| |
PrEP/(ART) Update- Once Monthly or Every 3 Months PrEP & ART: Rilpivirine Long-Acting; tenofovir intravaginal ring for PrEP; combination PrEP & ART injections Every Month or 3 Months with 2 long-acting drugs GSK744
|
| |
| |
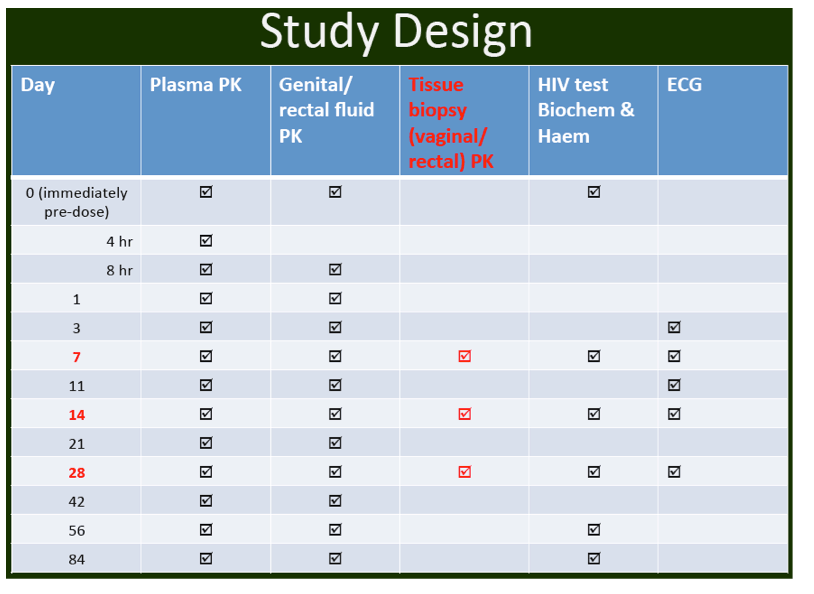
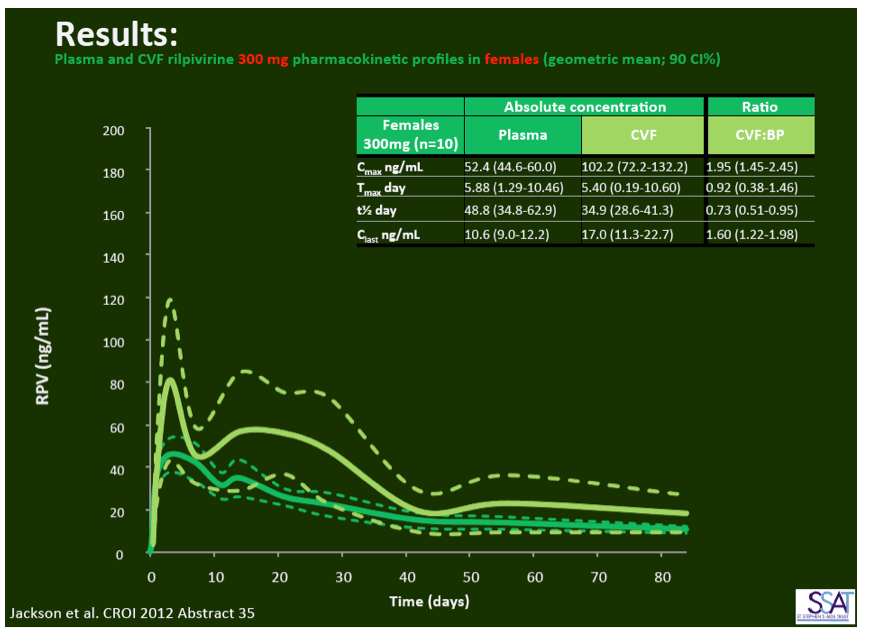
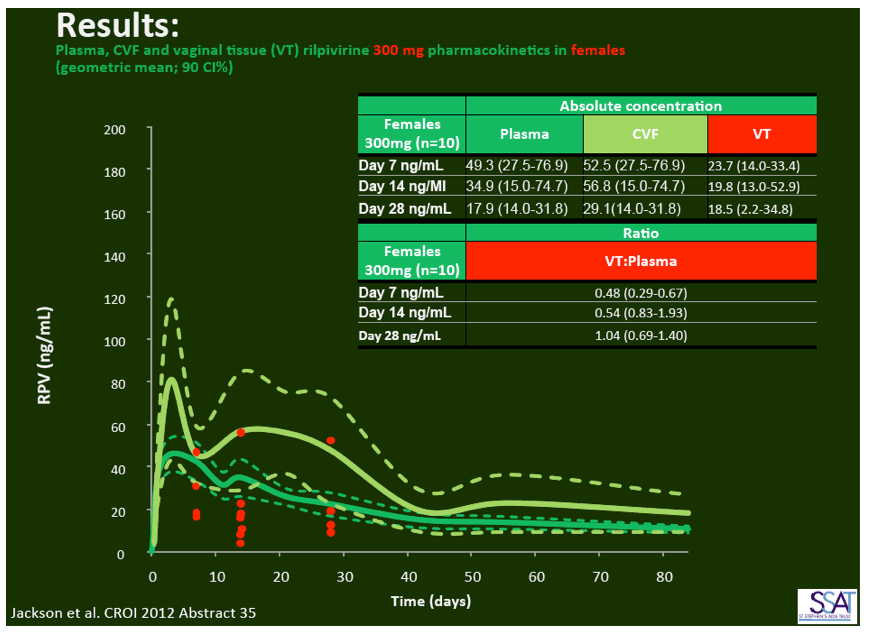
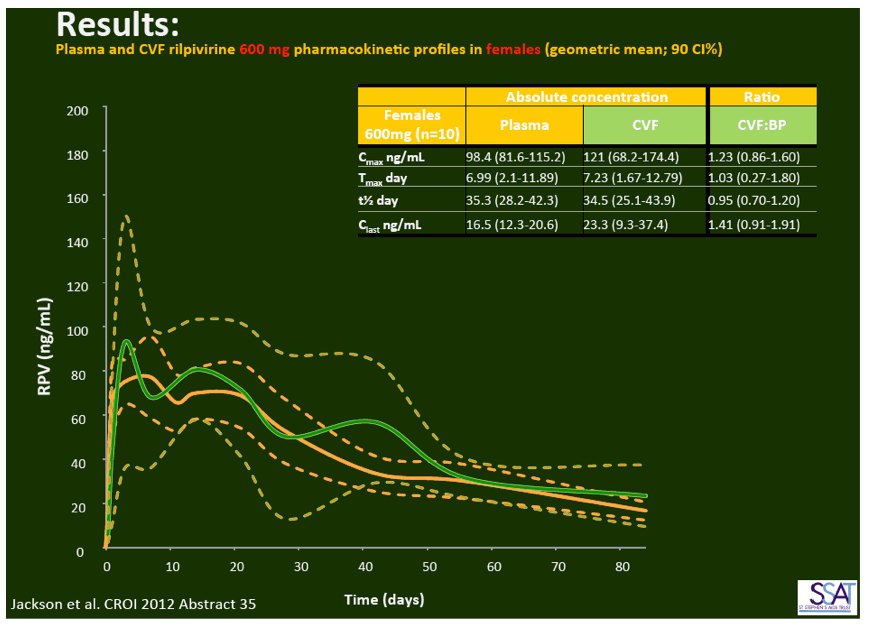
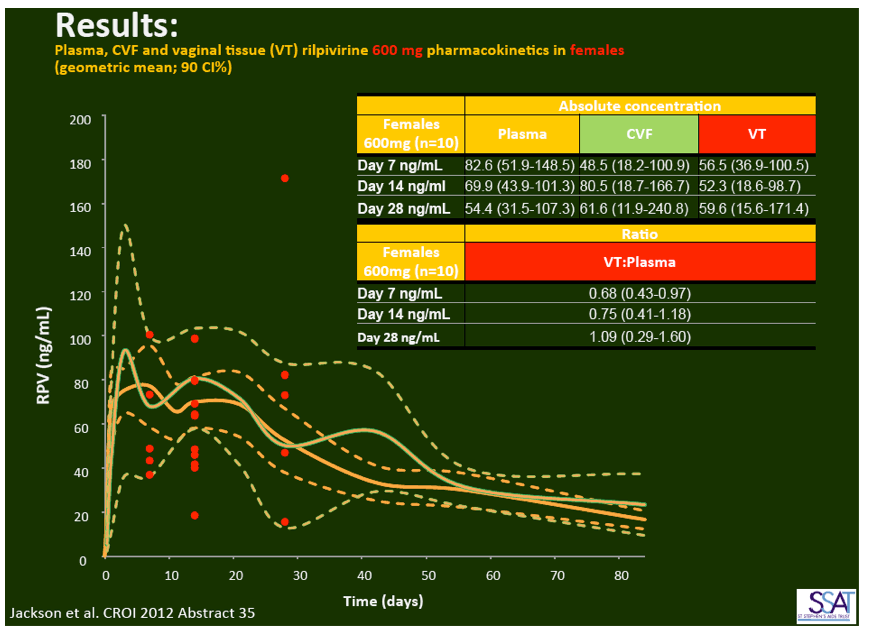
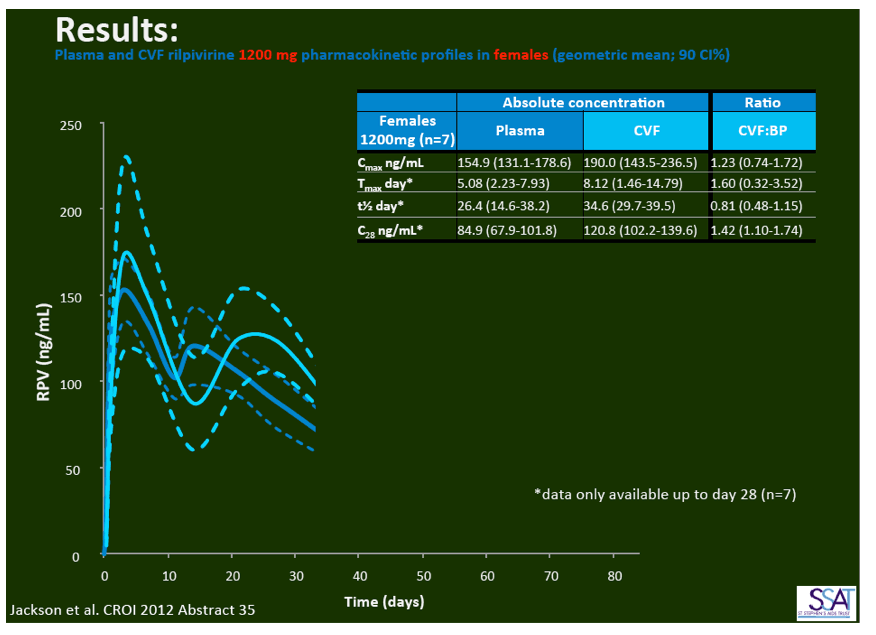
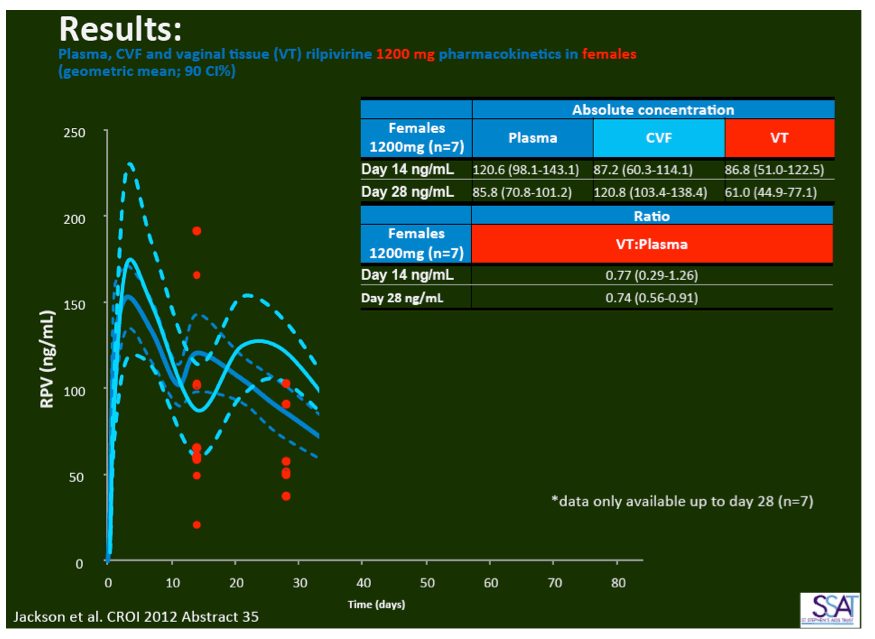
