| |
HAART & The Brain: "CNS Toxicity of Antiretroviral Drugs", "Can antiretroviral therapy prevent HIV-associated cognitive disorders?"
|
| |
| |
Download the PDF here
Download the PDF here
CNS Toxicity of Antiretroviral Drugs
17th CROI Feb 16-20 2010 SF
J. Liner, R.B. Meeker, and K. Robertson*
Department of Neurology and Neurobiology Curriculum, University of North
Carolina, Chapel Hill
http://www.natap.org/2010/CROI/croi_56.htm
-----------------------------
Can antiretroviral therapy prevent HIV-associated cognitive disorders?
Current Opinion in HIV & AIDS: Jan 2014
Winston, Alana,b; Vera, Jaime H.a,b
aFaculty of Medicine, Department of Medicine, Imperial College London, St Mary's Hospital Campus
bDepartment of HIV and Genitourinary Medicine, Imperial College Healthcare NHS Trust, London, UK
from Jules: in balancing the pros & cons of early HAART, I think early HAART is the better choice.
"Since the advent of effective cART, controversy has existed regarding the optimal timing for initiation of this therapy. This controversy continues, with the impact of cART on comorbidities, an important current consideration. With regards to cognitive function impairment, although cART has shown beneficial effects in individuals with symptomatic cognitive decline and in individuals commencing cART for the first time, the impact of cART has on cognitive function in asymptomatic individuals with high CD4+ lymphocyte counts remains unknown......A neurology sub-study with the Strategic Timing of Antiretroviral Therapy study; ClinicalTrials.gov Identifier NCT00867048 is assessing the impact of the earlier initiation of cART on cognitive function.......comorbidities and lifestyle factors have been associated with higher rates of cognitive impairment. Comorbidities such as cardiovascular disease and diabetes are among the strongest predictors of cognitive function impairment within the Strategic Management of ART study.....Coinfections are also important risk factors for cognitive impairment. HIV-infected individuals coinfected with chronic viral hepatitis-C infection have an increased risk of cognitive impairment compared to those infected with HIV alone [29]. Similarly, poorer cognitive function has been described in HIV-infected individuals with higher cytomegalovirus antibody concentrations and those with past history of syphilis infection [30,31]. It is possible that these coinfections are associated with increased neuroinflammatory responses and nervous system injury......Lifestyle factors such as cigarette smoking, recreational drug and alcohol use are also reported to be highly prevalent in HIV-disease as are diagnoses of depressive symptoms, which may further fuel the incidence of this condition. Not only may these clinical risk factors themselves directly affect cognitive function (for instance, recreational drug-use), these may also contribute to the pathogenesis of other diseases leading to cognitive decline (for instance, cardiovascular disease contributing to vascular dementia). Moreover, the presence of these conditions may also lead to increased peripheral inflammatory responses [32], which could in turn also lead to neuroinflammation [33]."
"cerebral toxicities may ensue after the initiation of cART, which may cause cognitive decline. Laboratory studies suggest that many of the antiretroviral agents in current clinical use may have neuronal toxicities, even at the concentrations one would expect to observe in the CNS during general clinical usage [39]. In this study, cultured rat cortical neurones were challenged with several antiretroviral agents in current clinical use such as abacavir, efavirenz, etravirine, nevirapine and atazanavir within the range of concentrations one would expect to observe in plasma and CSF with standard clinical dosing. The study described a considerable loss of neurones caused by alterations of the neuronal calcium homeostasis and mitochondrial membrane potentials suggesting one possible mechanism for neuronal injury may be due to antiretroviral therapy itself.
Clinical data describing cerebral toxicities from antiretroviral therapy are sparse, however, knowledge in this field is expanding [40]. Cerebral imaging studies suggest toxicities may be present and are associated with the duration [41] or number [42] of nucleoside-reverse-transcriptase-inhibitor agents used within a cART regimen, which may be markers of mitochondrial toxicity. Clinical data regarding specific drug-toxicities are also starting to emerge. Within cohort studies, efavirenz use has been associated with poorer cognitive function [43] and switching from this agent associated with improvements in CNS-symptomatology [44]."
Abstract
Purpose of review: In general, the initiation of combination antiretroviral (cART) is associated with improvement in cognitive function. However, the impact of cART has on cognitive function in neurologically asymptomatic HIV-infected individuals initiating therapy at high CD4+ lymphocyte cell counts is unknown.
