| |
Telaprevir Alone or With Peginterferon and Ribavirin Reduces HCV RNA in Patients With Chronic Genotype 2 but Not Genotype 3 Infections
|
| |
| |
Download the PDF here
Gastroenterology September 2011
Received 8 December 2010; accepted 16 May 2011. published online 01 June
2011.
"For treatment-naïve patients with genotype 2 HCV, studies to examine a shortened duration of therapy that includes telaprevir should be considered."
----------------------------------
Telaprevir Effective for Genotype 2 But Not For Genotype 3
www.natap.org/2009/EASL/EASL_13.htm -
In this study genotype 2 treatment naive patients achieved a mean 4 log reduction in viral load after 6 days on monotherapy with telaprevir
--------------------------------------
Activity of Telaprevir Alone or in Combination with Peginterferon ...
www.natap.org/2010/EASL/EASL_15.htm -
Activity of Telaprevir Alone or in Combination with Peginterferon Alfa-2a and Ribavirin in Treatment-naïve, Genotype 2 and 3, Hepatitis C Patients: Final
ABSTRACT
Background & Aims
We evaluated antiviral activity of 2 weeks therapy with telaprevir alone, peginterferon alfa-2a and ribavirin (PR), or all 3 drugs (TPR) in treatment-naïve patients with chronic hepatitis C virus (HCV) genotype 2 or 3 infections.
Methods
We performed a randomized, multicenter, partially blinded study of patients (23 with HCV genotype 2, 26 with genotype 3) who received telaprevir (750 mg every 8 h), placebo plus PR (peginterferon, 180 μg, once weekly and ribavirin, 400 mg, twice daily), or TPR for 15 days, followed by PR for 22 or 24 weeks. Plasma levels of HCV RNA were quantified.
Results
Levels of HCV RNA decreased in all patients with HCV genotype 2, including those who received telaprevir monotherapy. The decrease was more rapid among patients who received telaprevir. By day 15, 0% (telaprevir), 40% (TPR), and 22% (PR) of patients with HCV genotype 2 had undetectable levels of HCV RNA; rates of sustained virologic response were 56%, 100%, and 89%, respectively. Overall, 6 of 9 HCV genotype 2 patients that received only telaprevir had viral breakthrough within 15 days after an initial response. HCV RNA levels decreased slightly among patients with HCV genotype 3 who received telaprevir and decreased rapidly among patients given PR or TPR (telaprevir had no synergistic effects with PR). Sustained virologic response rates were 50%, 67%, and 44% among patients given telaprevir, TPR, or PR respectively; 7 patients with HCV genotype 3 relapsed after therapy (2 given telaprevir, 3 given TPR, and 2 given PR) and 3 patients with HCV genotype 3 had viral breakthrough during telaprevir monotherapy. The incidence of adverse events was similar among groups.
Conclusions
Telaprevir monotherapy for 2 weeks reduces levels of HCV RNA in patients with chronic HCV genotype 2 infections, but has limited activity in patients with HCV genotype 3.
Current therapy for patients with chronic hepatitis C virus (HCV) who are infected with genotype 2 or 3 comprises peginterferon (Peg-IFN) and ribavirin (RBV) administered for 24 weeks.1 The aim of therapy is to achieve a sustained virologic response (SVR; defined as undetectable HCV RNA 6 months after treatment cessation), which is believed to be equivalent to a cure since long-term follow-up studies show very limited disease recurrence.2 In general, patients with genotype 2/3 treated with Peg-IFN/RBV achieve SVR rates approaching 80%, although some patient subgroups respond less well.1, 3
Agents that directly target viral replication have the potential to improve SVR rates and shorten treatment duration. Telaprevir is an orally available, highly potent, specific inhibitor of the HCV nonstructural (NS) 3 protease that increased SVR rates and reduced treatment duration in phase III clinical trials of patients with genotype 1 HCV when added to Peg-IFN/RBV therapy vs Peg-IFN/RBV alone.4, 5, 6, 7 Telaprevir was generally well-tolerated in genotype 1 clinical trials, but with a slightly higher rate of some adverse events (AEs), including rash, than with Peg-IFN/RBV.4, 5, 6, 7 In vitro data suggest that telaprevir is also active against genotype 2 and 3 HCV, and modeling data imply that the drug has activity against nongenotype 1 protease enzymes.8 However, to date, phase II studies have not assessed protease inhibitors in patients with genotype 2 or 3 chronic HCV. It is therefore unclear whether they will play a role in increasing response rates and reducing the duration of therapy in these patients.