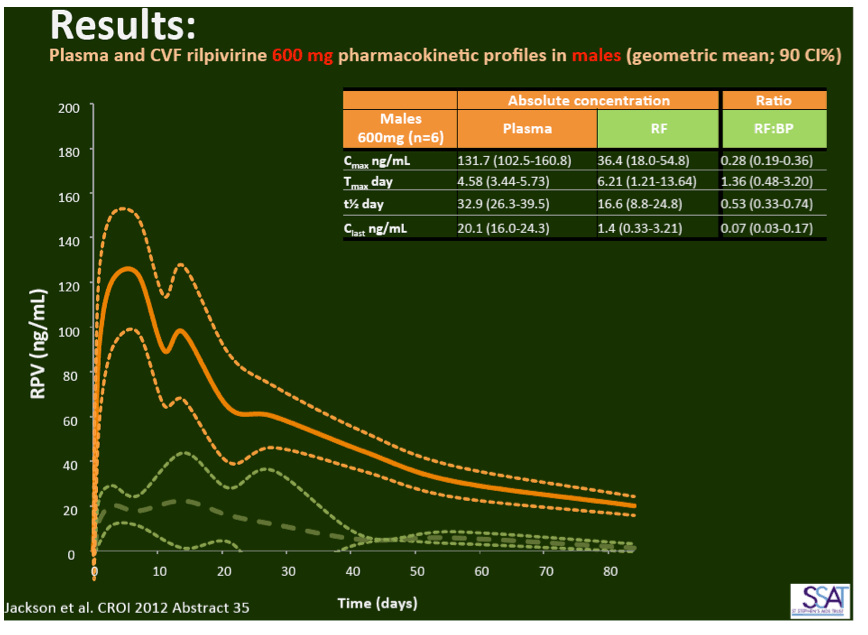
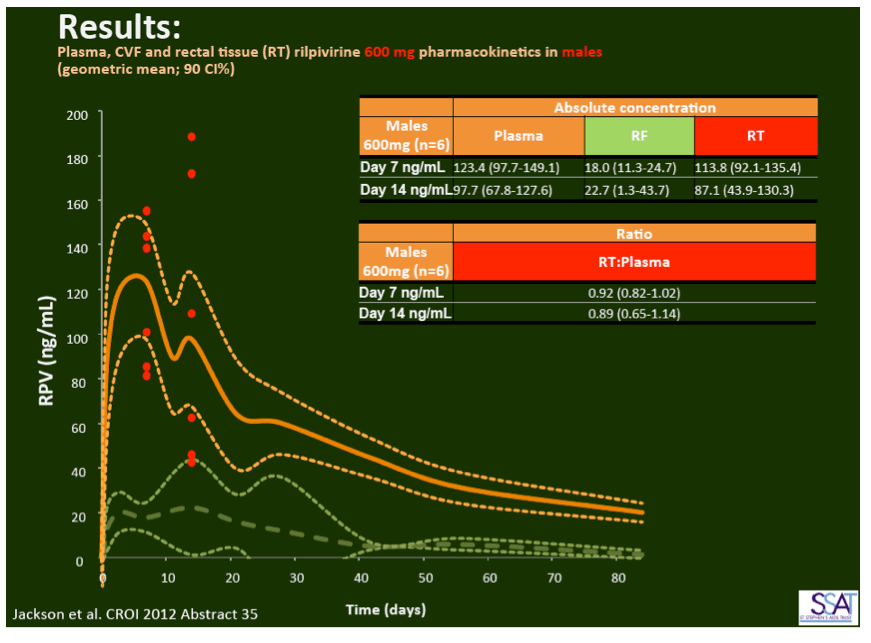
- below are slides presented at CROI/2012: Rilpivirine Long-Acting (RPV- LA) formulation: Pharmacokinetics (PK) in Plasma, Genital Tract (GT) in HIV-negative Females and Rectum (RT) in Males; SSAT040
Reported by Jules Levin
ViiV is studying the combination of 2 long-acting drugs including rilpivirine long-acting & GSK744 long-acting that could be used for PrEP & ART, at ICAAC this update was