| |
Silymarin for HCV infection - Review
|
| |
| |
Download the PDF here
Antiviral Therapy April 2013
Stephen J Polyak1,2,3,*, Nicholas H Oberlies4, Eve-Isabelle Pecheur5, Harel Dahari6,7,8, Peter Ferenci9, Jean-Michel Pawlotsky10,11
Silibinin Is a Potent Antiviral Agent in Patients With Chronic HCV not Responding to Pegylated Interferon/Ribavirin Therapy
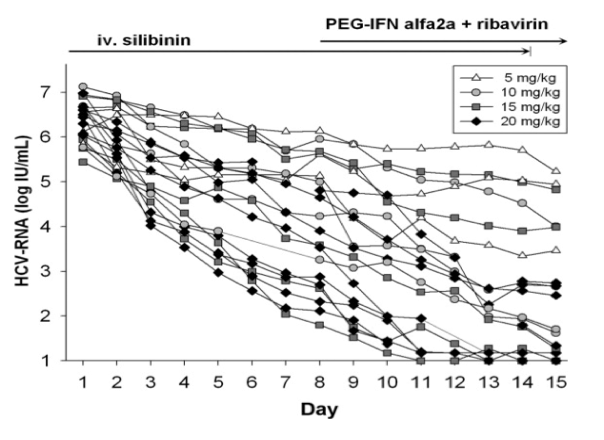
http://www.natap.org/2008/HCV/090808_01.htm.........After 7 days of silibinin monotherapy the 5-mg/kg dose was marginally effective (n = 3; log drop, 0.55 ± 0.5), whereas the 10 mg/kg (n = 19 [including the patients in protocol 1], log drop 1.41 ± 0.59), 15 mg/kg (n = 5; log drop, 2.11 ± 1.15), and 20 mg/day doses (n = 9; log drop, 3.02 ± 1.01) led to a highly significant decrease in viral load (P < .001
Silibinin monotherapy prevents graft infection after orthotopic liver transplantation in a patient with chronic hepatitis C
http://www.natap.org/2011/HCV/021611_06.htm
Silymarin for HCV infection
Antiviral Therapy April 2013
Stephen J Polyak1,2,3,*, Nicholas H Oberlies4, Eve-Isabelle Pecheur5, Harel Dahari6,7,8, Peter Ferenci9, Jean-Michel Pawlotsky10,11
1Department of Laboratory Medicine, University of Washington, Seattle, WA, USA
2Department of Microbiology, University of Washington, Seattle, WA, USA
3Department of Global Health, University of Washington, Seattle, WA, USA
4Department of Chemistry and Biochemistry, University of North Carolina at Greensboro, Greensboro, NC, USA
5UMR INSERM 1052/CNRS 5286, Lyon, France
6Department of Medicine, University of Illinois at Chicago, Chicago, IL, USA
7Theoretical Biology and Biophysics, Los Alamos National Laboratory, Los Alamos, NM, USA
8Present address: Department of Medicine, Division of Hepatology, Loyola University Chicago, Maywood, IL, USA
9Internal Medicine 3, Medical University of Vienna, Vienna, Austria
10National Reference Center for Viral Hepatitis B, C and Delta, Department of Virology, Henri Mondor Hospital, University of Paris-Est, Creteil, France
11INSERM U955, Creteil, France
Abstract
Silymarin, an extract of milk thistle seeds, and silymarin-derived compounds have been considered hepatoprotective since the plant was first described in ancient times. Hepatoprotection is defined as several non-mutually exclusive biological activities including antiviral, antioxidant, anti-inflammatory and immunomodulatory functions. Despite clear evidence for silymarin-induced hepatoprotection in cell culture and animal models, evidence for beneficial effects in humans has been equivocal. This review will summarize the current state of knowledge on silymarin in the context of HCV infection. The information was collated from a recent workshop on silibinin in Germany.
