| |
Outcome of sustained virological responders with histologically advanced chronic hepatitis C (Early HCV Therapy?)
|
| |
| |
Download the PDF Here
(from Jules: again, the 2nd recent study providing I think good evidence to consider early HCV treatment)
Hepatology Sept 2010
Timothy R. Morgan1,2,*, Marc G. Ghany3, Hae-Young Kim4, Kristin K. Snow4, Mitchell L. Shiffman5, Jennifer L. De Santo6, William M. Lee7, Adrian M. Di Bisceglie8, Herbert L. Bonkovsky9, Jules L. Dienstag10, Chihiro Morishima11, Karen L. Lindsay12, Anna S. F. Lok13
"Durability of SVR: Ninety-one SVR patients had follow-up HCV R NA testing performed an average of 78.6 ± 15.9 months (range: 22.1-99.6 months) after achieving SVR, and 90 of the 91 (99%) had undetectable HCV RNA in serum"
"In summary, we found that patients with advanced chronic hepatitis C who achieved SVR had significantly lower rates of death from any cause or liver transplantation, and of liver-related morbidity and mortality, compared to patients who failed to eliminate HCV with treatment (NR). Still, patients who achieved SVR remained at risk of HCC for at least 6 years after achieving SVR. Our study also showed that patients who had temporary, but complete viral suppression (BT/R) were less likely to die or undergo liver transplantation, or to experience liver-related complications than patients in the NR group, indicating that the duration of clinical benefit may outlast the period of actual viral suppression. Importantly, laboratory tests associated with liver-disease severity (e.g., platelet count, albumin) continued to improve after patients achieved SVR. Overall, our data indicate that patients with chronic hepatitis C and advanced hepatic fibrosis who achieve SVR have a marked reduction in the risk for death or liver transplantation, or of liver-related complications, and continued improvement in laboratory markers of liver function in the 5-6 years following successful viral eradication."
"An interesting and heretofore unreported finding was the intermediate risk of clinical outcomes in the BT/R group, between the risk of that for the NR and the SVR groups. In particular, the adjusted risk of death from any cause/liver transplantation or of any liver-related outcome was significantly lower in the BT/R group than in the NR group. The risks of decompensated liver disease, HCC, and liver-related death or liver transplantation were also lower in the BT/R group than in the NR group, although these differences did not reach statistical significance. These findings suggest that complete viral suppression is associated with a reduced risk of clinical outcomes and that the benefits may outlast the period in which HCV RNA is undetectable.16"
Abstract
Retrospective studies suggest that subjects with chronic hepatitis C and advanced fibrosis who achieve a sustained virological response (SVR) have a lower risk of hepatic decompensation and hepatocellular carcinoma (HCC). In this prospective analysis, we compared the rate of death from any cause or liver transplantation, and of liver-related morbidity and mortality, after antiviral therapy among patients who achieved SVR, virologic nonresponders (NR), and those with initial viral clearance but subsequent breakthrough or relapse (BT/R) in the HALT-C (Hepatitis C Antiviral Long-Term Treatment Against Cirrhosis) Trial. Laboratory and/or clinical outcome data were available for 140 of the 180 patients who achieved SVR. Patients with nonresponse (NR; n = 309) or who experienced breakthrough or relapse (BT/R; n = 77) were evaluated every 3 months for 3.5 years and then every 6 months thereafter. Outcomes included death, liver-related death, liver transplantation, decompensated liver disease, and HCC.
Median follow-up for the SVR, BT/R, and NR groups of patients was 86, 85, and 79 months, respectively.
At 7.5 years, the adjusted cumulative rate of death/liver transplantation and of liver-related morbidity/mortality in the SVR group (2.2% and 2.7%, respectively) was significantly lower than that of the NR group (21.3% and 27.2%, P < 0.001 for both) but not the BT/R group (4.4% and 8.7%). The adjusted hazard ratio (HR) for time to death/liver transplantation (HR = 0.17, 95% confidence interval [CI] = 0.06-0.46) or development of liver-related morbidity/mortality (HR = 0.15, 95% CI = 0.06-0.38) or HCC (HR = 0.19, 95% CI = 0.04-0.80) was significant for SVR compared to NR. Laboratory tests related to liver disease severity improved following SVR.