Recent findings: Cognitive function impairment remains prevalent despite effective cART. Several clinical risk factors for this condition have been described, including low nadir CD4+ lymphocyte cell count which may be associated with greater neuroinflammatory process, a potential pathogenic mechanism underlying this cognitive impairment. The earlier initiation of antiretroviral therapy could theoretically avoid this risk factor and limit the degree of neuroinflammation. On the converse, the earlier initiation of cART may be associated with the development of neuronal toxicities.
Summary: This review article highlights the recent literature and arguments for and against the earlier initiation of cART with regards to cognitive function.
INTRODUCTION
The advent of effective combination antiretroviral (cART) therapy has led to dramatic reductions in the incidence and prevalence of HIV-associated central-nervous-system (CNS) opportunistic-infections [1] and other AIDS-defining illnesses such as HIV-encephalopathy [2]. With the development of cART regimens with reduced toxicity and increased tolerability, the incidence of these conditions continues to decline [3].
Despite these advances, milder forms of HIV-associated cognitive impairment are reported to persist. Clinically significant cognitive function impairment has been reported in approximately 10-15% of effectively treated HIV-infected individual within cohorts worldwide [4-6]. Although higher prevalence rates of cognitive impairment in up to 50% of individuals has been described in recent times [7,8], these reports may be overestimations because of the analysis of cohorts including individuals not receiving effective cART and cohorts which included individuals who were otherwise asymptomatic who under-performed on formal cognitive testing wherein control data-sets were unavailable to interpret such findings [9,10]. Incidence rates are also high with the onset of de-novo cognitive symptomatology reported in 22% of individuals with clinical risk factors annually [11].
This cognitive decline is of clinical relevance. Typically, the domains of memory, attention and information processing are affected with the clinical consequences being reduced quality of life [12], reduced functioning on activities of daily living [13] and even increased mortality [14]. Furthermore, HIV-associated cognitive impairment may undermine sustained adherence to lifelong cART, jeopardizing both the long-term effectiveness of treatment for the individual and the prevention of onward HIV transmission.
Several underlying pathogenic mechanisms for this condition have been proposed, and many clinical risk factors have been identified. One important clinical risk factor is an individual's CD4+ lymphocyte count at nadir [15]. This had led to the hypothesis that the avoidance of low nadir CD4+ lymphocyte count, by commencing cART at an earlier stage of HIV-disease, could lead to a reduced incidence of HIV-associated cognitive impairment. In this review article, we firstly summarize some of the proposed pathogenic mechanisms that may underlie this condition with the associated clinical risk factors, and then in detail outline the impact of antiretroviral therapy in differing stages of HIV-disease.

MAIN REVIEW
Numerous pathogenic mechanisms have been implicated with the development of cognitive impairment in the cART era. These include on-going neuroinflammation despite effective cART, antiretroviral factors and general patient factors such as other comorbidities, coinfections and lifestyle factors Table 1.
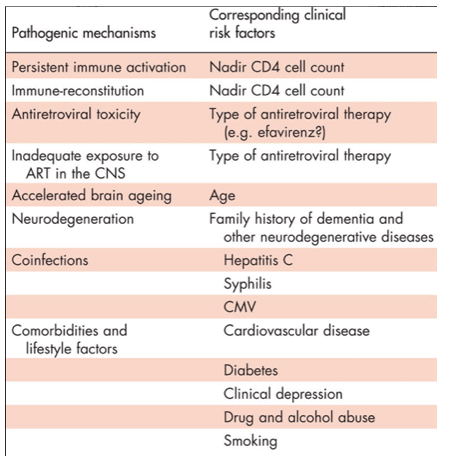
NEUROINFLAMMATION AND NADIR CD4+ LYMPHOCYTE COUNT
The main effectors of immune responses and inflammatory processes with the CNS are microglia, which constitutes approximately 10% of the entire brain cell population. Compelling evidence exists suggesting that activation of microglial cells which leads to neuroinflammation plays an important role in HIV-associated brain disease.
