| |
Multiple ascending dose study of BMS-790052, an NS5A replication complex inhibitor, in patients infected with hepatitis C virus genotype 1
|
| |
| |
Download the PDF here
Hepatology Aug 2011 accepted articles
Richard E. Nettles1, Min Gao2, Marc Bifano1, Ellen Chung1, Anna Persson3, Thomas C. Marbury4, Ronald Goldwater5, Michael P. DeMicco6, Maribel Rodriguez-Torres7, Apinya Vutikullird8, Ernesto. Fuentes9, Eric Lawitz10, Juan Carlos Lopez-Talavera2, and Dennis M. Grasela1
Affiliations:
1. Bristol-Myers Squibb, Research and Development, Hopewell, NJ; 2. Bristol-Myers Squibb, Research and Development, Wallingford, CT; 3. Bristol-Myers Squibb, Research and Development, Princeton, NJ; 4. Orlando Clinical Research Center, Orlando, FL; 5. P AREXEL, Baltimore, MD; 6. Advanced Clinical Research Institute, Anaheim, CA; 7. Fundacion de Investigacion de Diego, Santurce, Puerto Rico; 8. West Coast Clinical Trials, Cypress, CA; 9. Elite Research Institute Miami, FL; 10. Alamo Medical Research, San Antonio, TX
Abstract
The antiviral activity, resistance profile, pharmacokinetics (PK), safety and tolerability of BMS-790052, an NS5A replication complex inhibitor, were evaluated in a double-blind, placebo-controlled, sequential panel, multiple ascending dose study. Thirty patients with chronic hepatitis C virus (HCV) genotype 1 infection were randomized to receive a 14-day course of BMS-790052 (1, 10, 30, 60 or 100 mg once daily or 30 mg twice daily) or placebo in a ratio of 4:1.
Results:
The mean maximum decline from baseline in HCV RNA ranged from 2.8 to 4.1 log10 IU/mL; the placebo group showed no evidence of antiviral activity. Most patients experienced viral rebound on or before day 7 of treatment with BMS-790052 monotherapy; viral rebound was associated with viral variants that had been previously implicated in resistance development in the in vitro replicon system. The PK profile was supportive of once-daily dosing with median peak plasma concentrations at 1-2 hours postdose and mean terminal half-life of 12-15 hours. Steady state was achieved following 3-4 days of daily dosing. BMS-790052 was well tolerated in all dose groups with adverse events occurring with a similar frequency in BMS-790052- and placebo-treated groups. There were no clinically relevant changes in vital signs, laboratory, or electrocardiogram parameters.
Conclusion:
BMS-7590052 is the first NS5A replication complex inhibitor with multiple dose proof-of-concept in clinic. At doses of 1-100 mg daily, BMS-790052 was well-tolerated, had a PK profile supportive of once-daily dosing, and produced a rapid and substantial decrease in HCV-RNA levels in patients chronically infected with HCV genotype 1.
The current treatment of chronic hepatitis C virus (HCV) infection, a regimen of pegylated interferon alpha (PEG-IFN)-2a or -2b, and ribavirin (RBV) remains unsatisfactory, particularly in the large number of patients with HCV genotype 1 infection whose sustained viral response rates are currently ~40% (1). However, treatment for HCV infection is rapidly evolving with the introduction of direct-acting antiviral (DAA) agents (2, 3). The combination of telaprevir, an HCV NS3 protease inhibitor, with PEG-IFN alpha-2a and RBV has been associated with sustained viral response rates of 61%-67% in patients with genotype 1 infection (2). Telaprevir is administered three times a day and has been associated with adverse events (AEs) such as rash and anemia (4). There continues to be an unmet medical need for additional DAA agents with different mechanisms of action and resistance patterns that are easy to administer, more effective, and well tolerated. Focusing on the critical importance of non-structural protein 5A (NS5A) for HCV replication, BMS-790052 was identified as a potent and highly selective inhibitor of HCV based on inhibitor binding and mapping, inhibitor-induced resistant substitutions, and crystal structure modeling. In vitro data have shown that BMS-790052 inhibits HCV genotype 1 replicons with a median 50% effective concentration of ≤50 pM, while BMS- 790052-resistant variants remain fully sensitive to interferon alpha and small-molecule inhibitors of HCV protease and polymerase (5). NS5A is a multifunctional protein required for in vivo and in vitro HCV replication and has no known human homologs, making it an attractive target for therapeutic intervention (6).
BMS-790052 was previously found to be safe and well tolerated administered in healthy non- HCV-infected subjects at doses up to 200 mg as a single dose, and up to 60 mg once daily for 14 days. In a previous trial of patients chronically infected with HCV, administration of a single 100-mg dose of BMS-790052 was associated with a 3.3 log10 reduction in mean viral load measured 24 hours postdose. This response was sustained for an additional 120 hours in two patients infected with genotype 1b virus (6). Here we report the results of the first placebo- controlled, multiple ascending dose clinical study to evaluate the antiviral activity, resistance profile, pharmacokinetics (PK), safety, and tolerability of an HCV NS5A replication complex inhibitor, BMS-790052, in patients chronically infected with HCV genotype 1.