Introduction
Despite clear evidence for silymarin-induced hepatoprotection in cell culture and animal models [1], evidence for beneficial effects in humans has been equivocal. This may be attributable to a relative dearth of well-designed and controlled clinical trials and a generalized pervasiveness of inadequate attention to silymarin nomenclature and composition. The potential of silymarin-derived natural products for hepatitis C originated from cell-culture-based studies showing that silymarin blocked HCV infection [2,3].
However, recent reports exemplify a bipolarity of silymarin action. On one hand, there is the striking observation by Ferenci et al. [4] that Silibinin-C-2', 3-dihydrogen succinate, disodium salt (Legalon SIL), an intravenous, aqueous soluble formulation of silybin A and silybin B (synonymously called silibinin A and B; Figure 1) that collectively comprise the mixture known as silibinin, reduces viral load in HCV-infected patients. This has led to other studies showing that Legalon SIL prevents re-infection of the graft during liver transplantation through its pretransplant antiviral effect [5-7]. At the opposite end of the spectrum is the report from the randomized double-blind placebo-controlled SyNCH trial using the highest oral doses of silymarin to date (700 mg per day) that showed lack of any demonstrable effect of orally administered silymarin in reduction of HCV viral loads and alanine aminotransferase in infected patients [8]. In the last few years, new investigators have entered the field with the goals of understanding how silymarin, silibinin, Legalon SIL and other silymarin-derived flavonolignans inhibit HCV infection, as well as defining other potential clinical applications of Legalon SIL.
With these goals in mind, the first workshop on silibinin was held on 10 February 2012, in Cologne, Germany. Organized by Ralf-Torsten Pohl from Madaus Rottapharm in Cologne, Germany, the manufacturer of Legalon SIL, and an affiliate of the international Rottapharm-Madaus-Group headquartered in Monza, Italy, the workshop was chaired by Peter Ferenci and Jean-Michel Pawlotsky, and attracted an international group of researchers in this small but growing field.
History and composition of silymarin and silibinin
Ralf Torsten-Pohl (Madaus Rottapharm) gave a brief overview of the history of silymarin and silibinin, stressing the ancient origins of silymarin, where the herbal extract has been referenced in early medical texts. He discussed briefly the taxonomy of milk thistle (Silybum marianum [L] Gaertn [Asteraceae]). Dr Pohl's talk also highlighted how silymarin is extracted from the seeds (achenes), purified to form the mixture of the two isomeric silybin components, and then esterified with succinic acid to form the disuccinate disodium salt, Legalon SIL. Moreover he reviewed the large body of published studies on silymarin components that describe their pleiomorphic biological and pharmacodynamic behaviour in different preclinical and clinical settings covering diverse fields of medicinal use.
Nicholas Oberlies (University of North Carolina at Greensboro, Greensboro, NC, USA) presented an overview of the components of silymarin, with particular emphasis on the structures and nomenclature of the various mixtures of flavonolignans. Focusing on the most well-studied mixtures, silymarin and silibinin, he compared and contrasted these through a series of structural diagrams, chromatograms and lists.
The mixture silymarin constitutes major flavonolignans silybin A, silybin B, isosilybin A, isosilybin B, silychristin, isosilychristin and silydianin. These flavonolignans are likely derived from the parent flavonoid, taxifolin, which represents the left-hand side of all those compounds, condensing with coniferol alcohol. The various ways in which coniferyl alcohol combines with taxifolin imparts a fair degree of molecular diversity. By contrast, silibinin is a two-component mixture, made up of only silybin A and silybin B, and as noted by Pohl, Legalon SIL results from a synthetic addition of two succinic acid moieties to both silybin isomers. Dr Oberlies also showcased some of the chromatographic methods his group has developed to purify the individual components to >98% purity [9]. In the structural descriptions, Dr Oberlies stressed the importance of proper nomenclature of silymarin-derived compounds [10]. Indeed, the literature is rife with misnaming of compounds, and this lack of attention to detail likely adds to the confusion surrounding the biology of these compounds in the literature. He stressed that in the interest of furthering the medical potential of these compounds, scientists should strive to be clear, correct, and consistent in the nomenclature of silymarin-derived compounds and mixtures. Please refer to Table 1 for a guide to milk thistle nomenclature.
