| |
Host targeting cyclophilin inhibitor alisporivir (DEB025)
presents a high barrier to resistance with no
cross-resistance to direct acting antivirals, in Phase 3 Now
|
| |
| |
Reported by Jules Levin
6th International Workshop on Hepatitis C, Resistance
and New Compounds. Cambridge, MA, June 24th, 2011
"Phase III study with DEB025 commenced recently with previously untreated patients infected by the most common form of hepatitis C virus....DEB025 is the first in a new class of drugs called cyclophilin inhibitors. Unlike other compounds in development that target the virus directly, DEB025 is a host targeting antiviral (HTA) that targets so-called host proteins which are essential for the replication of HCV.....Phase II study with the first-in-class antiviral DEB025 (alisporivir) met its primary endpoint for achieving viral cure (24 weeks after stopping treatment) in 76% of patients with chronic hepatitis C[1]. The study involved nearly 300 previously untreated patients infected with the most common form of hepatitis C virus (HCV), the genotype 1 (G1)[1].".....The findings show that 76% of G1 chronic hepatitis C patients treated with DEB025 plus standard of care (pegylated-interferon alfa 2a/ribavirin) achieved superior viral cure (known as sustained viral response, or SVR) compared to 55% of patients on standard of care alone (p=0.008)[1]. Treatment with DEB025 demonstrated a low incidence of adverse events, with discontinuation rates comparable between treatment groups[1]."
EASL: Once daily alisporivir (DEB025) plus Peg-IFN-alfa-2A/ ribavirin results in superior sustained virologic response (SVR24) in chronic hepatitis C genotype 1 treatment-naïve patients - The ESSENTIAL study - (03/31/11)
EASL: Once daily alisporivir (DEB025) plus Peg-IFN-alfa-2A/ ribavirin results in superior sustained virologic response (SVR24) in chronic hepatitis C genotype 1 treatment-naïve patients - The ESSENTIAL study - (03/31/11)
New HCV Drugs at EASL Apr 3, 2011 Đ TMC435 & BI201335 are in phase 3. ... EASL: Once daily alisporivir (DEB025) plus Peg-IFN-alfa-2A/ ribavirin results in superior sustained ...www.natap.org/2011/EASL/EASL_109.htm
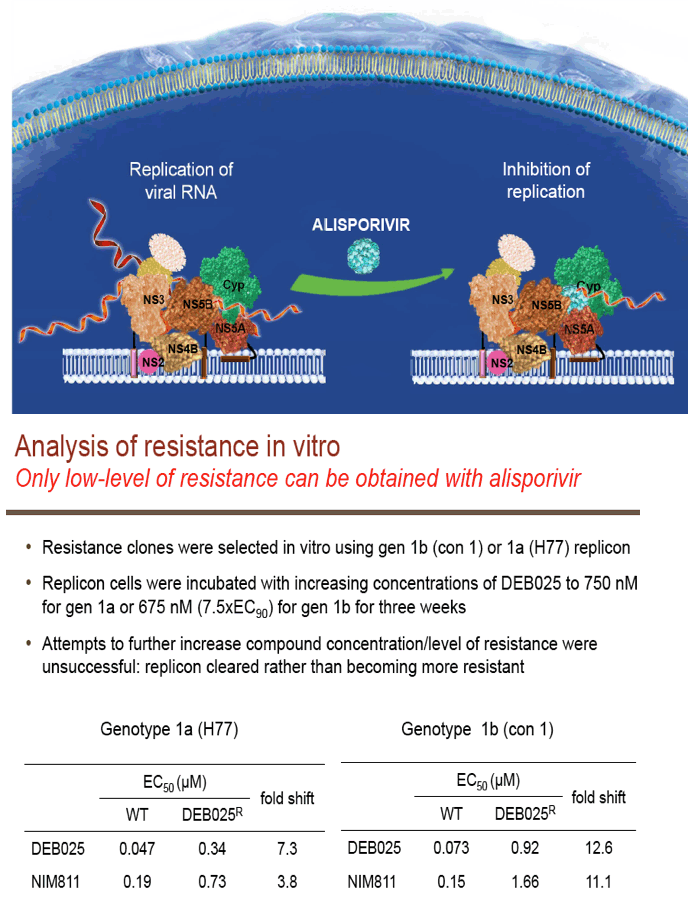
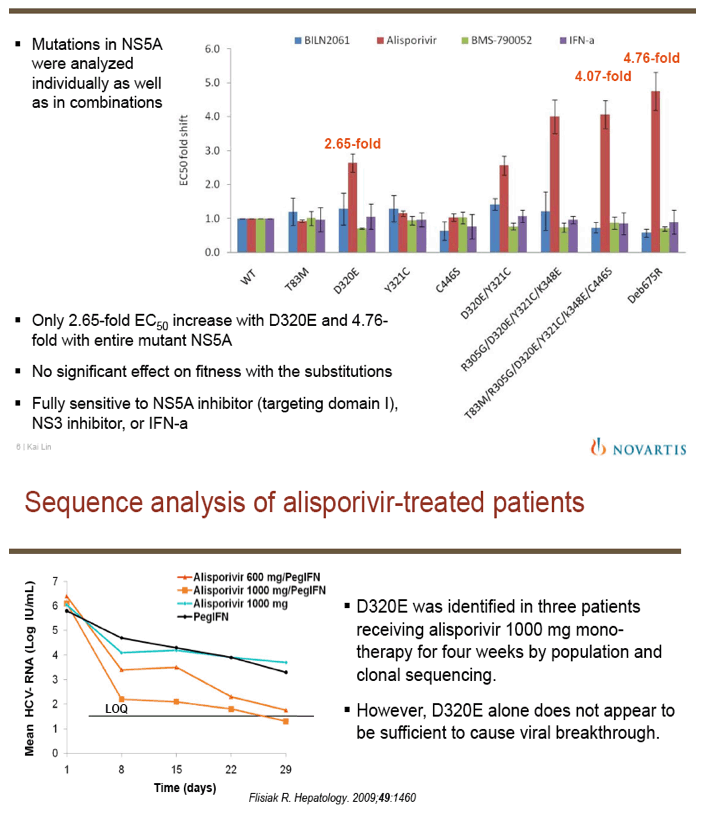
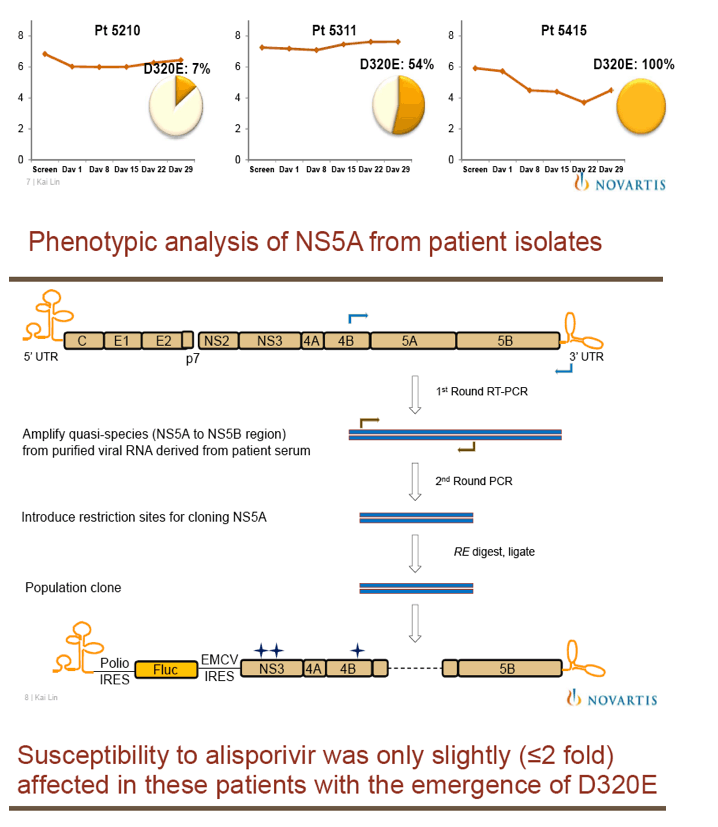
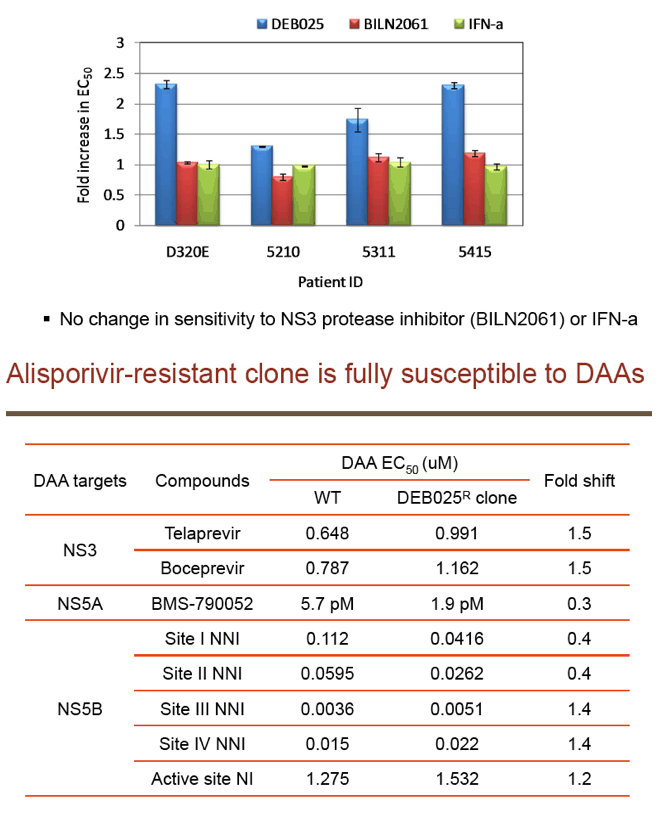
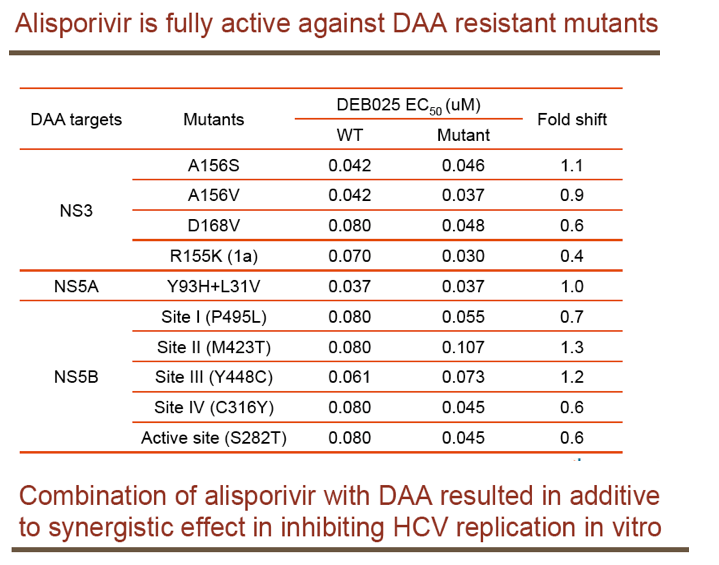
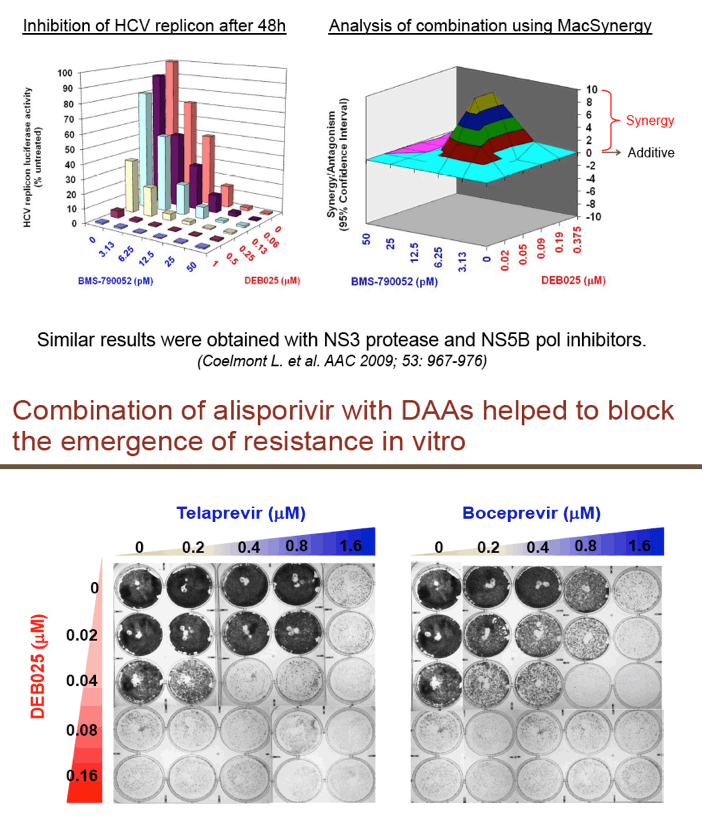