Accordingly, we conducted a multicenter, randomized, controlled, phase II clinical trial to evaluate the early kinetics of genotype 2 and 3 HCV in treatment-naïve patients treated with telaprevir-based therapy vs Peg-IFN/RBV alone. To allow a clear evaluation of telaprevir's intrinsic antiviral activity, a small number of patients also received the drug as monotherapy during the early phase of the study.
Results
Patient Disposition and Baseline Characteristics
Patient disposition is shown in Figure 1. Twenty-six patients with HCV genotype 2 were randomized, and 23 were treated (2 patients in the telaprevir plus Peg-IFN/RBV group withdrew consent before first intake and another patient in the telaprevir plus Peg-IFN/RBV group was infected with HCV genotype 4). Twenty-six patients with genotype 3 HCV were randomized and all were treated. All treated patients completed the 2-week investigational treatment phase. Nineteen patients (83%) with HCV genotype 2 and 21 patients (81%) with HCV genotype 3 completed the entire study (including follow-up). Baseline demographics and disease characteristics were generally well balanced across genotype and treatment groups (Table 1) and differences between treatment groups at baseline were probably due to the small sample size, and were thought unlikely to influence the outcome.
Efficacy
Efficacy in HCV Genotype 2-Infected Patients
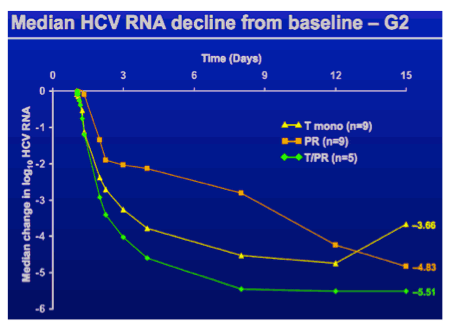
At baseline, median log10 HCV RNA values were similar across groups; 6.61 (telaprevir montherapy), 6.21 (telaprevir plus Peg-IFN/RBV), and 6.15 (Peg-IFN/RBV) log10 IU/mL. During the investigational treatment phase, HCV RNA values decreased in all groups (Figure 2A); in particular, there was a marked, rapid decrease in HCV RNA values in patients receiving telaprevir monotherapy (Figure 2A and B). At day 3, the median HCV RNA decreases were -3.27 (range, -4.3 to -2.8), -4.03 (range, -4.4 to -3.2), and -2.04 (range, -3.9 to 0.1) log10 IU/mL in the telaprevir monotherapy, telaprevir plus Peg-IFN/RBV, and PegIFN/RBV groups, respectively. The difference in mean HCV RNA levels between baseline and day 3 in the telaprevir montherapy group was statistically significant (P < .001). The rapid decreases in both telaprevir regimens continued, resulting in large differences between changes in HCV RNA levels vs Peg-IFN/RBV up to days 8 to 12 (Figure 2A). Between days 12 and 15, median HCV RNA levels increased in the telaprevir monotherapy group. On day 15, the median HCV RNA decreases were -3.66 (range, -5.4 to -0.9), -5.51 (range, -6.0 to -4.6), and -4.83 (range, -6.0 to -0.2) log10 IU/mL in the telaprevir monotherapy, telaprevir plus Peg-IFN/RBV, and Peg-IFN/RBV groups, respectively (Figure 2A).
The proportion of patients with undetectable HCV RNA on day 8 were 0% (telaprevir monotherapy), 20% (1 of 5; telaprevir plus Peg-IFN/RBV), and 0% (Peg-IFN/RBV). The proportions on day 15 were 0%, 40% (2 of 5), and 22% (2 of 9), respectively, increasing to 89-100% at EOT (Table 2).