Conclusion: Patients with advanced chronic hepatitis C who achieved SVR had a marked reduction in death/li ver transplantation, and in liver-related morbidity/mortality, altho ugh they remain at risk for HCC.
Clinical Outcomes of SVR Patients.
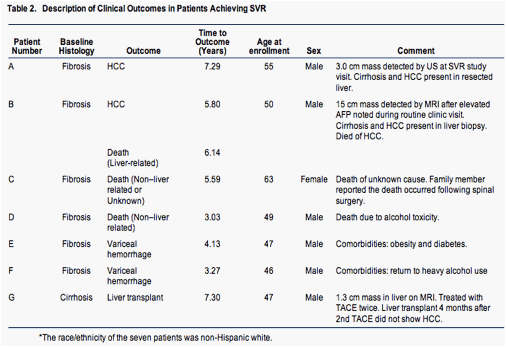
Five patients who achieved SVR (3.6%) had six liver-related clinical outcomes (Table 2). One patient (patient A) had a 3-cm lesion detected on ultrasound performed for his amended study clinic visit, 7.3 years after his baseline visit and 5.8 years after achieving SVR. At entry into the HALT-C Trial, he had a liver biopsy with an Ishak fibrosis score of 4 and his platelet count was 112,000/mm3. The resected specimen revealed a well-differentiated HCC; cirrhosis was present in the nontumor liver. Another patient (patient B) who had an Ishak fibrosis score of 3 on his baseline liver biopsy was found to have a 15-cm lesion on magnetic resonance imaging performed to evaluate an elevated AFP during a routine follow-up visit 5.8 years after his baseline visit and 4.4 years after achieving SVR. Biopsy of the lesion confirmed the presence of HCC and cirrhosis in the adjacent liver. This patient died of progressive HCC 4 months later. After magnetic resonance imaging was performed, a third patient (patient G) was found to have a 1.3-cm liver mass and underwent transarterial chemoembolization twice, followed by liver transplantation, but no tumor was found in the liver explant. This patient did not meet the HALT-C Trial criteria for presumed or definite HCC. Two patients with SVR experienced variceal hemorrhage (patients E and F).
Chronic hepatitis C virus (HCV) infection is a common cause of cirrhosis, hepatocellular carcinoma (HCC), and liver transplantation. Follow-up studies of patients who achieved a sustained virological response (SVR) after antiviral therapy have demonstrated that the majority of patients continue to have undetectable serum HCV RNA, improvement in liver fibrosis, including reversal of cirrhosis, and a reduction in the incidence of decompensated liver disease and HCC compared with subjects who did not achieve an SVR.1-3 These studies notwithstanding, the beneficial effect of achieving an SVR on the outcome of patients with advanced chronic hepatitis C remains incompletely defined because prior studies were retrospective4-7 and included a small number of patients with cirrhosis2 and a relatively limited period of follow-up.8 In addition , few data are available on patients in the United States, because most of these studies were conducted in Japan or Europe.4-8 Furthermore, the beneficial effect of interferon and ribavirin treatment on the outcomes of patients with advanced hepatitis C who achieved viral clearance during treatment and who relapsed after discontinuation of treatment has not been established clearly.6
The Hepatitis C Antiviral Long-Term Treatment Against Cirrhosis (HALT-C) Trial was a multicenter study involving more than 1000 patients in the United States with advanced chronic hepatitis C and nonresponse to previous treatment with interferon-based therapy.9 During the lead-in phase of the HALT-C Trial, 1145 patients were treated with a combination of pegylated interferon and ribavirin; of these, 180 achieved SVR. Patients who did not achieve SVR entered the randomized phase of the HALT-C Trial and were followed prospectively for the development of fibrosis progression, decompensated liver disease, HCC, and death. The aim of the current study was to evaluate the effect of achieving SVR on overall mortality and on liver-related morbidity and mortality in this large, prospectively followed cohort of patients from the United States with advanced chronic hepatitis C.
