| |
2 New Studies - HIV Antibodies Advance the search for a cure/Supercharged antibodies fight HIV-related virus in monkeys
|
| |
| |
View the PDF here
View the PDF here
View the PDF here
'Next-generation' antibodies could complement current therapies.
Nature | News
Heidi Ledford
30 October 2013
Powerful antibodies against HIV show promise for bolstering antiviral therapies, studies in rhesus macaques suggest.
Two papers published online today in Nature1, 2 report the impact of antibody therapy on an HIV-related virus that is capable of infecting monkeys and was engineered to carry key portions of the genome of the human virus. Levels of that modified virus, called SHIV, plummeted after the macaques were infused with human antibodies isolated from HIV patients and selected for their potency against a broad array of HIV viruses.
"The results were profound and surprising," says Dan Barouch, a professor of medicine at the Beth Israel Deaconess Medical Center in Boston, Massachusetts, and an author in one of the studies. "It clearly calls for evaluation of these antibodies in clinical trials."
Indeed, both teams plan to move their research into human trials. It would not be the first time that scientists have tested antibodies against HIV in humans, but earlier attempts met with little success1.
Those attempts, however, were made with less potent antibodies. Since then, techniques have improved for harvesting powerful anti-HIV antibodies - called 'broadly neutralizing antibodies' - from unusual patients who are able to keep the virus in check without therapy.
Now, techniques have improved for harvesting powerful anti-HIV antibodies - called 'broadly neutralizing antibodies'. In 2010, several labs reported the isolation of a new breed of broadly neutralizing antibodies, rejuvenating the field and reviving the possibility of using the antibodies therapeutically.
"Most scientists would have been laughed out of the room for proposing these studies prior to 2010," says Gary Nabel, chief scientific officer at Sanofi, a pharmaceutical firm headquartered in Paris. "It's really almost staggering to see how quickly this whole problem has broken open."
Powerful cocktails
Barouch and his team tested a cocktail of three of the antibodies, infusing them into four SHIV-infected rhesus monkeys. Within a week, the virus was undetectable in all four monkeys and remained so for at least 84 days.
Subsequent tests that used exclusively one particularly potent component of the cocktail, called PGT121, yielded similar results. Altogether, therapuetic effects were seen in a total of 18 treated monkeys.
In the other study, virologist Malcolm Martin of the National Institutes of Health in Bethesda, Maryland, and his colleagues gave a combination of two other antibodies to macaques that tested positive for the virus but had not yet developed symptoms. The treatment drove SHIV levels below the detection threshold within ten days. The therapy however was less effective in macaques in advanced stages of the disease.
In both studies, viral levels almost always rebounded, and the researchers could not test the long-term effects of the therapy in the monkeys for fear of triggering an immune response against the human antibodies themselves. As a result, it is unclear whether the significant responses they saw can be maintained in the long term, notes Louis Picker, an immunologist at Oregon Health & Science University in Portland.
Antibody therapy also faces logistical hurdles. Whereas current antiretroviral therapies used to combat HIV can be taken orally, antibodies must be injected. Therapeutic antibodies also tend to be significantly more expensive to make than small-molecule drugs. "Whether they're going to be available for the developing world is questionable," says Picker.
Nevertheless, these antibodies could tackle the virus in ways that current therapies do not. Cocktails of antiretroviral drugs have revolutionized HIV therapy, turning a once-fatal diagnosis into a chronic disease, says Barouch. But these drugs only block active viral-replication machinery and cannot fight the virus that circulates free in the blood or hides in cells without replicating.
The ultimate goal - curing HIV infection - will probably require a variety of therapies, and antibody therapy may well be a key player among them, says Picker. "It's not going to be easy," he says. "But every tool we have to move closer to the goal helps."
----------------------------------
Nature | News & Views
HIV: Antibodies advance the search for a cure
· Louis J. Picker
· & Steven G. Deeks
Published online
30 October 2013
Efforts to make a prophylactic HIV vaccine have identified monoclonal antibodies that potently suppress viral replication. Studies in monkeys show that these reagents effectively treat HIV infection.
A major breakthrough from the HIV-vaccine research community in recent years was the isolation and characterization of novel antibodies from HIV-infected people that have the remarkable ability to efficiently neutralize most circulating HIV strains1, 2. The antibodies' mechanism of action involves the recognition and blockade of evolutionarily conserved, functionally crucial structures of the HIV viral envelope. These unusual antibodies have reinvigorated the effort to develop an antibody-based prophylactic HIV vaccine by defining effective human antibody responses to the virus and providing a 'map' for reverse engineering of vaccines that recapitulate these responses. Because these antibodies develop infrequently, emerge only after many years of HIV infection and are characterized by a high degree of mutation1, 2, this task is not likely to be quickly accomplished. But that does not necessarily mean that the clinical benefit of these antibodies is relegated to the distant future. On the contrary, two reports published on Nature's website today, by Barouch et al.3 and Shingai et al.4, demonstrate that combinations of such antibodies drastically reduce virus levels in chronically infected rhesus macaques, bolstering the hope that such therapies might be effective in humans.