Human clinical experience
Over 400 subjects have received oral silymarin in multiple clinical trials [8,11-17]. In approximately half of these studies, some improvement in liver histology or reduction in liver enzymes was observed [11-13,16]. In the HALT-C trial, approximately one-quarter of the 1,145 patients were using silymarin at baseline [18]. In a follow-up study of 1,049 HALT-C subjects [19], 34% of subjects were consuming silymarin at baseline, which was associated with lower hepatic collagen content on study biopsies and less histological progression. However, no effect was seen for clinical outcomes in this study. In all studies cited above, no reduction in HCV viral loads was observed.
Peter Ferenci (University of Vienna, Vienna, Austria) reviewed his pioneering studies on Legalon SIL administration to HCV-infected patients [4]. The first study demonstrated that Legalon SIL induced dose-dependent, log-fold reductions in HCV RNA level in 20 subjects. However, the virological response to Legalon SIL was heterogeneous from one patient to another. Particularly intriguing were his data showing that 2-3 weeks of daily Legalon SIL intravenous infusion may 'rescue' pegylated interferon plus ribavirin non-responders, obtaining 40% sustained virological response rates [20]. Similar data were recently described by Biermer et al. [21] who also successfully rescued patients ailing on treatment with protease inhibitor containing triple regimes. Thus, Legalon SIL (like other inhibitors of HCV replication) may be interferon-sensitizing, enabling the treatment of interferon non-responders with triple therapy. Moreover, Legalon SIL appears to suppress viral load in patients infected with genotypes 1, 3 and 4, whereas the effect of Legalon SIL on genotype-2-infected patients is currently unknown. Dr Ferenci also reviewed the cases in which Legalon SIL prevented re-infection of the graft following liver transplantation [5-7]. These data suggest that Legalon SIL may prove clinically valuable in the transplant setting, where therapeutic options are still limited.
Mechanisms of silibinin action based on in vitro investigations
Despite the clear reductions in viral load in HCV-infected patients treated with Legalon SIL, the mechanisms by which HCV suppression by silymarin, silibinin, and Legalon SIL occurs have not been fully characterized. In vitro studies have suggested multiple mechanisms may be operative.
Abdelhakim Ahmed-Belkacem, who is in Jean-Michel Pawlotsky's lab (INSERM U955, Henri Mondor University Hospital, Creteil, France), reviewed data showing that silymarin-derived compounds, Legalon SIL, and structurally related flavonoids can inhibit HCV RNA-dependent RNA polymerase (RdRp) activity in in vitro assays with recombinant HCV non-structural 5B (NS5B) protein with inhibitory concentrations 50% (IC50) in the range of 75-100 μM [22]. A focused screen of 44 compounds belonging to different subgroups of flavonoids, including flavones, flavonols, isoflavones, flavanols and flavonoid glycosides revealed that the flavonoid, quercetagetin, had the strongest RdRp inhibitory activity with IC50 of 6.7 ±1.0 μM and 4.3 ±0.7 μM against genotype 1a H77 and genotype 1b J4 NS5B isolates, respectively. Interestingly, quercetagetin was fivefold less efficient against RdRp from genotype 2a strain JFH1, with an IC50 of 20.5 ±2.8 μM. These data suggest that Legalon SIL may differentially modulate RNA polymerase of different HCV genotypes. Kinetic analyses showed non-competitive inhibition with nucleoside triphosphates. However, although the comparisons were made on key structural components of highly related flavonolignans, the compared structures differ in other regions of the molecules. Thus, it is not clear how changes at distant molecular positions affect overall structure and function of flavonoids.