Prior to the advent of effective cART, in individuals with frank HIV-encephalopathy, increased microglial activation was demonstrated on postmortem examination [16] and utilizing PET imaging in which ligands which bind to activated microglial cells can be visualized in vivo[17]. More recently, microglial activation has also been reported in cART era, again both on postmortem examination in in vivo. The 'Edinburgh Brain Banks' group have described microglial activation to be present in effectively treated HIV-infected individuals deceased from non-AIDS and non-CNS-related diseases suggesting neuroinflammation is on-going despite cART [18]. PET imaging supports these findings with increased binding of PET-ligands which adhere to activated microglial cells in effectively treated HIV-infected individuals with mild forms of cognitive impairment and in those without any evidence of cognitive disease [19].
Furthermore, cerebrospinal-fluid (CSF) biomarker studies measuring concentrations of inflammatory markers produced by activated microglia have revealed that despite a reduction in the level of intrathecal immune activation observed with cART, significant on-going inflammation activity is evident [20]. Letendre et al.[21] measured the concentration of three inflammatory chemokines macrophage inflammatory protein-1α (MIP-1α), RANTES (regulated on activation, normal T cell expressed and secreted) and MIP-ß in 73 HIV-infected individuals and reported increased concentrations of all three biomarkers in the CSF of individuals with HIV-associated cognitive impairment compared with individuals without cognitive impairment. A prospective study looking at the effects of 12 weeks of cART on changes in immune-markers in 12 patients with cognitive impairment, demonstrated a reduction in MIP-1α and RANTES following the initiation of cART [22]. Other markers studied include IP-10 [23] and S100ß [24] in which increased concentrations in HIV-infected individuals with either more advanced or more rapidly progressive cognitive impairment have been reported. The literature in this field includes many studies reporting differences in both blood CSF biomarkers in HIV-infected individuals on effective cART compared with HIV-uninfected individuals and differences in such biomarkers in HIV-infected individuals with cognitive impairment. Overall, the data suggest that patients with HIV-associated cognitive disorders tend to have higher levels of inflammatory blood and CSF biomarkers.
An individual's CD4+ lymphocyte count nadir is a marker of historical HIV-disease progression and a well described risk factor for the presence of cognitive impairment in many cohorts [4,10,15,25]. On the converse, for individuals on effective antiretroviral therapy, current CD4+ lymphocyte count has not been reported to be a strong risk factor for the presence of cognitive impairment. This may be due to HIV-disease progression, whereby as HIV-disease progresses, increased peripheral and CNS immune activation occurs. It is possible this process may persist despite effective cART and be a potential explanation for the strong clinical correlation observed between nadir CD4+ lymphocyte count and cognitive function, in which individuals with lower nadir CD4+ lymphocyte counts are more likely to display poorer cognitive function, despite higher CD4+ lymphocyte counts after effective cART.
PATIENT FACTORS
Many comorbidities and lifestyle factors have been associated with higher rates of cognitive impairment. Comorbidities such as cardiovascular disease and diabetes [26,27] are among the strongest predictors of cognitive function impairment within the Strategic Management of ART study, one of the largest HIV-treatment studies to date. Such noncommunicable comorbidities are reported to be highly prevalent in treated HIV-infected individuals and may potentially increase prematurely with ageing [28]. Therefore, within ageing HIV-infected populations, the presence of cognitive impairment could increase exponentially over time.
Coinfections are also important risk factors for cognitive impairment.
HIV-infected individuals coinfected with chronic viral hepatitis-C infection have an increased risk of cognitive impairment compared to those infected with HIV alone [29]. Similarly, poorer cognitive function has been described in HIV-infected individuals with higher cytomegalovirus antibody concentrations and those with past history of syphilis infection [30,31]. It is possible that these coinfections are associated with increased neuroinflammatory responses and nervous system injury.
Lifestyle factors such as cigarette smoking, recreational drug and alcohol use are also reported to be highly prevalent in HIV-disease as are diagnoses of depressive symptoms, which may further fuel the incidence of this condition. Not only may these clinical risk factors themselves directly affect cognitive function (for instance, recreational drug-use), these may also contribute to the pathogenesis of other diseases leading to cognitive decline (for instance, cardiovascular disease contributing to vascular dementia). Moreover, the presence of these conditions may also lead to increasedperipheral inflammatory responses [32], which could in turn also lead to neuroinflammation [33].
THE IMPACT OF ANTIRETROVIRAL THERAPY
On a population level, there is compelling evidence to suggest cART is beneficial with regards to cognitive function. The incidence of severe HIV-associated brain disease fell dramatically with the advent of effective cART and continues to decline with the development of regimens with reduced toxicity [3], and moreover cognitive function improves in HIV-infected individuals commencing cART for the first time [34,35].
