| |
(Issentress) Raltegravir for Pediatrics: Interim Results from IMPAACT P1066: Raltegravir (RAL) Oral Chewable Tablet (CT) Formulation in Children 2-5 Years - CROI 2011 Presentation & attached FDA Label containing pediatric safety, PK, dosing, efficacy
|
| |
| |
Dowload the PDF here
CROI Feb 2011
Sharon Nachman1, Edward Acosta2, Nan Zheng3, Hedy Teppler4, Brenda Homony4, Terence Fenton3, Edward Handelsman5, Carol Worrell6, Bobbie Graham7, Andrew Wiznia8, and the P1066 Team
1SUNY Stony Brook, NY, 2U Alabama, AL 3Harvard School Public Health, MA, 4 Merck Sharp & Dohme Corp., NJ, 5 NIAID, MD, 6NICHD, MD, 7 Frontier Science Research Foundation Inc., NY, 8 Jacobi Med Center, NY

ABSTRACT
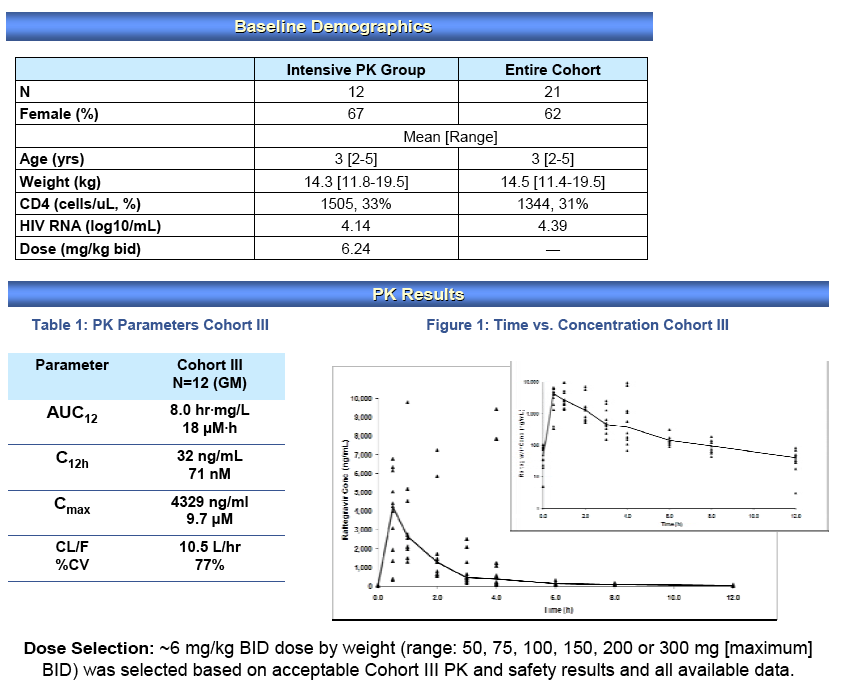
Background: RAL is an HIV-1 integrase inhibitor approved for use in adults1. P1066 is an open label study of RAL in treatment experienced HIV+ youth; acceptable pharmacokinetics (PK), safety and short term efficacy have been described in youth 6-18 yrs receiving the adult formulation (AF), and youth 6-12 yrs receiving a chewable tablet (CT). Here we report initial PK, efficacy, and all safety data as of Oct 1, 2010 from Cohort III, 2-5 yr-old subjects receiving the RAL CT.
Methods: HIV+ 2-5 yr-old children enrolled: dose-finding with intensive PK, safety and virologic response. Entry criteria included HIV RNA>1000 c/mL, prior ARV experience but naïve to integrase inhibitors. RAL CT 6 mg/kg BID was added to the existing ARV regimen, intensive PK samples drawn between d 5 - 12, then ARVs were optimized. Summary PK parameters were evaluated and a dose was selected for continued study using an area-under-the-curve (AUC12h) targeted design (target range of 14-25 μM*h) based on available PK data with a C12h target to exceed the protein-adjusted IC95 of raltegravir against WT
virus (31 nM). PK parameters were compared to existing data from children 6-18 yrs receiving the AF and children 6-11 yrs receiving RAL CT. Virologic success was defined as vRNA < 400 c/mL or 1 log drop from baseline.
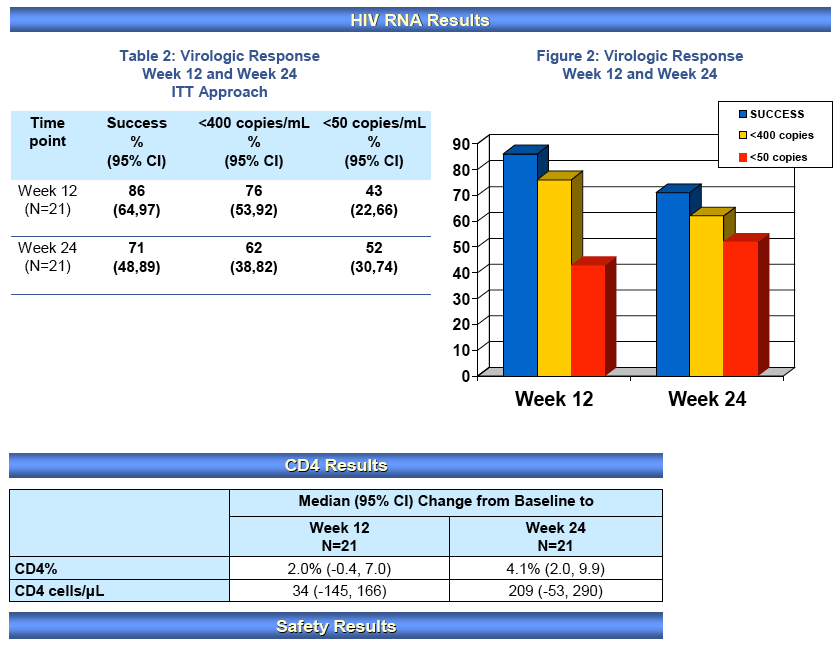
Results: N=12, Demographics: 67% female, 75% Black, mean (SD): age, 3 years (1); log10 RNA, 4.14 copies/mL (0.89); CD4%, 33% (17%); CD4 count, 1505 cells (1568); weight, 14.3 kg (2.4), dose: 6.24 mg/kg (0.67); AUC12h, 21 μM*h (13); Cmax, 11.7 μM (6.6); and C12h, 89 nM (49), compared to the 6-11 yr olds dosed at 6 mg/kg mean AUC12h, Cmax, C12h and weight; 24 μM*h, 12 μM, 171 nM and 24 kg, respectively. There were 5 patients with 7 grade ≥3 adverse events (5 low ANC); none were related to study drug. There were no treatment discontinuations. Virologic success was noted in 75% (95% CI: 43%, 95%) of 12
subjects at wk 12 and 60% (26%, 88%) of 10 subjects who reached wk 24. There was a net gain (95% CI) in %CD4 cells of 3 (1, 9) points at wk 12 and 7 (1, 18) at wk 24.
Conclusions: Using weight based dosing of RAL CT @6mg/kg BID, in children 2-5 yrs, PK data were similar to that observed in children 6-11 yrs. A RAL CT dose of 6 mg/kg (max 300 mg) BID was chosen for continued study in this age group. In these subjects, RAL CT appeared to be generally safe and well tolerated. Partial efficacy data through wk 24 were favorable.


| |
| |
| |
|
|
|