Stephen J Polyak (University of Washington, Seattle, WA, USA) described the characterization of the hepatoprotective activities of silymarin and the seven major flavonolignans in silymarin using cell culture-based assays that measure antiviral, antioxidant and anti-inflammatory actions in liver cells, and immunomodulatory actions on T-cells [2,23]. A comparison of the effects of Legalon SIL versus the parent natural product mixture silibinin was also presented, focusing on the relationships between NS5B polymerase inhibition and antiviral activities. It was emphasized that Legalon SIL inhibits NS5B-1b better than silibinin, and silibinin is a poor inhibitor of NS5B-2a [24]. Regarding inhibition of HCV replication in subgenomic replicon cell lines, Legalon SIL inhibits genotype 1b but not 2a, while silibinin does not inhibit replication in 1b or 2a replicon cell lines. Since JFH-1 infection is more effectively inhibited by silibinin than by Legalon SIL, the data suggest that if indeed polymerase activity is involved in suppression of HCV infection by Legalon SIL, there may be genotype-dependent differential susceptibilities of NS5B polymerases to Legalon SIL. This is in line with the NS5B polymerase inhibition study described by Dr Ahmed-Belkacem, although in their study, silibinin and Legalon SIL had anti-HCV activity in both the genotype 1b subgenomic replicon and the JFH1 model [14]. These differences could be explained by different experimental conditions and/or cell lines. Moreover, Legalon SIL is by design soluble in aqueous solution while silibinin, silymarin, and all silymarin-derived flavonolignans are insoluble in aqueous solution and require organic solutions such as DMSO or methanol for solubilization. In this respect, Legalon SIL is thought to partition well into the membrane lipid-water interface [25]. Thus, the question arises as to whether differences in solubility contribute to the clear and demonstrable differences in HCV infection, replication, and polymerase inhibition between Legalon SIL and silibinin.
Julie Blaising, who is in Eve-Isabelle Pecheur's lab (UMR INSERM 1052/CNRS 5286, Lyon, France), presented exciting new data that continue the story on the silibinin inhibition of HCV entry at the fusion stage previously described by her group [3,24]. Using a variety of sophisticated techniques including spinning disk confocal microscopy, Ms Blaising showed that during HCV entry, cell culture-derived HCV virions (HCVcc) colocalize in clathrin-containing vesicles, and as a consequence of the interaction with vesicles that have markers of early endosomes, the virus particles transiently adopt low velocity movement. As the endosomes mature into late endosomes, the interaction with clathrin ceases, and HCVcc resume high velocity movement. Ms Blaising demonstrated that in presence of silibinin, HCVcc dissociation from clathrin vesicles does not occur, the particles become trapped in early endosomes, retain low velocity movement, and never become associated with late endosomes. Thus, silibinin inhibits key steps in the clathrin-dependent entry of HCV into hepatocytes. The data suggest that silibinin may impact fundamental cellular processes that could have profound influence on pathogens beyond HCV. These studies typify how one can use natural products or natural product containing mixtures to probe cellular functions to elucidate key biological processes and their effects on host-pathogen interactions.
Application of HCV kinetics to understanding how Legalon SIL works
Harel Dahari (University of Illinois, Chicago, IL, USA; Los Alamos National Laboratory, Los Alamos, NM, USA) reviewed the principles of mathematical modelling of HCV dynamics during antiviral treatment and examined the kinetics of HCV RNA changes observed during directly-acting antiviral (DAA) drug administration that might be relevant to understanding the modes of action of Legalon SIL against HCV. The original model published in 1998 [26] posits that HCV RNA decline is biphasic, consisting of an early, rapid phase lasting approximately 2 days that reflects the direct antiviral effects of interferon-α (IFN) in blocking virus production/release. The second phase is protracted in time (days-weeks), has a slower viral RNA decline, and presumably reflects the loss/death of infected cells. Dr Dahari reviewed the current state of modelling HCV kinetics and the challenges to the original biphasic viral decline model that arise when fitting HCV kinetics during DAA treatment [27].
Jeremie Guedj (Los Alamos National Laboratory) [28] described a recent publication presenting a modelling analysis of HCV RNA kinetics from 25 patients infected with HCV genotype 1 or 4, who were treated for 7 days with monotherapy of 10, 15 or 20 mg/kg/day of Legalon SIL. Drs Guedj, Dahari and colleagues attempted to resolve the controversy in the field of whether Legalon SIL inhibits viral production, which is expected if the RdRp activity is inhibited [3,22,24,29], or whether Legalon SIL blocks other components of the HCV lifecycle such as virus entry or release [3,24]. Interestingly, the antiviral profile of Legalon SIL resembles that of IFN during the first phase, characterized as a rapid, dose-dependent decrease in viral RNA levels in serum.
