| |
New DAA FDA Guidance March 2012
|
| |
| |
Reported by Jules Levin
presented by Russell Fleischer, PA-C, MPH, Senior Clinical Analyst, Division of Antiviral Products, US Food and Drug Administration, at the AASLD special topic meeting March 16-17 2012 in Atlanta
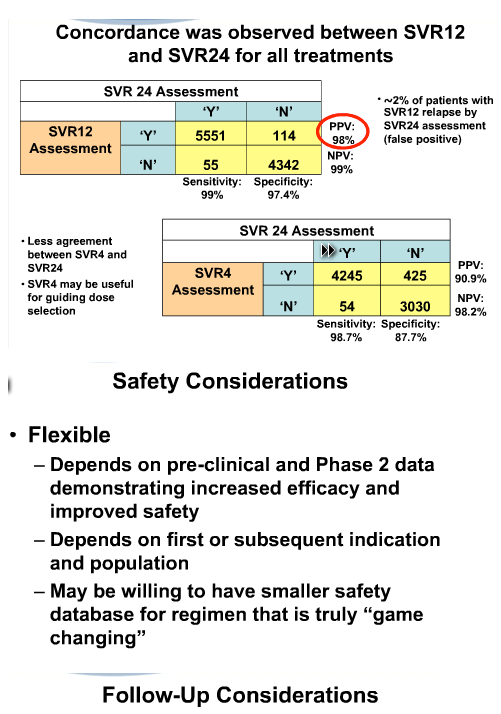
You have attended this conference or know already about the talk. I have attached the slide presentation but you can view the webcast & hear the talk on the AASLD website in the Liver Learning Portal.
from Jules: The language used in this presentation makes it clear in general guidance is fluid but the FDA also clearly steps into new territory with this new guidance saying they will consider historical controls for IFN-free regimens in phase 3, but didn't go beyond that. Inclusion of specifically mentioned special populations are encouraged, see slides. The biopsy discussion is interesting.
I have highlighted immediately below key points & slides presented, followed by the full slide presentation.
"I want to briefly update you on some of the activities we are engaged in around updating our HCV DAA guidance and what these changes may mean for the future development of DAA therapies. At the end I will discuss fast track as Ray asked me too, and I think there are some misconceptions about what this designation actually means."
Phase 3 trials should enroll adequate numbers of patients representative of the disease demographics: "we want to see" African-Americans, Latinos, compensated Cirrhotics, and "we want to open the door to 2 populations typically excluded from clinical trials": IVDU/opiate substitution.
For IFN-contraindicated we understand you cant randomize them to interferon "so if we have an IFN-free regimen thats going forward we are considering single arm historical control trials, if the phase 2 data is supportable of that approach, its probably only going to fly for really short duration regimens. This will work until 1 or 2 regimens are approved and then they will work as de facto standards of care, then we'll probably go with non-inferiority vs standard of care; another option would be an immediate vs deferred PBO controlled standard of care"

"it's on a case by case basis"
"For experienced patients regimens are needed that maximize the response rate because there are likely to be very limited options for retreatment.
For IFN-free regimens in nulls and prior partial responders we believe that we can use historical data from the PI studies as it would not really be reasonable to randomize patients to retreatment with PR alone or P/R/PI. Similar to naives, this will likely be the way forward until a regimen is approved at which time that regimen will become the control.
For IFN-containing regimens, nulls again we would allow historical controls. In partial responders, the need for an active control will be driven by the Phase 2 data."

"until nonivasive tests are FDA approved in the USA sponsors need to provide rationale & validation"

"We continue to want and have been prodding sponsors to get data on these populations at time of NDA submission. To start evaluating a product in these subgroups, some safety and efficacy data in the general population of HCV subjects is needed, hepatic impairment studies and relevant drug-drug interaction studies should be completed in order to understand how the drugs should be administered. This data should be amassed by EOP2 so that some groups could be enrolled into Phase 3 trials."
"peg-RBV control arm not required"

"The 300 came from what we have historically asked for in HIV monotherapy to expand an indication. We believe this number should be acceptable for co-infected, but could be adjusted based on the first few applications."

"We need data from a hepatic Impairment Study to understand if any dose adjustments are necessary and again, this study should be conducted early in development. SVR is an important endpoint not withstanding disease progression requiring transplantation, because SVR will likely translate into prevention of infection of a newly transplanted liver"

"Kids are important too. Sponsors are encouraged to begin discussions of their pediatric formulation and clinical development plan early in development once PK, PD, antiviral activity and safety data are available."




Codifies what DAVP has always done. Sponsors like to issue press releases suggesting that FDA holds their product to be something super special, but in fact the overwhelming majority of products targeting HCV have been granted fast track status.
the company gets to issue press release suggesting their drug is "super special" the fact of the matter is the designation can be withdrawn.

FULL SLIDE PRESENTATION











|
|
| |
| |
|
|
|