MATERIALS AND METHODS
Study Design
This study was a double-blind, placebo-controlled, sequential panel, multiple ascending dose study. Six dose regimens of BMS-790052 in HCV genotype 1-infected patients were evaluated (1 mg once daily, 10 mg once daily, 30 mg once or twice daily, 60 mg once daily, and 100 mg once daily) (ClinicalTrials.gov number, NCT00663208). Five patients in each panel were randomized to receive a 14-day course of orally administered BMS-790052 or placebo in a ratio of 4:1. Patients were admitted to one of eight clinical facilities in the United States between May 2008 and June 2009, and required to remain in-house from day -1 (screening day) to day 2, and from day 13 to day 15. Patients were permitted to be furloughed from the clinical facility from day 3 to day 12 and from day 16 to study discharge, which occurred at approximately day 182 for patients receiving active drug, following completion of additional blood sampling for analysis of HCV RNA and genomic substitutions. Patients treated with placebo were not required to return for follow-up visits beyond day 28. The majority of patients were treated as inpatients from day -1 to day 15. BMS-790052 or placebo was administered under fasting conditions. No intrapatient dose escalation was allowed. Dose escalation between panels occurred only after all safety data (through the final dose) from four patients receiving BMS-790052 in the prior dose panel were reviewed and deemed safe by the Bristol-Myers Squibb medical monitor in consultation with the investigators. No concomitant medications (prescription, over-the-counter or herbal) were permitted to be administered during the study, unless they were prescribed by the investigator for treatment of specific clinical events or were approved by the medical monitor prior to dosing. The study was approved by the institutional review boards in all study centers and conducted in accordance with Good Clinical Practice, as defined by the International Conference on Harmonization, in accordance with the ethical principles underlying European Union Directive 2001/20/EC and the United States Code of Federal Regulations, Title 21, Part 50 (21CFR50), and in accordance with the ethical principles that have their origin in the Declaration of Helsinki. Informed written consent was obtained from all patients.
Patients were randomly assigned to receive BMS-790052 or placebo according to a computer- generated randomization scheme prepared by Bristol-Myers Squibb. An Interactive Voice Response System was used to assign a unique subject number and a blinded container number, which was provided to the blinded study staff who supervised and recorded the drug administration.
The sample size was based on the primary endpoint of the study, defined as the change in log10 HCV RNA from baseline to day 7. A mean decrease of at least 1.5 log10 HCV RNA within one dose panel would suggest that BMS-790052 was sufficiently active to proceed to late phase development. If BMS-790052 had no effect, administration of drug to four patients within a dose panel would yield a probability of 0.01 to observe a mean decrease in log10 HCV RNA of more than 1.5. If the true mean decrease was 2.0, the probability of observing a mean decrease in log10 HCV RNA of more than 1.5 would be 0.78. These calculations are based on the assumption that log10 HCV RNA is normally distributed, with a standard deviation for the change of 1.3.
Patients
Eligible patients for this study were men and women, ages 18-60 years inclusive, with a body mass index of 18-35 kg/m2, who were chronically infected (longer than 6 months) with HCV genotype 1, and who were treatment-naive to interferon and RBV. Additional inclusion criteria were: plasma HCV RNA ≥100,000 IU/mL; documented FibroTest score of ≤0.72 and APRI ≤2, or the absence of cirrhosis based on liver biopsy within 12 months; women of childbearing potential were not to be nursing or pregnant and had to be willing to agree to use double barrier contraception for at least 1 month before dosing, during dosing, and at least 12 weeks after the last dose of study medication.
The main exclusion criteria were: patients with prior documented cirrhosis on liver biopsy; previous exposure to a NS5A replication co-factor inhibitor; coinfection with human immunodeficiency virus; coinfection with hepatitis B virus.
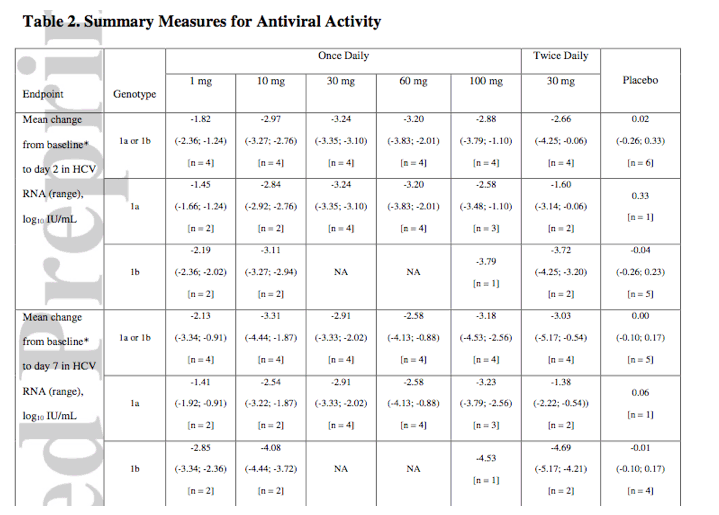
DISCUSSION
The results of this study indicate that BMS-790052 is a potent NS5A replication complex inhibitor that produces a substantial decline in HCV RNA in patients chronically infected with either HCV genotype 1a or genotype 1b. BMS-790052 was shown to be generally well tolerated, and had a PK profile supportive of once-daily dosing.