DISCUSSION
We report here the results of a prospective, long-term follow-up study to evaluate the effect of achieving SVR with pegylated interferon and ribavirin treat ment on death from any cause or liver transplantation, and on liver- related morbidity and mortality, in a large cohort of patients in the United States with chronic hepatitis C and bridging fibrosis or cirrhosis. Patients who achieved SVR were compared with two groups of patients who were enrolled into the HALT-C Trial at the same time: (1) patients who failed to respond to peginterferon and ribavirin (NR) and (2) patients with virologic clearance at Week 20 but subsequent virologic breakthrough during combination antiviral therapy or relapse after completion of therapy (BT/R). In this cohort of patients with advanced chronic hepatitis C, we found that those who achieved SVR after peginterferon and ribavirin treatment had a significantly reduced risk of death from any cause/liver transplantation, and of liver-related morbidity and mortality, when compared with patients in the NR group. Importantly, achieving SVR significantly reduced the risk of developing each component of liver-related morbidity and mortality (i.e., hepatic decompensation, HCC, and liver-related death or liver transplantation) when compared with NR patients.
A strength of this study was the long duration of prospective follow-up. Patients were identified at entry into the HALT-C Trial and were followed for a median of 79 (NR) to 86 (SVR) months after starting their final course of peginterferon/ribavirin. Our findings on the effect of SVR on liver-related clinical outcomes are similar to those of retrospective, and often smaller, studies from Japan5, 7, 13-15 and Europe,6 the results of which supported an approximately 70%-90% reduction in the risk of liver-related clinical outcomes over a follow-up period of 2-6 years in patients achieving an SVR. An interesting observation in our study was the relative rapidity of the effect of achieving an SVR on hepatic decompensation; within 1 year, rates of decompensation among patients with an SVR and those with NR began to diverge. Both SVR patients in whom HCC developed had no discernable cause for ongoing liver damage . These data underscore the continued risk of HCC in patients with advanced chronic hepatitis C, even in those who achieved SVR, as has been noted previously.2, 4, 7-8, 13 Because both SVR patients in whom HCC developed were diagnosed more than 4 years (4.4 and 5.9) after achieving SVR, HCC surveillance should continue for more than 5 years after SVR, and probably for life.
Based on Cox proportional hazard analyses, we found that baseline platelet count was associated independently with all five outcomes, whereas albumin level was associated independently with four outcomes (not with HCC). Age and alkaline phosphatase were associated with the risk of HCC but not with any other outcome. This observation could suggest that the development of HCC follows a different pathway than, and is temporally independent of, the development of other complications of liver disease. In prior studies, age and sex have been associated with risk of HCC, and we reported previously that alkaline phosphatase is associated with the risk of HCC in the HALT-C Trial cohort.11
An interesting and heretofore unreported finding was the intermediate risk of clinical outcomes in the BT/R group, between the risk of that for the NR and the SVR groups. In particular, the adjusted risk of death from any cause/liver transplantation or of any liver-related outcome was significantly lower in the BT/R group than in the NR group. The risks of decompensated liver disease, HCC, and liver-related death or liver transplantation were also lower in the BT/R group than in the NR group, although these differences did not reach statistical significance. These findings suggest that complete viral suppression is associated with a reduced risk of clinical outcomes and that the benefits may outlast the period in which HCV RNA is undetectable.16
Laboratory tests commonly associated with liver disease severity, such as albumin and platelet count, improved in patients achieving an SVR but worsened in patients not achieving SVR. Of particular interest, in these patients with advanced fibrosis who achieved SVR, platelet count and albumin continued to improve between Week 72 and the final visit approximately 5.5 years later. In the only prior report of laboratory tests among SVR patients followed for 5 years, George et al.2 were unable to demonstrate improvement in laboratory tests. Therefore, improvement in liver-related blood tests after achieving an SVR in patients with advanced fibrosis is an original finding. One possible explanation for the difference between the prior report and ours is that the majority of patients followed by George and colleagues2 had mild liver fibrosis, with minimal changes in albumin and platelets that would not be expected to improve during follow-up monitoring. Overall, our data demonstrating improvement in liver-related blood tests, when combined with prior studies demonstrating reduction in liver fibrosis,1-3 suggest that liver function continues to recover in the years following an SVR in patients with advanced fibrosis/cirrhosis.