presented, "GSK744 is currently in phase2b clinical trials in treatment-naive subjects; both oral & long-acting (LAP) formulations are under development:
ICAAC: Meta-analysis of Safety Data From 8 Clinical Studies With GSK1265744, an HIV Integrase Inhibitor, Dosed Orally or as Injection of Long-Acting Parenteral Nanosuspension (LAP) - (09/12/13)
ICAAC: Good Safety Profile With Long-Acting Integrase Inhibitor, GSK744 - Written by Mark Mascolini - (09/11/13)
At IAS this past summer at Kuala Lumpur this study was presented on this long-acting drug combination:
High Levels of Two Antiretrovirals With Monthly or Quarterly Injections in Healthy Volunteers: GSK744 LAP + TMC278 LA
http://www.natap.org/2013/IAS/IAS_63.htm
First study of repeat dose co-administration of GSK1265744 and TMC278 long-acting parenteral nanosuspensions: pharmacokinetics, safety, and tolerability in healthy adults
IAS 2013 Kuala Lumpur June 30-July 3. W. Spreen1, P. Williams2, D. Margolis1, S. Ford1, H. Crauwels2, Y. Lou1, E. Gould1, M. Stevens2, S. Piscitelli1. 1GlaxoSmithKline, Infectious Diseases Research & Development, Research Triangle Park, United States, 2Janssen Infectious Diseases BVBA, Beerse, Belgium
http://www.natap.org/2013/IAS/IAS_36.htm
INDUCTION/MAINTENANCE ORAL GSK744+Rilpivirine. ViiV is planning to conduct a similar study of induction/maintenance evaluating once a month injections (intramuscular) of long-acting GSK744+Rilpivirine following an induction with orally taken GSK744+abacavir/3TC+rilpivirine.