A further hypothesis underlying the observed cognitive impairment in the cART era is inadequate of antiretroviral agents in the CNS compartment. A great amount of recent attention has focused on this hypothesis, which assumes that although cART regimens may suppress HIV-viraemia in the plasma compartment, the efficacy of viral suppression in the CNS compartment may not be optimal. This thinking has led to scorning systems, whereby antiretroviral agents are graded based on their pharmacokinetic properties with agents with greater potential exposure in the CNS having greater scores [36]. However, there is no conclusive evidence that different cART regimens with different pharmacokinetic properties are associated with differences in cognitive function [37]. Separate consideration should be given to cases of 'cerebro-spinal-fluid (CSF) viral escape', in which suppressive control of HIV RNA in the CNS is lost. This rare condition, distinct from HIV-associated cognitive impairment, is often promptly diagnosed, as symptoms are overt and generally responds to modifying cART following HIV-resistance testing [38].
On the converse, cerebral toxicities may ensue after the initiation of cART, which may cause cognitive decline. Laboratory studies suggest that many of the antiretroviral agents in current clinical use may have neuronal toxicities, even at the concentrations one would expect to observe in the CNS during general clinical usage [39]. In this study, cultured rat cortical neurones were challenged with several antiretroviral agents in current clinical use such as abacavir, efavirenz, etravirine, nevirapine and atazanavir within the range of concentrations one would expect to observe in plasma and CSF with standard clinical dosing. The study described a considerable loss of neurones caused by alterations of the neuronal calcium homeostasis and mitochondrial membrane potentials suggesting one possible mechanism for neuronal injury may be due to antiretroviral therapy itself.
Clinical data describing cerebral toxicities from antiretroviral therapy are sparse, however, knowledge in this field is expanding [40]. Cerebral imaging studies suggest toxicities may be present and are associated with the duration [41] or number [42] of nucleoside-reverse-transcriptase-inhibitor agents used within a cART regimen, which may be markers of mitochondrial toxicity. Clinical data regarding specific drug-toxicities are also starting to emerge. Within cohort studies, efavirenz use has been associated with poorer cognitive function [43] and switching from this agent associated with improvements in CNS-symptomatology [44].
---------------------------------------------
39. Robertson K, Liner J, Meeker RB. Antiretroviral neurotoxicity. J Neurovirol 2012; 18:388-399
"Antiretroviral neurotoxicity"
Kevin Robertson & Jeff Liner & Rick B. Meeker
J. Neurovirol. (2012)
Neurotoxicity of individual ARVs ranged from undetectable to moderate and was not due to neuron death No compound was highly toxic but neural damage in the form of dendritic beading and pruning was a common observation. This pathology was reflected in the loss of MAP-2 staining. Since beading and pruning have been documented under a variety of pathological conditions (Bellizzi et al. 2005; Greenwood et al. 2007; Takeuchi et al. 2005), they most likely reflect a general endpoint associated with neuronal dysfunction (Greenwood et al. 2007; Kim et al. 2010; Masliah et al. 1992). Under these in vitro test conditions, a wide range of median toxic concentrations (TC50) was seen with a low value of 2.2 ng/ml and high values of >5 μg/ml (Table 1). The TC50 of ABC, ddI, and NVP fell within the range of concentrations seen in CSF of patients on ARV therapy. The median toxic concentration of ABC, APV, ATV, ddI, EFV, ETR, NVP, RTV, TDF and 3TC, fell within the range of concentrations seen in the plasma suggesting that they may have significant neurotoxic effects if present in CNS at therapeutic concentrations. DRV, FTC, and MVC produced little toxicity at relevant plasma and CSF concentrations. The ratio of the therapeutic plasma concentration to the TC10 provided an index of the potential toxicity at concentrations that would suppress HIV replication. The TC10 value was used to reflect the earliest drop in the dose-response curve (approximately a 10 % decrease) in an effort to provide a conservative estimate of the concentration at which toxicity might begin. Twelve of the compounds were above this theoretical threshold and some exceeded the threshold by two logs. The range was greater than three logs indicating considerable variation in the relative risk. The lower toxicity indices for ddC, FTC, DRV, RTV and MVC indicate that it may be possible to deliver therapeutic concentrations of some drugs into the CNS with minimal relative risk.
