| |
Janssen HCV Drug Research & Development
|
| |
| |
Reported by Jules Levin
In the Spring Janssen & Gilead submitted New Drug Applications to the FDA requesting approval. The FDA hearing appears to be scheduled for Oct 24-25.
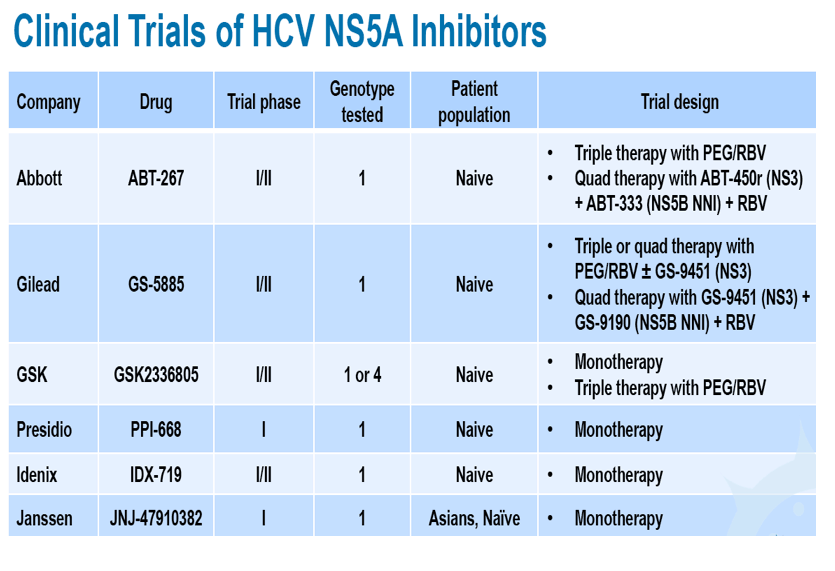
A Study of the Safety, Tolerability, Pharmacokinetics, and Antiviral Activity of JNJ-47910382 at Different Doses and Dose Regimens in Asian Genotype-1, Chronic, HCV-Infected Patients
Study in HCV-Infected Patients to Determine the Safety, Tolerability, Pharmacokinetics and Antiviral Activity of JNJ-47910382
A Study in Healthy Participants Investigating the Safety, Tolerability and Plasma Pharmacokinetics (PK) of Single Oral Doses of JNJ-47910382
IDX719 in Combination With Simeprevir and Ribavirin for 12 Weeks in Subjects With Chronic Hepatitis C Infection
http://www.clinicaltrials.gov/ct2/show/NCT01852604?term=Janssen+AND+Hepatitis+C&recr=Open&no_unk=Y&rank=9
Antiviral Drugs Advisory Committee .........October 24-25
http://www.fda.gov/AdvisoryCommittees/Calendar/ucm153468.htm
Simeprevir (TMC435) with peginterferon/ribavirin for chronic hCV genotype 1 infection in treatment-naïve patients: results from QUEST-1, a Phase III trial
http://www.natap.org/2013/EASL/EASL_10.htm
Simeprevir (TMC435) with peginterferon-α2a or -α2b and ribavirin in treatment-naïve HCV genotype 1 patients: QUEST-2, a randomised Phase III trial
http://www.natap.org/2013/EASL/EASL_18.htm
Simeprevir with Peginterferon/ribavirin for Treatment of Chronic HCV Genotype 1 Infection in Patients Who Relapsed After Previous Interferon-based Therapy: Results from PROMISE, a Phase III Trial
http://www.natap.org/2013/DDW/DDW_06.htm
COSMOS Study: SVR4 results of a once daily regimen of simeprevir (TMC435) plus sofosbuvir (GS-7977) with or without ribavirin in HCV genotype 1 null responders
http://www.natap.org/2013/CROI/croi_34.htm
Medivir Press Release: New Drug Application has been filed with FDA for Simeprevir (TMC435) for combination treatment of adult patients with genotype 1 chronic hepatitis C
http://www.natap.org/2013/HCV/040213_01.htm
Idenix Pharmaceuticals Announces Initiation of Phase II All-Oral Combination Study of Samatasvir (IDX719) and Simeprevir (TMC435) for the Treatment of Hepatitis C Virus (HCV) Infection
http://www.natap.org/2013/HCV/060213_04.htm
Combination Therapy of TMC647055 With Simeprevir (TMC435) in Genotype 1 HCV Patients
http://www.natap.org/2013/EASL/EASL_41.htm
TMC055 Monotherapy data:
Human safety, pharmacokinetics and antiviral activity of TMC647055, a novel HCV non-nucleoside polymerase
inhibitor.......
http://www.natap.org/2011/AASLD/AASLD_04.htm
Here is the link to the study on clinical trials.gov where you can see low dose 30mg RTV is used:
A Study to Evaluate the Safety, Tolerability, and Effectiveness of a 12-Week Combination Therapy of TMC647055 and TMC435 With a Pharmacokinetic Enhancer With and Without Ribavirin in Chronic Genotype-1 Hepatitis C Infected Patients.......http://clinicaltrials.gov/ct2/show/NCT01724086?term=tmc647055&rank=3
EASL: Combination Therapy of TMC647055 With Simeprevir (TMC435) in Genotype 1 HCV Patients - (04/30/13)
2nd generation NS5As
In Vitro Resistance Analysis of Merck's HCV NS5a Inhibitor MK-8742 Demonstrates Increased Potency AgainstClinical Resistance Variants and Improved Resistance Profile
http://www.natap.org/2012/EASL/EASL_46.htm
GS-5816, a Second-Generation HCV NS5A Inhibitor With Potent Antiviral Activity, Broad Genotypic Coverage, and a High Resistance Barrier
http://www.natap.org/2013/EASL/EASL_34.htm
Healthy Volunteer First-in-Human Evaluation of GS-5816, a Novel Second Generation Broad-Genotypic NS5A Inhibitor With Potential for Once-Daily Dosing - (04/29/13)
ACH-3102, A Second Generation NS5A Inhibitor, Demonstrates Potent Antiviral Activity in Patients with Genotype 1A HCV Infection Despite the Presence of Baseline NS5A-Resistant Variants - (05/09/13)
FINDINGS FROM CLINICAL VIROLOGY STUDIES ON ACH-3102 ARE CONSISTENT WITH PRECLINICAL OBSERVATIONS ON ITS IMPROVED POTENCY AGAINST GENOTYPE-1A HCV AND RESISTANT VARIANTS - (05/09/13)
No Clinically Significant Pharmacokinetic Interaction Between Sovaprevir and ACH-3102 in Healthy Volunteers - (05/09/13)
Synergistic Interactions of HCV NS5A Replication Complex Inhibitors Sensitize Resistant Variants and Enhance the Efficacy of Daclatasvir (DCV, BMS-790052) In Vitro and In Vivo - (04/27/13)
PRECLINICAL CHARACTERISTICS OF ACH-3102: A NOVEL HCV NS5A INHIBITOR WITH IMPROVED POTENCY AGAINST GENOTYPE-1A VIRUS AND VARIANTS RESISTANT TO 1ST GENERATION NS5A INHIBITORS
http://www.natap.org/2012/EASL/EASL_79.htm
NS5A BMS052
BMS052 HCV-NS5A Monotherapy in Phase 3 Now
http://www.natap.org/2012/HCV/082412_03.htm
NS5A BMS052 + nuc GS-7977 100% or 95% SVR for Patients Who Did Not Achieve SVR with Boceprevir/Telaprevir Triple Therapy, Resistance & Gt3
http://www.natap.org/2013/EASL/EASL_98.htm
|
|
| |
| |
|
|
|