Table 2. Summary of Antiviral Activity
Following initiation of standard therapy after 2 weeks, HCV RNA values decreased in the telaprevir monotherapy group and continued to decrease in the Peg-IFN/RBV group to week 4, while low HCV RNA levels were maintained in the telaprevir plus Peg-IFN/RBV group. At week 24/26, median HCV RNA decreases were -5.91 (range, -6.6 to -3.7), -5.51 (range, -6.6 to -4.6), and -5.45 (range, -6.7 to 0.7) log10 IU/mL in the telaprevir monotherapy, telaprevir plus Peg-IFN/RBV, and Peg-IFN/RBV groups, respectively.
The median times to first undetectable HCV RNA were 31, 12, and 43 days in the telaprevir monotherapy, telaprevir plus Peg-IFN/RBV, and Peg-IFN/RBV groups, respectively, and the proportions of patients with an SVR were 56% (5 of 9), 100% (5 of 5), and 89% (8 of 9) in these groups (Table 2). One patient in the telaprevir monotherapy group had undetectable HCV RNA at follow-up week 12, but was lost to follow-up by week 24, and consequently was a treatment failure. One patient in the telaprevir monotherapy group relapsed; time to relapse was 85 days (after EOT) in this patient.
Efficacy in Genotype 3-Infected Patients
Baseline median log10 HCV RNA values were similar across groups: 6.65 (telaprevir monotherapy), 6.79 (telaprevir plus Peg-IFN/RBV), and 6.92 (Peg-IFN/RBV) log10 IU/mL. Similar HCV RNA decreases from baseline were seen over time in response to telaprevir plus Peg-IFN/RBV and Peg-IFN/RBV (Figure 2C). In contrast to the observed findings in genotype 2 HCV, telaprevir elicited only slight decreases in HCV RNA levels when used as monotherapy (Figure 2D). At day 3, median HCV RNA decreases were -0.54 (range, -2.0 to 0.1), -3.05 (range, -4.1 to -1.5), and -2.38 (range, -3.7 to -0.4) log10 IU/mL in the telaprevir monotherapy, telaprevir plus Peg-IFN/RBV, and Peg-IFN/RBV groups, respectively. Although the absolute change was smaller than in genotype 2 HCV, the difference in mean HCV RNA levels between baseline and day 3 in the telaprevir monotherapy group was statistically significant (P = .036). On day 15, median changes in HCV RNA were -0.54 (range, -1.0 to -0.1), -4.85 (range, -6.1 to -2.3), and -4.72 (range, -6.1 to -3.2) log10 IU/mL in the telaprevir monotherapy, telaprevir plus Peg-IFN/RBV, and Peg-IFN/RBV groups, respectively (Figure 2C).
HCV RNA levels markedly decreased upon commencement of standard therapy (Peg-IFN/RBV) in the telaprevir monotherapy group, and continued to decrease slightly with time in the other groups. At week 24/26, the median changes from baseline were -5.71 (range, -6.4 to -0.9), -5.50 (range, -6.7 to -4.7), and -6.22 (range, -6.6 to -3.2) log10 IU/mL in the telaprevir monotherapy, telaprevir plus Peg-IFN/RBV, and Peg-IFN/RBV groups, respectively.
No genotype 3-infected patients had undetectable HCV RNA on day 8. At day 15, 0%, 22% (2 of 9) and 11% (1 of 9) of patients, respectively, had undetectable HCV RNA levels, and these proportions were higher at EOT (Table 2). Median times to first undetectable HCV RNA were 99, 43, and 29 days, respectively. The proportions of patients with an SVR were 50% (4 of 8), 67% (6 of 9), and 44% (4 of 9), respectively (Table 2). Two patients in each of the telaprevir monotherapy and Peg-IFN/RBV groups, and 3 patients in the telaprevir plus Peg-IFN/RBV group, relapsed. In addition, 1 patient (Peg-IFN/RBV group) had undetectable HCV RNA at EOT but was a treatment failure because no follow-up data were available.