The potent antiviral effect of BMS-790052 observed in a previous study was confirmed in the present study (6). In this study, HCV-RNA levels decreased by ~3 logs after a single dose in all BMS-790052-treated groups, other than the 1 mg group. This is consistent with single ascending dose results (6), and demonstrates that the in vitro picomolar potency of BMS-790052 translates in vivo to substantial antiviral activity. In addition, although the sample size was small, it appears that patients infected with HCV genotype 1b virus responded better than patients infected with HCV genotype 1a virus, with a more marked and sustained viral RNA decline. This is consistent with both the difference in the intrinsic potency of BMS-790052 for genotype1a and 1b replicons (50 pM versus 9 pM), and the higher level of resistance observed in vitro for genotype 1a variants (6). The early suppression of HCV replication with BMS-790052 monotherapy is comparable with, and in some cases exceeds, that observed for other DAA agents (7, 8).
Using the standard model of HCV infection and treatment (9), treatment with BMS-790052 in a prior monotherapy study (6) was associated with improved estimation of HCV RNA clearance rate, shorter delay in viral clearance, and shorter HCV RNA T1/2 as compared with PEG-IFN + RBV therapy and telaprevir therapy (10). In the current study, exposure to BMS-790052 again resulted in a rapid decline in HCV RNA in most patients. This early suppression of HCV replication with BMS-790052 monotherapy was commonly followed by viral rebound, as typically observed for short courses of DAA agents when administered as monotherapy (7, 11). In the current study, viral rebound generally occurred on or before day 7 of dosing and was associated with the emergence of previously described viral variants linked with high levels of viral resistance in the replicon system (5). A more detailed description of observed viral variants will be presented elsewhere. Importantly, preliminary data suggest that the combination of BMS- 790052 with PEG-IFN + RBV therapy or other DAA agents will be effective at markedly reducing viral rebound (12, 13).
While the development of DAA agents to treat HCV has focused in part on inhibitors of the viral enzymes NS3 protease and NS5B RNA-dependent RNA polymerase (2), BMS-790052 was developed as a small molecule inhibitor targeting the HCV NS5A protein (6). The precise role of NS5A in HCV replication has not been defined; however, observations of inhibition of viral replication in both in vitro replicon systems and single and multiple dose clinical trials confirm the essential role of NS5A in HCV replication. NS5A is a multifunctional viral protein that functions not only as an essential component of the HCV replication complex, but also as a modulator of cellular signaling pathways (14). The observed antiviral effects provide a rationale for the use of BMS-790052 in interferon-based combination therapy. A working model that may explain the potency of BMS-790052 is that its antiviral effect is amplified by the NS5A interactions with viral and cellular proteins. We have observed that BMS-790052 inhibits multiple stages of viral replication, such as the formation of replication complexes and active RNA replication (manuscript submitted). Furthermore, BMS-790052 exhibits additive or synergistic effects in replicon system studies with NS5B, NS3, and non-nucleoside NS5B inhibitors (6).
The PK profile of BMS-790052 supports once-daily dosing, with plasma concentrations throughout the 14-day dosing period above the protein binding-adjusted EC90 concentrations required for effective inhibition of HCV replication in the replicon systems. The exposure response observed in the current study suggests that the ranges evaluated in this study support a proposed therapeutic dose of 3-60 mg.
BMS-790052 was generally well tolerated over the study period for all doses evaluated. Adverse events occurred with a similar frequency in BMS-790052- and placebo-treated groups. All AEs were considered by the investigators to be unrelated to the medication.
In conclusion, the results of this study suggest that the novel NS5A replication complex inhibitor BMS-790052 can be administered orally once daily at doses of 10-100 mg daily and is well tolerated. BMS-790052 produces a substantial decline in HCV RNA levels following multiple doses in patients chronically infected with either HCV genotype 1a or genotype 1b. These results confirm the importance of inhibiting NS5A-mediated HCV replication and the potential of BMS- 790052 as part of combination therapy in the treatment of HCV. Additional clinical trials are ongoing to further confirm the safety and efficacy of BMS-790052 in patients with chronic HCV infection.
|
|
| |
| |
|
|
|