This study has several limitations. A total of 17% of patients who achieved SVR were lost to follow-up and an additional 6% declined to participate. Potentially, decompensated liver disease or HCC may have developed in these patients; therefore, our results may be an underestimate of the rate of clinical outcomes in patients who achieved SVR. We were able to determine, however, that none of the 30 patients who were lost to follow-up died according to a search of the SSDI performed at the end of amended study. Another potential limitation was the fact that the patients who achieved SVR were not monitored as closely as the BT/R and NR patients and that not all SVR patients were evaluated in person. Nevertheless, medical records with physical examination, blood tests, and/or liver imaging of the patients who were interviewed by phone were reviewed and added reliability to the ascertainment of the occurrence of decompensated liver disease or HCC as of the time of their last follow-up assessment.
In summary, we found that patients with advanced chronic hepatitis C who achieved SVR had significantly lower rates of death from any cause or liver transplantation, and of liver-related morbidity and mortality, compared to patients who failed to eliminate HCV with treatment (NR). Still, patients who achieved SVR remained at risk of HCC for at least 6 years after achieving SVR. Our study also showed that patients who had temporary, but complete viral suppression (BT/R) were less likely to die or undergo liver transplantation, or to experience liver-related complications than patients in the NR group, indicating that the duration of clinical benefit may outlast the period of actual viral suppression. Importantly, laboratory tests associated with liver-disease severity (e.g., platelet count, albumin) continued to improve after patients achieved SVR. Overall, our data indicate that patients with chronic hepati tis C and advanced hepatic fibrosis who achieve SVR have a marked reduction in the risk for death or liver transplantation, or of liver-related complications, and continued improvement in laboratory markers of liver function in the 5-6 years following successful viral eradication.
RESULTS
Demographic and Clinical Data.
Data were obtained on 140 (78%) of the 180 HALT-C Trial patients who achieved SVR. Thirty patients could not be located, and 10 declined to participate. The 40 patients who did not participate did not differ from the 140 who did at baseline or at Week 72 in demographic characteristics, baseline Ishak fibrosis score, or routine blood tests. Specifically, at Week 72 no difference was found between the SVR nonparticipants (n = 40) and participants (n = 140) for key predictors of clinical outcome such as age (49.8 ± 8.02 years versus 50.0 ± 6.12 years for nonparticipants and participants, respectively; P = 0.87), albumin (4.3 ± 0.4 versus 4.2 ± 0.4 g/dL; P = 0.26), platelet count (191 ± 56 versus 191 ± 59 x 1000/mm3; P = 0.97), AFP (3.3 ± 1.5 versus 3.3 ± 1.7 ng/mL; P = 0.88) or alkaline phosphatase (72 ± 20 versus 78 ± 20 IU/mL; P = 0 .27). Three of the 140 SVR patients had died, and copies of death certificates for two of the three were obtained. Of the 137 surviving participants, 70 were seen in clinic whereas 67 were evaluated by telephone interviews supplemented by examination of external medical records. None of the 30 patients with SVR who could not be located were listed as deceased in the online SSDI.