"For all DAAs discovered to date, a single mutation can confer high-level resistance: Resistance develops quickly both in vitro and in patients (except nucs); Thus, it may take ≥3 DAA's to completely suppress resistance in an IFN-free regimen.....A complementary approach is to target host factors that are essential for viral replication, which may present a higher genetic barrier to resistance.....Resistance clones were selected in vitro using gen 1b (con 1) or 1a (H77) replicon: Replicon cells were incubated with increasing concentrations of DEB025 to 750 nM for gen 1a or 675 nM (7.5xEC90) for gen 1b for three weeks.....D320E in NS5A was the only mutation consistently selected in both gen 1a (H77) and gen 1b (con 1) replicons.....Only 2.65-fold EC50 increase with D320E and 4.76-fold with entire mutant NS5A: No significant effect on fitness with the substitutions; Fully sensitive to NS5A inhibitor (targeting domain I), NS3 inhibitor, or IFN-a....D320E was identified in three patients receiving alisporivir 1000 mg monotherapy for four weeks by population and clonal sequencing; However, D320E alone does not appear to be sufficient to cause viral breakthrough.....No change in sensitivity to NS3 protease inhibitor (BILN2061) or IFN-a, [or NSB5 (polymerase)].....Alisporivir is fully active against DAA resistant mutants (protease156, 168)"




|
|
| |
| |
|
|
|