Virology Findings
Sequences were available for 22 of 23 (HCV genotype 2) and 24 of 26 (HCV genotype 3) baseline samples. None of these had any of the following NS3 mutations at baseline: V36A/M, T54A, R155K/T/I/M, or A156V/T/S, which have previously been observed in telaprevir-treated patients infected with HCV genotype 1 with on-treatment virologic failure or relapse.
Virology Findings in HCV Genotype 2-Infected Patients
In the telaprevir monotherapy group, confirmed viral breakthrough occurred in 2 patients during the investigational treatment phase (ie, while receiving telaprevir monotherapy). An additional 4 patients had unconfirmed viral breakthrough at the end of this period and were analyzed for the emergence of viral variants. No patients in the other groups had viral breakthrough during the investigational treatment phase. One patient (Peg-IFN/RBV group) had viral breakthrough during the standard treatment phase.
Viral sequences were available for 5 of the 6 patients in the telaprevir monotherapy group at breakthrough. This analysis focused on identifying mutations observed to emerge in HCV genotype 1 patients (V36A/M, T54A, R155K/T/I/M, or A156V/T/S). All 5 patients had emerging mutations at the time of viral breakthrough. Mutations were detected in all of the analyzed samples; specific mutations are shown in Table 3.

Of the 6 patients with viral breakthrough (confirmed and unconfirmed) in the telaprevir monotherapy group, 5 had undetectable HCV RNA at the end of the standard therapy phase. Three of these patients achieved an SVR (all with an unconfirmed viral breakthrough). The only patient with HCV genotype 2 who relapsed (telaprevir monotherapy group without prior viral breakthrough, noncompliant during standard therapy phase) did not have any relevant emerging mutations at follow-up week 24.
Virology Findings in HCV Genotype 3-Infected Patients
Viral breakthrough was not seen in the telaprevir monotherapy, telaprevir plus Peg-IFN/RBV, or Peg-IFN/RBV groups. As telaprevir monotherapy only resulted in a slight decrease in HCV RNA in HCV genotype 3-infected patients, the majority of these patients did not meet the criteria for viral breakthrough (ie, they did not experience a >1 log10 reduction in HCV RNA values from nadir during the investigational treatment phase). Three patients in the telaprevir monotherapy group were found to have viral breakthrough in the investigational treatment phase; 1 confirmed and 2 unconfirmed. The mutation R155K was found at time of breakthrough in 2 of these patients, and at a later time point in the third patient (Table 3). One of these 3 patients achieved an SVR (with a confirmed viral breakthrough) and 1 had undetectable HCV RNA at EOT but relapsed during follow-up (with unconfirmed virologic breakthrough).
Among patients who did not meet the criteria for viral breakthrough but had detectable HCV RNA at the end of telaprevir treatment, viral sequences were available for 4 of 5 patients in the telaprevir monotherapy group and 1 patient in the telaprevir plus Peg-IFN/RBV group. No known mutations associated with decreased susceptibility to telaprevir were detected. Of the 5 patients in the telaprevir monotherapy group, 4 had undetectable HCV RNA at EOT, 3 went on to achieve an SVR following standard treatment, and 1 relapsed. Two patients in the telaprevir monotherapy group and 3 in the telaprevir plus Peg-IFN/RBV group relapsed at the end of the 26-week treatment period. Viral sequences were available for 4 of these patients. In the telaprevir monotherapy group, 1 sample had no detectable mutations and the other had the R155K mutation. Neither of the 2 patients analyzed from the telaprevir plus Peg-IFN/RBV group at relapse had detectable mutations.
Safety and Tolerability
There were no apparent differences in the incidence, severity, or type of AEs, or laboratory and cardiac safety parameters in HCV genotype 2-infected patients vs those with HCV genotype 3 infection. Thus, the safety data were combined.