Baseline demographic data as well as clinical and laboratory data on the SVR group and the two comparison groups (BT/R and NR) are shown in Table 1. The three groups differed significantly in race/ethnicity, presence of cirrhosis, hepatitis C genotype, and laboratory values associated with advanced liver disease. SVR patients were more likely to be Caucasian, infected with HCV genotypes other than 1, to have fibrosis (rather than cirrhosis) on baseline biopsy and less likely to have laboratory values associated with advanced liver disease (e.g., low blood counts and albumin, or high INR and AFP) compared with patients in the BT/R and NR groups.
Durability of SVR.
Ninety-one SVR patients had follow-up HCV RNA testing performed an average of 78.6 ± 15.9 months (range: 22.1-99.6 months) after achieving SVR, and 90 of the 91 (99%) had undetectable HCV RNA in serum. The patient with reappearance of HCV RNA was presumed to have a relapse because there were no risk factors for reinfection and genotype 1b was detected at enrollment and at HCV reappearance 15 months following discontinuation of combination treatment. This patient had persistently detectable HCV RNA but no evidence of hepatic decompensation or HCC when last seen 108 months after enrollment in the lead-in phase of the HALT-C Trial.
Clinical Outcomes of SVR Patients.
Five patients who achieved SVR (3.6%) had six liver-related clinical outcomes (Table 2). One patient (patient A) had a 3-cm lesion detected on ultrasound performed for his amended study clinic visit, 7.3 years after his baseline visit and 5.8 years after achieving SVR. At entry into the HALT-C Trial, he had a liver biopsy with an Ishak fibrosis score of 4 and his platelet count was 112,000/mm3. The resected specimen revealed a well-differentiated HCC; cirrhosis was present in the nontumor liver. Another patient (patient B) who had an Ishak fibrosis score of 3 on his baseline liver biopsy was found to have a 15-cm lesion on magnetic resonance imaging performed to evaluate an elevated AFP during a routine follow-up visit 5.8 years after his baseline visit and 4.4 years after achieving SVR. Biopsy of the lesion confirmed the presence of HCC and cirrhosis in the adjacent liver. This patient died of progressive HCC 4 months later. After magnetic resonance imaging was performed, a third patient (patient G) was found to have a 1.3-cm liver mass and underwent transarterial chemoembolization twice, followed by liver transplantation, but no tumor was found in the liver explant. This patient did not meet the HALT-C Trial criteria for presumed or definite HCC. Two patients with SVR experienced variceal hemorrhage (patients E and F).
Two additional SVR patients died, one from alcohol toxicity (patient D) and the other from an unconfi rmed cause, although a family member reported that the death had occ urred after spinal surgery (patient C). These two deaths were not considered to be liver-related.
Models to Predict Clinical Outcomes.
The numbers of patients with death from any cause/liver transplantation and with liver-related outcomes in the SVR, BT/R, and NR groups are presented in Table 3. SVR patients had fewer deaths from any cause/liver transplantation (four or 2.9%) and liver-related outcomes (six outcomes in five [3.6%] patients) compared to BT/R (four or 5.2%) death from any cause/transplant; 15 liver-related outcomes in eight (10.4%) patients and NR (64 or 20.7%) death from any cause/transplant; 148 liver-related outcomes in 78 (25.2%) patients. Because the three patient groups differed in baseline severity of liver disease (e.g., Ishak fibrosis score, platelet count, albumin level; Table 1), we performed a Cox proportional hazard regression analysis (Table 4), adjusting for histological stratum (fibrosis or cirrhosis), age, race, platelet count, AST/ALT ratio, albumin, alkaline phosphatase, AFP, and treatment response (SVR, B T/R, and NR). These variables were selected because they have been associated with liver disease severity or clinical outcomes in prior HALT-C Trial analyses.11, 12 Separate multivariate models were developed to assess risk factors associated with the five outcomes analyzed in this study.
