| |
New protease inhibitors for HCV - Help is on the way
|
| |
| |
Jnl of Hepatology March 2011
Articles in Press
Uncorrected Proof
Dahlene N. Fusco, Raymond T. Chung
Massachusetts General Hospital, GI Unit, 55 Fruit Street, Warren 1007, Boston, MA 02114, USA
" HCV DAAs (direct-acting antivirals in the development pipeline include inhibitors of the viral NS3/4A protease and the viral NS5B RNA-dependent RNA polymerase [2]. Inhibitors of the NS3/4A protease are particularly attractive, since these drugs may have the double-edged action of not only directly inhibiting HCV replication, but also abrogating HCV NS3/4A's subversion of the innate immune response [6]. The NS3/4A compounds currently furthest along in development, the linear protease inhibitors (PIs) boceprevir (Merck) and telaprevir (VX 950, Vertex)......represent an important step forward toward the eradication of HCV [19]. Their major contributions include potent antiviral effects with a lower pill burden and manageable side effect profiles. .....Great care will be necessary to counsel against unnecessary therapy in persons who have low likelihood of responding to PEG/RBV and a PI, including those persons with prior null response to PEG/RBV, and limited hepatic fibrosis. We will therefore likely require a substantial re-education campaign for providers in the coming years. The future appears bright, provided we proceed with an appropriate measure of circumspection......For the particular case of prior null responders, added caution will be required, since retreatment with a single DAA added to SOC may amount to the functional equivalent of DAA monotherapy. Indeed, phase 2 and 3 data in null responder populations have shown high rates of residual viremia enriched for NS3/4A mutations. The success rates of about 30% SVR for prior null responders treated with PEG/RBV and telaprevir suggests that only those null responders with more advanced histologic disease should be offered the combination of PEG/RBV/PI therapy [20]. For prior null responders, referral to a clinical trial including at least two DAA agents with PEG and RBV may need to be considered. While sequencing studies have suggested the gradual regression of most PI-resistant variants to wildtype virus over time [14], the long term implications of these findings is unclear. For instance, it is unknown how quickly PI-resistant virus will be selected with re-exposure to PI-containing regimens. The clonal sequencing of viral populations using sensitive measures such as ultradeep pyrosequencing will help to clarify the correlates of rapid re-selection of resistance. The additional finding that some PI-resistant variants may retain fitness comparable to wildtype virus [15] bolsters the argument that monotherapy, even for a short time, could limit future treatment options for patients enrolled in clinical trials."
CV infects an estimated 170 million people and is the leading indication for liver transplantation. For patients harboring infection with HCV-1, the predominant genotype worldwide, treatment with peginterferon and ribavirin produces sustained virologic response (SVR) in only 45% of patients and is limited by substantial side effects [1]. In the coming months, HCV therapy will enter a new era with the introduction of direct acting antiviral agents (DAAs) [2], [3], [4], [5].
The most advanced HCV DAAs in the development pipeline include inhibitors of the viral NS3/4A protease and the viral NS5B RNA-dependent RNA polymerase [2]. Inhibitors of the NS3/4A protease are particularly attractive, since these drugs may have the double-edged action of not only directly inhibiting HCV replication, but also abrogating HCV NS3/4A's subversion of the innate immune response [6]. The NS3/4A compounds currently furthest along in development, the linear protease inhibitors (PIs) boceprevir (Merck) and telaprevir (VX 950, Vertex), have shown that they can substantially improve SVR when used in combination with peginterferon and ribavirin, and may also permit truncation of therapy [7], [8], [9]. Phase 3 trials of these PIs have been recently completed [2], and their results are anxiously awaited by patients and HCV providers alike. The arrival of these drugs heralds a major advance for HCV therapy, though they are associated with important limitations. Both agents require thrice daily dosing; telaprevir has also been associated with skin reactions, and boceprevir with anemia and dysgeusia. As DAAs, both drugs are associated with rapid development of resistance mutations when given as monotherapy [7], [8], [9] (Table 1).
Table 1.Comparison of 1st and 2nd generation HCV NS3/4A protease inhibitors.
Seiwert et al. Antimicrobial Agents and Chemotherapeutics 2008. % Susser et al. Hepatology 2009.
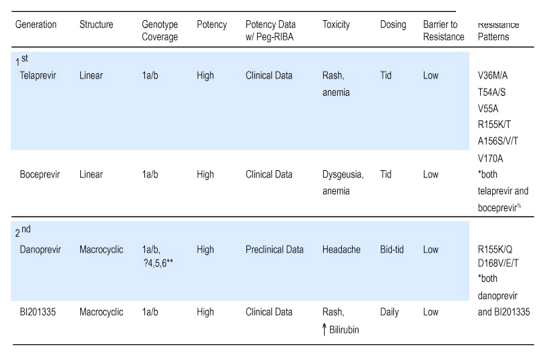
Two phase 1b studies of new NS3/4A protease inhibitors published in this issue of Journal of Hepatology [10], [11] provide promising evidence that HCV treatment options will continue to improve. Forestier et al. present phase 1b study results for the NS3/4A protease inhibitor danoprevir (ITMN 191/RG7227). Danoprevir is a macrocyclic PI notable for its high specificity [12], [13] and high hepatic concentrations [13]. Their study evaluated safety, pharmacokinetics, and antiviral activity of 14days of danoprevir monotherapy administered two or three times daily in multiple ascending doses versus placebo in two groups: 40 treatment naïve genotype 1 infected patients (TNs) and 10 peginterferon and ribavirin non-responders (NRs). Subjects were predominantly male, non-obese, and non-cirrhotic. Danoprevir monotherapy was safe and well tolerated in both groups, with limited side effects. HCV RNA declines occurred in all treatment groups and were dose dependent, with median maximal declines ranging from 1.8 to 3.9logs. Viral kinetics revealed that, overall, 27% of patients experienced viral rebound, 35% plateau, and 38% continuous viral decline, including 3/8 prior non-responders, although nonresponse category (e.g., null versus partial versus relapser) was not specified.
