| |
Primary Efficacy and Safety Findings from Four Phase 3 Japanese Studies of Simeprevir Administered Once Daily Demonstrate Sustained Virologic Response in Genotype 1 Chronic Hepatitis C Adult Patients
|
| |
| |
Regulatory Application has now been filed for Simeprevir (TMC435) in Japan for the treatment of genotype 1 hepatitis C patients
22-Feb-13 Janssen now has submitted a regulatory application to the Japanese Ministry of Health & Welfare authorities seeking approval for simeprevir, administered with pegylated interferon (Peg-IFN) and ribavirin (RBV) for the treatment of genotype 1 ......http://www.natap.org/2013/HCV/030513_01.htm
CORK, Ireland (June 6, 2013) -- Janssen R&D Ireland (Janssen) today announced primary efficacy and safety results from four Japanese Phase 3 clinical studies demonstrating that the use of the investigational NS3/4A protease inhibitor simeprevir (TMC435) led to sustained virologic response 12 weeks after the end of treatment (SVR12) in patients with genotype 1 hepatitis C, when administered once daily with pegylated interferon and ribavirin. The four studies examined the use of simeprevir in patients who were treatment naïve, as well as patients who were non-responders to prior therapy or relapsed following treatment with pegylated interferon with or without ribavirin.
The data were presented today at The Japan Society of Hepatology's 49th Annual Meeting in Tokyo. The CONCERTO studies supported the new drug application for simeprevir, which was submitted to Japanese regulatory authorities in February 2013.
Approximately 1.5 to 2 million people in Japan are infected with the hepatitis C virus (HCV). After infection with HCV occurs, the infection persists in about 70 percent of cases, leading to the onset of chronic hepatitis C.
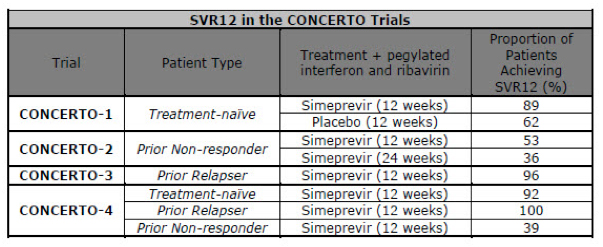
In CONCERTO-1, treatment-naïve patients were randomized to receive simeprevir or placebo for 12 weeks plus pegylated interferon and ribavirin for 24 or 48 weeks. Eighty-nine percent of patients in the simeprevir arm achieved SVR12 compared to 62 percent in the placebo arm.
In CONCERTO-2, patients who failed to respond to previous interferon-based therapy were randomized to two groups. In group one, patients received simeprevir plus pegylated interferon and ribavirin for 12 weeks, followed by response-guided treatment of pegylated interferon and ribavirin alone for 12 or 36 weeks. In group two, patients received simeprevir plus pegylated interferon and ribavirin for 24 weeks, followed by response-guided treatment of pegylated interferon and ribavirin alone for up to 24 weeks. Fifty-three percent of patients in the 12-week arm and 36 percent in the 24-week arm achieved SVR12, respectively.
In CONCERTO-3, patients who relapsed after previous interferon-based therapy received simeprevir plus pegylated interferon and ribavirin for 12 weeks, followed by response-guided treatment of pegylated interferon and ribavirin alone for 12 or 36 weeks. Ninety-six percent of patients achieved SVR12.
In CONCERTO-4, treatment-naïve patients and patients who relapsed after previous interferon-based therapy received simeprevir plus pegylated interferon and ribavirin for 12 weeks, followed by response-guided treatment of pegylated interferon and ribavirin alone for 12 or 36 weeks. Non-responder patients to previous interferon-based therapy received simeprevir plus pegylated interferon and ribavirin for 12 weeks, followed by pegylated interferon and ribavirin alone for 36 weeks. Ninety-two percent of treatment-naïve patients, 100 percent of prior relapsers and 39 percent of prior non-responders achieved SVR12.
The most common adverse events seen in patients receiving simeprevir plus pegylated interferon and ribavirin in CONCERTO-1 were similar to those observed with pegylated interferon and ribavirin alone and were also similar in the other studies (decreased white blood cell count, fever, anemia, decreased neutrophil count, malaise, headache and rash). Treatment discontinuation rates due to an adverse event in CONCERTO-1 were five percent in the simeprevir arm and 8 percent in the placebo arm, four percent in CONCERTO-2, four percent in CONCERTO-3 and one percent in CONCERTO-4.
About The CONCERTO Studies
Janssen's Phase 3 clinical program for simeprevir in Japan consists of four studies in patients with genotype 1 HCV: CONCERTO-1 in treatment-naïve patients, CONCERTO-2 and -3 in prior non-responders or patients who relapsed after prior interferon-based treatment, and CONCERTO-4 using different pegylated interferon treatments (pegylated interferon alfa-2b) in a broad patient population.
About Simeprevir
Simeprevir (TMC435) is an investigational NS3/4A protease inhibitor jointly developed by Janssen R&D Ireland and Medivir AB for the treatment of genotype 1 chronic hepatitis C in adult patients with compensated liver disease, including all stages of liver fibrosis. Simeprevir works by blocking the protease enzyme that enables the hepatitis C virus to replicate in host cells. New drug applications were recently submitted for simeprevir in Japan and the United States for the treatment of genotype 1 hepatitis C, and a Marketing Authorisation Application was submitted to the European Medicines Agency seeking approval of simeprevir for the treatment of genotype 1 or genotype 4 chronic hepatitis C. The U.S. FDA has granted Priority Review to the New Drug Application. To date, 3,787 patients have been treated with simeprevir in clinical trials.
For additional information about simeprevir clinical trials, please visit www.clinicaltrials.gov.
About Hepatitis C
Hepatitis C, a blood-borne infectious disease of the liver and a leading cause of chronic liver disease, is the focus of a rapidly evolving treatment landscape. Approximately 150 million people are infected with hepatitis C worldwide and 350,000 people per year die from the disease globally.
When left untreated, hepatitis C can cause significant damage to the liver including cirrhosis. Additionally, hepatitis C may increase the risk of developing complications from cirrhosis, which may include liver failure.
About Janssen R&D Ireland
At Janssen, we are dedicated to addressing and solving some of the most important unmet medical needs of our time in oncology, immunology, neuroscience, infectious diseases and vaccines, and cardiovascular and metabolic diseases. Driven by our commitment to patients, we develop innovative products, services and healthcare solutions to help people throughout the world. Janssen R&D Ireland is part of the Janssen Pharmaceutical Companies of Johnson & Johnson. Please visit http://www.janssenrnd.com for more information.
Media Contact: Daniel De Schryver
Mobile: +49 173 76 89 149
Investor Contact: Stan Panasewicz
Office: +1 (732) 524-2524
|
|
| |
| |
|
|
|