A low baseline platelet count was significantly associated with all five outcomes, whereas a low baseline albumin was a significant risk factor for all outcomes except HCC (Model 4). Age and baseline alkaline phosphatase were also sig nificant risk factors for the development of HCC (Model 4). Achievin g an SVR, when compared with nonresponders, was associated with a significantly lower hazard ratio for each of the five clinical outcomes. Patients with BT/R had a significantly lower hazard ratio for death from any cause/liver transplantation (hazard ratio [HR] = 0.29; 95% confidence interval [CI] = 0.10-0.79) and for any liver-related outcome (HR = 0.46; 95%CI = 0.22-0.96) when compared with NR. Fibrosis stage, race, and baseline AST/ALT ratio were not statistically significant risk factors in any multivariate model.
Adjusted Rates of Clinical Outcomes in SVR, BT/R, and NR Patients.
The cumulative rates of death from any cause/liver transplantation, and of liver-related morbidity and mortality, adjusted for the significant risk factors identified in the Cox models, are shown in Fig. 2 and Supporting Information Table 1. At year 7.5 from enrollment, the adjusted cumulative incidence of outcomes for the SVR, BT/R, and NR patients was, respectively, 2.2%, 4.4%, and 21.3% for death from any cause or liver transplantation (P = 0.0002); 2.7%, 8.7%, and 27.2% for any liver-related outcome (P < 0.0001); 0.9%, 4.7%, and 11.7% for decompensated liver disease (P = 0.012); 1.1%, 5.5%, and 8.8% for HCC (P = 0.077); and 0.99%, 4.1%, and 14.7% for liver-related death or liver transplantation (P = 0.005). For each of the five outcomes, the adjusted cumulative proportion of patients with outcomes was lowest for the SVR group, intermediate for the BT/R group, and highest for the NR group of patients. Although the SVR patients had fewer outcomes than the BT/R patients, the adjusted cumulative incidence was not significantly different between the SVR and the BT/R groups for any of the five outcomes (SVR versus BT/R: P = 0.44 for death or liver transplantation, P = 0.05 for any liver-related outcome, P = 0.07 for decompensated liver disease, P = 0.05 for HCC, and P = 0.13 for liver-related death or liver transplantation). The adjusted cumulative proportion with death or liver transplantation (P = 0.02) or any liver-related outcome (P = 0.04) was significantly lower for the BT/R group when compared with patients from the NR group, but the difference between these two groups was not statistically significant when decompensated liver disease (P = 0.24), HCC (P = 0.93), or liver-related death or liver transplantation (P = 0.11) were analyzed individually.
Because there was no effect of long-term peginterferon treatment on the rate of clinical outcomes,9 the Cox proportional hazard analysis and the adjusted cumulative survival analysis were repeated after including 400 patients who were randomized to the peginterferon alfa-2a (90 µg/week) arm of the HALT-C Trial and who were followed after randomization. Including these patients increased the NR group to 638 and the BT/R group to 148 individuals. All HRs and cumulative outcome analyses were essentially unchanged, except that statistical significance for SVR versus NR was stronger, the HR and adjusted survival analyses for SVR versus BT/R were significant for any liver-related outcome (P < 0.05), and the HR and adjusted survival analyses for BT/R versus NR were significant for liver-related death or liver transplantation (P < 0.05) (data not shown).
Changes in Laboratory Test Results.
Figure 3 shows changes in selected blood tests over time among patients who had blood tests performed at each of the three time points. Among the SVR patients, platelet count and albumin (shown in Fig. 3) as well as AST, ALT, and AFP (data not shown) significantly improved from baseline to the most recent values. A significant improvement in platelet count and albumin was also observed between Week 72 (Month 18), when SVR was attained, and the time of the amended study visit. In contrast, patients from the BT/R and NR groups had a significant worsening of platelet count and bilirubin between baseline and Month 72 visits, and NR patients also had deterioration in albumin and INR during the same time period.
|
|
| |
| |
|
|
|