The second-phase decline induced by Legalon SIL is not dose-dependent and resembles that of an RdRp inhibitor [30]. Moreover, half of Legalon-SIL-treated patients had biphasic RNA decay profiles, while the other half had monophasic decay patterns. Thus, Legalon SIL appears to induce two classes of viral RNA decay profiles, suggesting the possibility that blocking both virus production/release (probably by RdRp inhibition and later steps in virion release) and virus entry mechanisms contribute to the anti-HCV effects of Legalon SIL.
Pharmacokinetics and pharmacodynamics of silymarin preparations
A recent study by Schrieber et al. [31], indicated that patients with non-alcoholic fatty liver disease show biphasic and in some cases triphasic plasma concentrations of silymarin flavonolignans. This may be due to enterohepatic recycling of silymarin flavonolignans, which involves the circulation of parent and conjugated (by glucuronidation and/or sulfation) versions of silymarin flavonolignans. The metabolized (that is, conjugated) flavonolignans are transported out of hepatocytes by hepatobiliary transporters into the bile. Upon encountering the small intestine, the parent flavonolignans can be reformed by deconjugation enzymes in intestinal flora. The deconjugated flavonolignans are then returned to the liver via the portal blood supply, where they are again taken up by hepatocytes. Thus, enterohepatic recycling can elicit profound differences in plasma flavonolignan pharmacokinetics, thereby influencing the hepatoprotective actions of Legalon SIL. Moreover, it has also been shown that patients with cirrhosis display higher plasma levels of silymarin flavonolignans [32]. Future studies should formally characterize flavonolignan metabolism following Legalon SIL versus silymarin administration.
Key issues and areas for future research
First, a key issue involves differences between in vitro and in vivo dosing of silymarin-derived compounds. The in vitro antiviral activities of silymarin and silymarin-derived flavonolignans are observable at concentrations generally >20 μM. In healthy subjects, oral dosing of silymarin results in very low plasma concentrations of major flavonolignans in the range of 50-300 ng/ml due to their rapid metabolism to glucuronide and sulfate conjugates [8,32-37]. The key point is that silymarin as an oral formulation is not highly bioavailable, with a pharmacokinetic profile that achieves peak plasma concentrations 1-2 h post-dosing, with elimination in 4-6 h. However, in HCV patients with advanced liver disease, three- to fivefold higher plasma concentrations of flavonolignan conjugates are achieved compared to healthy subjects [38], while in patients with prostate cancer, plasma levels of silybin A and B up to 40 μg/ml (approximately 80 μM) are achieved with high oral doses of silipide, a formulation of silibinin with phospholipids [39]. Thus, it is possible that with the correct formulation for oral dosing, plasma levels of flavonolignans that approach in vitro concentrations can be achieved. However, results from the SyNCH trial, which administered the highest oral doses of silymarin to date, were presented at the AASLD meeting in 2011 and recently published [40]. The patients achieved 2-2,000 ng/ml of silymarin flavonolignans and there was no significant change in serum alanine aminotransferase activity or RNA levels in the silymarin treatment arms [8]. While it is logical to assume that intravenous dosing of Legalon SIL leads to higher serum levels of silybin A and B as compared with oral dosing, it is not clear how much higher. Thus, there is a need for additional clinical trials of Legalon SIL administration to patients.
The effects of Legalon SIL on different HCV genotypes should also be evaluated. These studies should have frequent sampling so that pharmacokinetic profiles can be accurately assessed. The investigators at the workshop also urged the company to publish existing Legalon SIL pharmacokinetic data. Although the SyNCH trial provided the highest oral silymarin dosing to date and was shown to be well-tolerated and safe, it will likely be impractical to increase pill burden to increase dose in future studies. Therefore, improved formulations of silymarin and silibinin for oral dosing that overcomes the bioavailability limitations are also urgently needed.