AEs
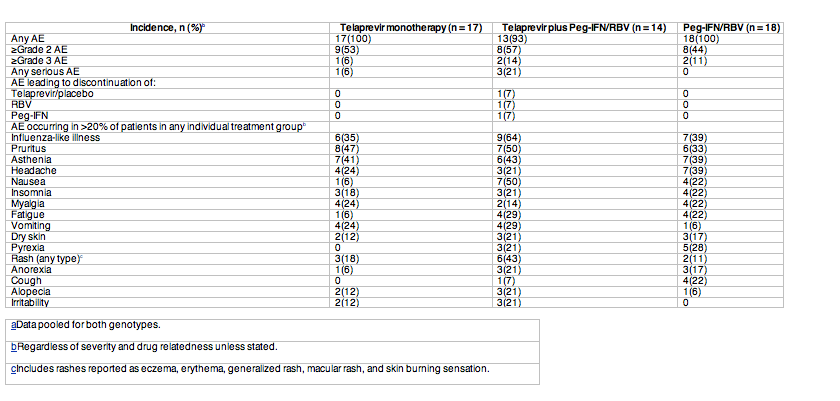
AEs occurring during the overall treatment period are listed in Table 4. The most frequently reported AEs (>20% of patients in any group; Table 4) were influenza-like illness, pruritus, asthenia, headache, nausea, insomnia, myalgia, fatigue, vomiting, dry skin, pyrexia, rash, anorexia, cough, alopecia, and irritability. All AEs were grade 1 or 2 in severity, except for 1 telaprevir-treated patient who had a grade 3 rash (telaprevir plus Peg-IFN/RBV group; investigational phase), grade 3 neutropenia (1 patient in the telaprevir monotherapy group, 2 patients in the Peg-IFN/RBV group; standard treatment phase), and pharyngolaryngeal pain and pneumonia in 1 patient (telaprevir plus Peg-IFN/RBV group; standard treatment phase). Five serious AEs were recorded in 4 patients during the overall treatment phase, only 1 of which (pneumonia) was considered to be possibly related to study medication (Peg-IFN/RBV). Any rash (including rashes reported as eczema, erythema, generalized rash, macular rash, and skin burning sensation) occurred in 3 (18%), 6 (43%), and 2 (11%) patients in the telaprevir monotherapy, telaprevir plus Peg-IFN/RBV, and Peg-IFN/RBV groups, respectively. All rashes were grade 1 or 2 in severity, except for the grade 3 rash in 1 patient on day 3 that led to permanent discontinuation of telaprevir; Peg-IFN/RBV was temporarily interrupted but later restarted. Another patient permanently discontinued Peg-IFN/RBV at week 17 due to grade 2 rash. Four patients experienced anemia and all events were grade 2 or lower. No life-threatening AEs or deaths occurred during the trial.
Laboratory Parameters and Cardiovascular Safety
The most frequently observed (at least 3 patients in any group) treatment-emergent toxicities were decreased neutrophils, white blood cells, lymphocytes, hemoglobin, and aspartate aminotransferase and alanine aminotransferase levels, and increased uric acid. No clinically relevant changes over time were observed for electrocardiogram parameters and the incidence of electrocardiogram abnormalities was low.
Pharmacokinetic Analyses
In the monotherapy groups, telaprevir exposure was comparable between genotype 2- and genotype 3-infected patients (Supplementary Table 1). Telaprevir exposure was higher in the telaprevir plus Peg-IFN/RBV group vs the telaprevir monotherapy group; this difference was more pronounced in genotype 2-infected patients. There was no clear association between telaprevir exposure and response (including viral breakthrough) or safety parameters (occurrence of rash-related AEs or hemoglobin toxicities) (data not shown).