( http://clinicaltrials.gov/ct2/show/NCT01641809?term=1265744+maintenance&rank=1)
This is an ongoing study of GSK744+rilpivirine taken orally/daily as maintenance therapy after successful completion of an induction regimen of GSK744+NRTIs: The Induction Phase consists of a 24 week dose-ranging evaluation of GSK1265744 at blinded doses of 10 mg, 30 mg and 60 mg once-daily and a control arm of open-label efavirenz (EFV) 600 mg once daily. The background dual nucleoside reverse transcriptase inhibitor (NRTI) antiretroviral therapy (ART) for all arms will be either abacavir/lamivudine (ABC/3TC) or tenofovir/emtricitabine (TDF/FTC) as selected by the Investigator. Maintenance Phase: The background NRTIs will be discontinued and the subjects will continue their randomized dose of GSK1265744 in combination with rilpivirine (RPV) 25 mg once-daily for an additional 72 weeks. The Maintenance phase will evaluate the ability of this two drug ART regimen to maintain virologic suppression through Week 48, Week 72 and Week 96. Subjects randomized to the EFV arm will continue on their randomized regimen through Week 96.
At 2013 Clin Pharm Wkp:
Split Dosing Versus Single Dose of Long-Acting Injected Integrase Inhibitor - written by Mark Mascolini - (04/24/13)
At CROI 2013, this study was presented: "Long-acting parenteral formulation of GSK1265744 protects macaques against repeated intrarectal challenges with SHIV". 20th Conference on Retroviruses and Opportunistic Infections
- Long-Acting Integrase Inhibitor Shields Macaques From Anal Simian HIV
http://www.natap.org/2013/CROI/croi_10.htm
- PrEP GSK744 Integrase Administered Monthly Perhaps Quarterly Prevents HIV-Infection in Monkeys:
"Long-Acting Parenteral Formulation of GSK1265744 Protects Macques against Repeated Intrarectal Challenges with SHIV"
Chasity Andrews from the Aaron Diamond AIDS Research Center in NYC
http://www.natap.org/2013/CROI/croi_38.htm
At ICAAC last week in Denver the CDC presented a study of tenofovir used in an intravaginal ring in monkeys challenged with higher HIV levels following a presentation of a study in monkeys at CROI 2013. The CDC is planning next step studies:
ICAAC: ICAAC: Pigtailed Macaques Under High Doses of Depot Medroxyprogesterone are Protected from Multiple SHIV Exposures with a Tenofovir Disoproxil Fumarate Intravaginal Ring - (09/24/13)
ICAAC: Tenofovir Intravaginal Ring - (09/24/13)
Then yesterday Janssen announced -
"Janssen R&D Ireland (Janssen) announced today it has signed a license agreement with PATH for the early development of the human immunodeficiency virus type 1 (HIV-1) medicine rilpivirine in a long-acting injection (depot formulation) as potential pre-exposure prophylaxis (PrEP) against HIV infection."
Current ongoing study:
A Study to Evaluate Safety, Acceptability, Pharmacokinetics, and ex Vivo Pharmacodynamics of TMC278 Long Acting Formulation in HIV-1 Seronegative Participants
to look at multi-dose PK and ex-vivo PD in the development of the product for prevention. It should complete early next year. This is an open-label (all people know the identity of the intervention), multi-arm (more than one treatment group), dose-ranging study (clinical study where different doses of study medication are tested against each other) to evaluate the safety, acceptability, pharmacokinetics, and ex vivo pharmacodynamics of a single and multiple intramuscular injections of long acting TMC278 to human immunodeficiency virus type 1 (HIV-1) seronegative (having a negative serum reaction) male and female participants.
At the 2012 Clin Pharm Wkp:
1. Injected Rilpivirine for PrEP Stays in Plasma for 84 Days in Women and Men
"Pharmacokinetics of long-acting rilpivirine in plasma, genital tract and rectum of HIV-negative females and males administered a single 600 mg dose". 13th International Workshop on Clinical Pharmacology of HIV Therapy, April 16-18, 2012, Barcelona
http://www.natap.org/2012/pharm/Pharm_06.htm
2. Excerpt from Report written from this Clin Pharm Wkp for NATAP by Courtney Fletcher, Pharm.D-
Clinical Pharmacology at the 13th Workshop on Clinical Pharmacology of HIV Therapy - Courtney V. Fletcher, Pharm.D. - (05/14/12)






|
|
| |
| |
|
|
|