HBV DNA. If HBV DNA was not detected by this method, we tried again, using the polymerase chain reaction (PCR)-based Amplicor Monitor test (Roche Diagnostics, Tokyo, Japan) [14]. The detection range of the PCR assay was between 2.6 and 7.6 log10 copies/mL. From 1 March 2008, HBV DNA was measured by the Taqman HBV test (Roche Diagnostics, Tokyo, Japan). The detection range of the Taqman HBV test is between 1.8 and 8.8 log10 copies/mL of HBV DNA [15].
LAM-resistant mutations in the tyrosineÐmethionineÐaspartateÐaspartate motif of the HBV polymerase gene, L80I, and ADV-resistant mutations were examined by a line-probe assay (INNO-LiPA HBV DR, Innogenetics NV, Belgium) [16].
Chemical markers in serum
Levels of alanine aminotransferase (ALT), creatinine, and phosphate were examined before and after combination therapy with ADV and LAM. Creatinine clearance was calculated with Cockcroft's formula before add-on treatment with ADV [17]. An increase in the serum creatinine level was defined as an increase equivalent to more than 130% of the creatinine level at the start of ADV add-on therapy, with no decrease in the absence of additional treatment.
Statistical analysis
Statistical analysis was performed with the Statview SE+Graphics program, version 5.0 (SAS Institute, Cary, NC, USA). The MannÐWhitney U-test was used to compare two continuous variables, and the chi-square test was used to compare two categorical variables. All tests were two-sided, and P values of <0.05 were considered to indicate statistical significance.
Ethical considerations
The study protocol complied with the ethical guidelines of the Declaration of Helsinki (1975) and was approved by the Ethics Committee of Osaka City University Graduate School of Medicine.
|
|
| |
| |
|
|
|