Second, hepatic levels of silymarin flavonolignans following oral and intravenous dosing in humans are unknown. Historically, liver biopsies have not been performed in patients receiving Legalon SIL and in the recently completed SyNCH trial [41]. However, it might be possible to justify studies in future trials, especially if Legalon SIL is used as a rescue therapy for previous non-responders to interferon-based therapies.
Third, despite the clear antiviral effects of silymarin and silymarin-derived compounds on HCV in vivo and in vitro, the mechanism(s) remain incompletely understood. While polymerase inhibition is demonstrable in vitro using purified NS5B proteins, the activity is modest. Cumulatively, the data support an important role for inhibition of virus entry in inhibition of HCV infection.
Silymarin, Legalon SIL, and silibinin also appear to inhibit release of progeny viruses [3,24], so it is possible that these compounds target other steps in the virus lifecycle that are dependent on host cell functions. Thus, additional studies should be performed to more clearly elucidate the mechanisms of antiviral action of silymarin. In doing so, these studies may reveal whether one or multiple mechanisms predominate.
Fourth, it is not known if Legalon SIL, which is the disuccinate versions of silybin A and silybin B, or silybin A and silybin B are the bioactive components. It is likely that the succinate moities on Legalon SIL could be cleaved by intracellular esterases, meaning that the parent silybin A and silybin B flavonolignans are the actual intracellular biological effectors of Legalon SIL. This issue is quite important because if it turns out that Legalon SIL is cleaved into silybin A and silybin B in cells, it likely means that the bioactivity of silibinin is more relevant than that of Legalon SIL, and as such, this information might help to settle the controversy over antiviral mechanisms of action of Legalon SIL versus silibinin. The situation becomes even more complex considering that silymarin flavonolignans are metabolized primarily through glucuronidation and also by sulfation [8,42], with multiple possible sites available for modification on each compound. Thus, metabolic studies on liver tissue are required to resolve these issues.
Fifth, Legalon SIL has shown promise in prevention of HCV infection during orthotopic liver transplantation [5-7]. Additional studies with recently FDA-approved protease inhibitors and other emerging (DAA) compounds are required to formally demonstrate the efficacy of Legalon SIL in conjunction with new antivirals.
Sixth, at present, Legalon SIL is administered daily for only 1-2 weeks before treatment is stopped. Despite having robust anti-HCV activity, this dosing regimen could conceivably create a scenario for development of resistance to Legalon SIL. Indeed, a recent study shows resistance to SIL can be selected both in vitro and in vivo [43]; thus, further studies on SIL resistance should be conducted.
With the recent approvals of new DAA compounds and second-generation drugs on the horizon, the renewed excitement and interest in compounds derived from the ancient botanical medicine known as silymarin may, at first glance, seem to be 'too little too late'. For patients who can afford and tolerate standard of care therapy, this statement may be true. However, many patients experience side effects that require cessation of therapy, and Legalon SIL may be useful as salvage therapy for prior interferon non-responders. Moreover, many developing countries may be unable to afford DAA combination therapy, let alone pegylated interferon plus ribavirin therapy. Thus, silymarin-derived compounds may provide clinical utility in these situations. Moreover, if hepatoprotective mechanisms of action and molecular targets for silymarin flavonolignans can be identified, this may lead to the structure-based development of potent new compounds that are orally bioavailable. The efficacy of Legalon SIL in orthotopic liver transplantation has been demonstrated, so continued research into this area may lead to identification of biomarkers of silymarin treatment and efficacy, novel targets for antiviral and anti-inflammatory drug design, and guide refinements in natural product-derived treatments for liver diseases in HCV- and HCV-HIV-coinfected patients. In this regard, we have recently shown that Legalon SIL inhibits in vitro HIV-1 infection of multiple cell types [44]. Furthermore, the antioxidant and anti-inflammatory effects of silymarin [45,46] may reduce pathogenesis of liver diseases of non-viral origin, as well as show efficacy in inflammatory diseases including cancer.
|
|
| |
| |
|
|
|