Discussion
Telaprevir monotherapy studies provided evidence for the activity of telaprevir against HCV genotype 1.9 In studies of patients with genotype 1 chronic HCV infection, telaprevir combined with Peg-IFN/RBV significantly increased the SVR rate and reduced the duration of therapy in a large proportion of patients.5, 6, 7, 10, 11, 12 Patients with genotype 2 and 3 HCV usually respond well to 24 weeks of therapy with Peg-IFN/RBV.1 However, 20-30% of patients do not achieve an SVR with this treatment regimen.1, 13 These difficult-to-treat patients include those with advanced fibrosis, higher baseline HCV RNA levels, a higher body weight, and a TT interleukin (IL)-28B genotype. In addition, genotype 2/3 patients who do not achieve undetectable HCV RNA at week 4 are also less likely to achieve an SVR.14, 15, 16 These patients are in need of additional therapies. The potential for telaprevir to shorten treatment regimens (in most cases) and increase SVR rates in patients with genotype 1 HCV5, 6, 7, 10, 11, 12 suggests that telaprevir may also be able to improve treatment outcomes in other genotypes. The purpose of this study was to evaluate, for the first time, the antiviral activity of telaprevir in patients infected with genotype 2 or 3 HCV.
In patients with genotype 2 HCV, telaprevir inhibited viral replication and synergized with Peg-IFN/RBV. Hence, there was a highly significant reduction in HCV RNA from baseline to day 3 in the telaprevir monotherapy group. However, there was limited activity of telaprevir in patients with genotype 3 HCV. The differences in response were not due to differences in telaprevir exposure, as patients infected with either genotype had sufficient telaprevir plasma concentrations. These observations, which could not be predicted from in vitro models or structural analysis of the viral protease,8 highlight the need for clinical studies involving short monotherapy arms to confirm the activity of direct-acting antiviral agents.
The use of telaprevir monotherapy for a period of 2 weeks allowed us to determine the intrinsic activity of the drug against HCV genotype 2 and 3, and to determine potential synergy between Peg-IFN/RBV and telaprevir. Although combining telaprevir with Peg-IFN/RBV showed antiviral activity in genotype 2-infected patients, the use of telaprevir monotherapy led to a rapid viral breakthrough, as described in genotype 1.4 This finding confirms that telaprevir monotherapy is not a feasible treatment option for HCV. We identified mutations previously associated with reduced susceptibility to telaprevir in genotype 1 in all genotype 2 and 3 viral breakthrough patients who could be tested and in some patients with relapse (further details to be published elsewhere). However, approximately half of the viral breakthrough patients went on to achieve an SVR following standard treatment, 2 were discontinued or lost to follow-up and only 1 relapsed (no relevant mutations detected). Therefore, as seen in genotype 1,4 Peg-IFN/RBV treatment can eradicate telaprevir-resistant variants that emerge during viral breakthrough. This finding underlines the importance of administering telaprevir and other direct-acting antiviral agents in combination with Peg-IFN/RBV to control low-level emergent variants and to ultimately maximize the chance of achieving an SVR.
Although the number of patients evaluated was small, the incidence and severity of AEs in this trial were comparable with other studies of patients infected with HCV genotype 1;5, 6, 7, 10, 11, 12 no new side effects were observed. Most AEs were grade 1 or 2, although 1 telaprevir-treated patient had grade 3 rash during the investigational phase and permanently discontinued telaprevir (Peg-IFN/RBV were temporarily interrupted). One patient permanently discontinued Peg-IFN/RBV due to grade 2 rash during Peg-IFN/RBV therapy, and no life-threatening AEs or deaths occurred during the study.
The study was limited primarily by its small sample size and exploratory nature. Indeed, these factors may account for the observed lack of association between telaprevir exposure and response. In addition, since the IL-28B polymorphism is a predictor of Peg-IFB/RBV response in patients with genotypes 2 and 3 HCV,14 examination of the impact of IL-28B on the observations in the study may have provided further insight. However, IL-28B was not discovered at the time of study enrollment and data were therefore not collected.
Conclusions
This study was the first to evaluate the activity of telaprevir in patients with genotype 2 or 3 HCV. The results showed that telaprevir had antiviral activity in treatment-naïve patients with genotype 2 HCV, but limited activity against genotype 3, indicating that this drug is unlikely to have major clinical utility in this patient subpopulation. However, the potential of telaprevir-based triple combination therapy in patients with HCV genotype 2 who have not responded to Peg-IFN/RBV should be explored. For treatment-naïve patients with genotype 2 HCV, studies to examine a shortened duration of therapy that includes telaprevir should be considered.
|
|
| |
| |
|
|
|