The ratio of the CSF concentration of each drug to the TC10 provided an index of the risk of toxicity based on current use. The relatively high penetration of ABC and NVP yielded toxicity risk values much greater than zero indicating that they may be present in CSF at levels capable of producing neurotoxicity. DDI, 3TC, APV and ATV also had scores greater than zero but the predicted amount of toxicity would be minimal. Even in cases where higher levels of toxicity were seen, it is noteworthy that the toxic effects were not due to cell death. The toxic effects associated with dendrite beading and possibly pruning are likely to be reversible, raising the possibility that neuroprotective treatments may be effective in preventing or reversing ARV-associated damage.
Conclusions
As we develop new antiviral approaches to target HIV in the CNS greater attention to strategies that minimize the risk to neurons will be needed. Our studies indicate that there is a risk of direct neurotoxic interactions that varies substantially between compounds and that informed selection of ARVs and ARV combinations used to target the CNS may minimize adverse effects. While these data begin to identify potential risks, they should not be viewed as guidelines for the clinical use of ARVs. Similar studies will need to correlate in vitro measures to clinical observations to establish the validity of the in vitro toxicity measures. In addition, longterm effects of the compounds may give rise to other forms of damage and will need to be evaluated. Our ability to make rational choices for therapies directed to the CNS is currently limited by a paucity of data on the complex interactions of ARV compounds with neural tissue. Our studies presented here illustrate that the potential for neural damage and dysfunction is significant and highlight the need for further exploration of the mechanism of neurotoxicity of these compounds individually and in combinations.
---------------------------------------------------------------
THE EARLIER INITIATION OF ANTIRETROVIRAL THERAPY
Current guidelines recommend the initiation of antiretroviral therapy in all HIV-infected individuals with clinically overt cognitive-symptomatology, which is considered related to HIV-disease regardless of CD4+ lymphocyte count [45].
However, could the early initiation of antiretroviral therapy, prior to current guidelines recommendation based on CD4+ lymphocyte count be beneficial to cognitive function in cognitively asymptomatic HIV-infected individuals? When considering this, several of the above principles should be taken into consideration.
First, the earlier initiation of antiretroviral therapy would avoid HIV-infected individuals obtaining low nadir CD4+ lymphocyte, and therefore avoiding this risk factor. However, one also needs to consider that this is a risk factor described within clinical cohorts and although this may be linked to increased neuroinflammation and other pathogenic mechanisms associated with the development of cognitive impairment, cohort biases should also be taken into consideration. If one considers two HIV-infected individuals, one with a very low nadir CD4+ lymphocyte count and one with a high nadir CD4+lymphocyte count then these individuals general phenotypes could be quite different. The underlying reasons that one individual has developed a low nadir CD4+ lymphocyte may include late HIV-disease presentation, greater risk-taking behaviour and general poorer engagement with healthcare services. Therefore, this individual may inherently have poorer cognitive function rather than this being directly related to HIV-disease, and as such the earlier initiation of antiretroviral therapy may have little if any impact on cognitive function.
Secondly, the earlier initiation of antiretroviral therapy would allow individuals to benefit from the effects of cART on cognitive function at an earlier stage. However, again on the converse, such a strategy would expose individuals to the potential cerebral toxicities of antiretroviral therapy for a longer duration of time and from an earlier stage of HIV-disease.
Therefore, there remains equipoise regarding the role of commencing cART in neurologically asymptomatic individuals with high CD4+ lymphocyte counts. A neurology sub-study with the Strategic Timing of Antiretroviral Therapy study; ClinicalTrials.gov Identifier NCT00867048 is assessing the impact of the earlier initiation of cART on cognitive function. In this study, individuals with CD4+ lymphocyte counts above 500 cells/ul are randomized to immediate cART or deferred cART (until CD4+ lymphocyte counts falls below 350 cells/ul) with prospective cognitive function assessed during the study period. Results from this study may definitively answer the above conundrum regarding the impact of the earlier initiation of cART on cerebral function.
CONCLUSION
Since the advent of effective cART, controversy has existed regarding the optimal timing for initiation of this therapy. This controversy continues, with the impact of cART on comorbidities, an important current consideration. With regards to cognitive function impairment, although cART has shown beneficial effects in individuals with symptomatic cognitive decline and in individuals commencing cART for the first time, the impact of cART has on cognitive function in asymptomatic individuals with high CD4+ lymphocyte counts remains unknown.
|
|
| |
| |
|
|
|