| |
New Data on Multiple Bristol-Myers Squibb Investigational Hepatitis C Compounds to be Presented at The International Liver Congress 2011 (EASL)
|
| |
| |
Data Demonstrate Focused Execution of the Company's Hepatitis C R&D Strategy
PRINCETON, N.J. --March 07, 2011---New Phase II data on multiple Bristol-Myers Squibb (NYSE: BMY) investigational hepatitis C compounds will be presented at The International Liver Congress (ILC), the 46th annual meeting of the European Association for the Study of the Liver (EASL) in Berlin, Germany, from March 30 to April 2. The data presentations, including three late-breaker presentations, demonstrate the rigorous execution of the company's strategy to develop potential improvements in the care of patients living with Hepatitis C infection by using multiple approaches to target the virus.
"The data at the International Liver Congress reflect the breadth of our hepatitis C pipeline and the multiple approaches we are taking to bring forward potential new options for a disease that today impacts approximately 170 million people worldwide."
Bristol-Myers Squibb will present three late-breaker presentations, including a poster presentation of the first public disclosure of 12-week data on sustained virologic response (SVR) with the NS5A inhibitor BMS-790052 in combination with PEG-Interferon alpha-2a and ribavirin (IFNα/RBV) in treatment-naïve HCV patients. Additionally, SVR 12-week data on quadruple therapy with BMS-790052, the NS3 inhibitor BMS-650032 (protease inhibitor) and IFNα/RBV in null responders will be presented in the late-breaker oral presentation session on Saturday, April 2. Complete early virology response (cEVR) data from the Phase IIb EMERGE study of PEG-Interferon lambda and ribavirin versus IFNα/RBV in treatment-naïve patients will also be presented in a late-breaker oral presentation.
"Bristol-Myers Squibb is focused on advancing the science to address significant unmet medical needs for patients with liver disease," said Brian Daniels, MD, senior vice president, Global Development and Medical Affairs, Research and Development, Bristol-Myers Squibb. "The data at the International Liver Congress reflect the breadth of our hepatitis C pipeline and the multiple approaches we are taking to bring forward potential new options for a disease that today impacts approximately 170 million people worldwide."
BMS-790052 and BMS-650032 were discovered by Bristol-Myers Squibb Research and Development. PEG-Interferon lambda was discovered by ZymoGenetics, Inc., a wholly-owned subsidiary of Bristol-Myers Squibb.
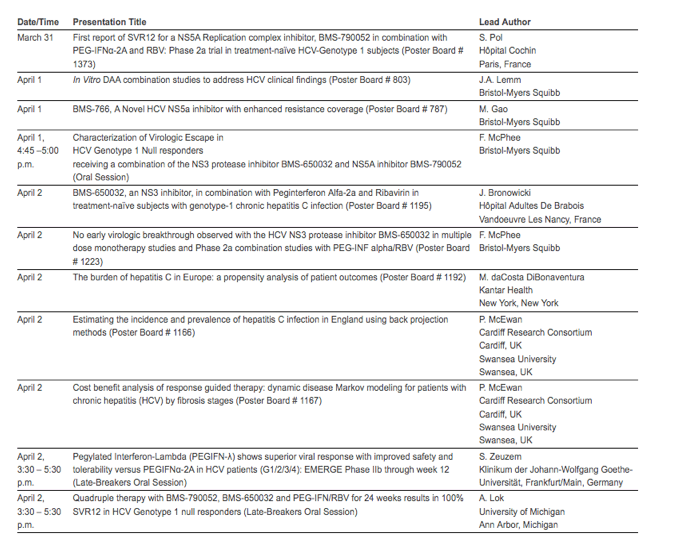
The Bristol-Myers Squibb data presentations at ILC are as follows:
About Bristol-Myers Squibb
Bristol-Myers Squibb is a global biopharmaceutical company whose mission is to discover, develop and deliver innovative medicines that help patients prevail over serious diseases. For more information, please visit http://www.bms.com or follow us on Twitter at http://twitter.com/bmsnews.
Contacts
Bristol-Myers Squibb
Media:
Cristi Barnett, 609-252-6028
cristi.barnett@bms.com
or
Investors:
John Elicker, 609-252-4611
john.elicker@bms.com
|
|
| |
| |
|
|
|