As anticipated, resistance mutations were detected. All patients with virologic rebound, including GT1b patients, selected R155K mutations; another two also had D168V/E/T mutations. Half of patients who experienced virologic plateau selected R155K or D168V/E/T mutations. Not surprisingly, GT1a patients exhibited rebound more often and selected a higher frequency of R155K mutations, likely since 1a virus requires only one nucleotide substitution to generate this mutant, whereas 1b requires two substitutions [14]. Of note, R155K mutants, which may confer cross class resistance [15], persisted following treatment cessation. This finding is consistent with the report that these mutants may display fitness resembling that of wild type HCV [15]. Conversely, D168 mutations may lead to macrocyclic PI resistance but may actually increase susceptibility to linear PIs [15]. These patterns will need to be confirmed with longer term follow-up. Finally, Forestier et al. observed an inferior virologic response in NRs versus TNs, despite equal to higher drug exposure in NRs. This phenomenon suggests an important role for the innate immune response, which is likely to be impaired in most peginterferon and ribavirin non-responders, and may still be critical even in the setting of monotherapy with DAAs. NS3/4A inhibitors may unmask an innate immune response ordinarily suppressed by the NS3/4A protein [6]. This liberated immune response may vary significantly from patient to patient, and may or may not correlate with IL28B genotype [16].
While it will be important to track the results of ongoing phase 2b studies of new generation PIs combined with standard of care, the greatest focus will and should be placed on efforts to combine these agents with other DAA classes, particularly nucleoside HCV NS5b polymerase inhibitors, a DAA class that offers an exceptionally high barrier to resistance [17], [18].
Also in this issue, Manns et al. present a phase 1b multiple ascending dose study of a second generation NS3/4A inhibitor, BI201335, in 34 treatment naïve patients and 19 treatment experienced patients with genotype 1 HCV [11]. Patients were middle aged, non-obese Caucasians and Asians. One major advantage of BI201335 is its once daily dosing schedule. In contrast to the study of Forestier, which was confined to study of monotherapy, Manns et al. evaluated monotherapy followed by combination therapy (BI201335/pegylated interferon/ribavirin) in treatment of naïve patients with >=1log HCV decline after 14days of DAA. Treatment experienced patients received combined therapy only, with no monotherapy phase. Ninety-six percent of patients receiving BI120335 monotherapy achieved a >=2log HCV RNA decline at some point, though, as with danoprevir, monotherapy was associated with the frequent selection of resistance mutations, including R155K and D168V/E. In contrast to danoprevir, however, B1201335 resistance mutations stratified almost completely by HCV subtype (1a developed R155, 1b developed D168V/E), again consistent with a lower barrier to selection of R155K in 1a infection [14]. Side effects were mild, although rash was reported in 4/34 patients on DAA with SOC, and the authors cite a slightly increased frequency of rash in ongoing phase II trials. In addition, indirect hyperbilirubinemia was detected in patients on high dose mono- and combination therapy; the mechanism underlying this phenomenon is currently under investigation [11].
Overall, these two DAA studies represent an important step forward toward the eradication of HCV [19]. Their major contributions include potent antiviral effects with a lower pill burden and manageable side effect profiles. The major drawback of these drugs, as expected, is selection of resistant variants during monotherapy. While monotherapy studies are important for the evaluation of safety, it is clear that monotherapy is not a viable long term strategy, and that DAAs will for now need to be added onto a backbone of peginterferon and ribavirin. For the particular case of prior null responders, added caution will be required, since retreatment with a single DAA added to SOC may amount to the functional equivalent of DAA monotherapy. Indeed, phase 2 and 3 data in null responder populations have shown high rates of residual viremia enriched for NS3/4A mutations. The success rates of about 30% SVR for prior null responders treated with PEG/RBV and telaprevir suggests that only those null responders with more advanced histologic disease should be offered the combination of PEG/RBV/PI therapy [20]. For prior null responders, referral to a clinical trial including at least two DAA agents with PEG and RBV may need to be considered. While sequencing studies have suggested the gradual regression of most PI-resistant variants to wildtype virus over time [14], the long term implications of these findings is unclear. For instance, it is unknown how quickly PI-resistant virus will be selected with re-exposure to PI-containing regimens. The clonal sequencing of viral populations using sensitive measures such as ultradeep pyrosequencing will help to clarify the correlates of rapid re-selection of resistance. The additional finding that some PI-resistant variants may retain fitness comparable to wildtype virus [15] bolsters the argument that monotherapy, even for a short time, could limit future treatment options for patients enrolled in clinical trials.
In summary, the two studies that have been described provide further welcome news that HCV protease inhibitors with improved side effect profiles and decreased dose frequency are on the horizon, just behind the first generation of PIs. We can anticipate that these agents will eventually supplant first generation PIs not only in combination with PEG and RBV, but, more importantly, as cardinal components of future IFN-sparing DAA cocktails. These considerations place a premium on both adherence and tolerability. In 2011 and beyond, HCV providers must now rise to the challenge of quickly assimilating and carefully applying DAA data as it unfolds. Great care will be necessary to counsel against unnecessary therapy in persons who have low likelihood of responding to PEG/RBV and a PI, including those persons with prior null response to PEG/RBV, and limited hepatic fibrosis. We will therefore likely require a substantial re-education campaign for providers in the coming years. The future appears bright, provided we proceed with an appropriate measure of circumspection.
|
|
| |
| |
|
|
|