| |
BMS HCV Study in Null Responders:
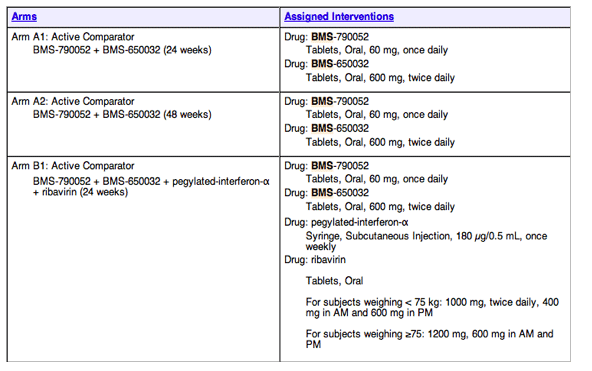
2 oral HCV Drugs Alone vs quad with peg/rbv plus 2 orals
|
| |
| |
'Study to Determine the Effectiveness of Antiviral Combination Therapy to Treat Hepatitis C Virus (HCV) Infected Patients Who Have Previously Failed Standard of Care' - HCV-Infected Genotype 1 Null responders to current standard of care
This study is currently recruiting participants.
Verified by Bristol-Myers Squibb, June 2010
First Received: November 12, 2009 Last Updated: June 29, 2010
Estimated Enrollment: 50
Study Start Date: December 2009
Estimated Study Completion Date: August 2012
Estimated Primary Completion Date: October 2011 (Final data collection date for primary outcome measure)
Eligibility
Ages Eligible for Study: 18 Years to 70 Years
Genders Eligible for Study: Both
Accepts Healthy Volunteers: No
Criteria
Inclusion Criteria:
* Male and female subjects ages 18 to 70 years
* HCV-Infected Genotype 1 Null responders to current standard of care
Exclusion Criteria:
* Evidence of a medical condition associate with chronic liver disease other than HCV
* History of variceal bleeding, hepatic encephalopathy, or ascites requiring management with diuretics or paracentesis
* History of Cancer within 5 years of enrollment
* History of gastrointestinal disease or surgical procedure (except Cholecystectomy)
* History of clinically significant cardiac disease
* History of G6PD deficiency
* Documented cirrhosis within 12 months prior to dosing
* Positive for HIV or HBV
* Pregnant
Contacts and Locations
Please refer to this study by its ClinicalTrials.gov identifier: NCT01012895
Contacts
Contact: For participation information at a USA site use a phone number below. For site information outside the USA please email: Clinical.Trials@bms.com
Contact: First line of email MUST contain NCT# & Site#. Only trial sites that are recruiting have contact information at this time.
Locations
United States, California
Advanced Clinical Res Inst Recruiting
Anaheim, California, United States, 92801
Contact: Michael Demicco, Site 004 714-774-7777
Local Institution Not yet recruiting
San Jose, California, United States, 95128
Contact: Site 018
United States, Colorado
University Of Colorado Denver & Hospital Recruiting
Aurora, Colorado, United States, 80045
Contact: Gregory Everson, Site 007 303-724-1877
United States, Maryland
Local Institution Not yet recruiting
Baltimore, Maryland, United States, 21202
Contact: Site 014
United States, Massachusetts
Llc Dba The Research Institute Recruiting
Springfield, Massachusetts, United States, 01107
Contact: Claudia Martorell, Site 002 413-747-5566
United States, Michigan
University Of Michigan Health System Recruiting
Ann Arbor, Michigan, United States, 48109
Contact: Anna S. F. Lok, Site 009
United States, North Carolina
Charlotte Gastroenterology & Hepatology, Pllc Recruiting
Charlotte, North Carolina, United States, 28207
Contact: John S. Hanson, Site 016 704-375-9485
Carolinas Center For Liver Disease Recruiting
Statesville, North Carolina, United States, 28677
Contact: Robert W. Reindollar, Site 013 704-978-1144
United States, Texas
The Liver Institute At Methodist Dallas Recruiting
Dallas, Texas, United States, 75208
Contact: Reem H Ghalib, Site 012
Alamo Medical Research Recruiting
San Antonio, Texas, United States, 78215
Contact: Eric J. Lawitz, Site 003 210-253-3426
United States, Virginia
Metropolitan Research Recruiting
Fairfax, Virginia, United States, 22031
Contact: Vinod K. Rustgi, Site 010 703-698-9254
Sponsors and Collaborators
Bristol-Myers Squibb
|
|
| |
| |
|
|
|