The two groups of investigators examined the ability of several of these potent and broadly HIV-neutralizing human monoclonal antibodies (mAbs) to suppress viraemia - the presence of the virus in the bloodstream - in rhesus macaques chronically infected with pathogenic (AIDS-causing) simian-human immunodeficiency virus (SHIV) hybrids that use the HIV envelope for cell entry. Their findings were remarkably consistent: administration of cocktails of two or more mAbs (directed at two different structural regions, the CD4-binding site and V3-loop/N332 glycans) profoundly suppressed viraemia, often to below the level of detection. This suppression persisted for weeks, as long as mAb levels were maintained. Furthermore, mutations associated with resistance to the neutralization activity of the administered mAbs were not generally observed in the virus that rebounded after mAb levels waned. Remarkably, Barouch and colleagues show that one antibody, the extraordinarily potent N332-glycan-dependent PGT121 mAb5, was able to provide prolonged viral suppression when used alone.
Given that current antiretroviral therapy (ART), which is based on small molecules that target different phases of the HIV life cycle, are effective and can be given orally, whereas mAbs need to be delivered by injection, why is this approach of interest? The answer lies in the different mechanisms of viral suppression by conventional ART versus neutralizing mAbs. Current ART drugs prevent the virus from spreading from an infected cell to the next round of target cells, but they do not directly kill infected cells nor inhibit the cells' production of virus particles6 (Fig. 1a). In the absence of viral spread, virus production declines as infected cells die by virus- or immune-mediated killing or by programmed cell death. But some infected cells persist and produce virus particles indefinitely. This rare but enduring reservoir of virus-producing cells is the source of viral rebound when people stop ART, and probably contributes to persistent inflammation and immune dysfunction (and associated morbidities) in individuals on ART7, 8.
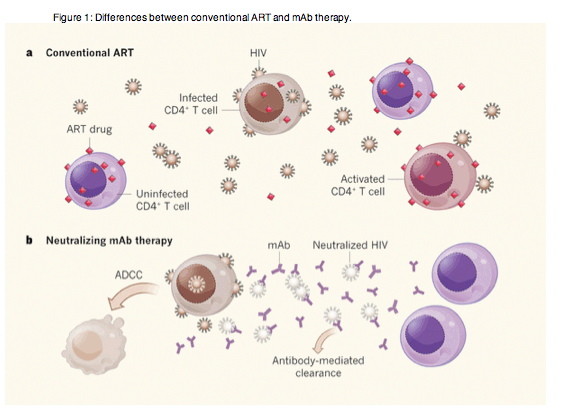
a, Conventional antiretroviral therapy (ART) involves combinations of small-molecule drugs that act at various cellular sites, protecting uninfected target CD4+ T cells from HIV infection by inhibiting viral entry or post-entry viral function. But this treatment does not affect the production of virus by infected cells, nor facilitate the clearance of virus particles. Persistent virus-producing cells and circulating virus can reignite progressive infection if ART is discontinued. Free virus can also activate cells, contributing to a chronic inflammatory environment. b, Monoclonal antibodies (mAbs) that bind to and inactivate viral envelope proteins effectively neutralize the virus and therefore also prevent the spread of infection. These may have the added advantage of facilitating the clearance of virus particles and killing virus-producing cells by antibody-dependent mechanisms, in particular antibody-dependent cell-mediated cytotoxicity (ADCC), thereby potentially diminishing the viral reservoir and preventing local immune activation.
Treatment with neutralizing mAbs would, like conventional ART, prevent viral spread, and this undoubtedly accounts for much of the antibodies' observed activity in these two studies. However, mAbs have intrinsic effector functions that would not only facilitate rapid clearance of virus particles produced by residually infected cells, but might also accelerate the destruction of virus-producing cells by antibody-dependent cell-mediated cytotoxicity, complement lysis or phagocytosis (Fig. 1b). Although these processes were not directly demonstrated in either of the new studies, Barouch et al. show accelerated viral clearance in mAb-treated macaques compared with that seen in humans given conventional ART. They also provide evidence for declining levels of cell-associated HIV DNA (a measurement of the number of infected cells) and improved function of SHIV-specific CD8+ T cells (antiviral immune cells) in mAb-treated animals. Perhaps more interestingly, Barouch and colleagues report that the viral-load set points - the stable level of viraemia reached once a balance is established between viral replication and the immune system - of the animals after therapy cessation were consistently lower than before treatment, suggesting that mAb treatment enhanced the antiviral immune response.
There are several caveats to this exciting work that deserve mention. One is the substantial animal-to-animal variability in pharmacokinetic outcomes, which would have to be addressed if long-term administration of these antibodies were pursued in people. Another is the fact that the high mutability of HIV means that slightly different envelope proteins will be expressed by virus strains both within an individual and across populations, making it likely that some people will harbour viruses that are resistant to one or more of the therapeutic mAbs. In addition, whether the mAbs can cross the blood-brain barrier and suppress viral replication in the central nervous system remains an open question. Finally, the extent to which viral envelope protein is exposed on the surface of long-lived infected cells is unknown. Additional treatment with virus-activating agents will probably be needed for antibody-mediated destruction to have a meaningful impact on this reservoir.
Despite these limitations, the findings of these two papers could revolutionize efforts to cure HIV. The combination of conventional ART and neutralizing mAb therapy might reduce viral replication more effectively than conventional ART alone, mediate destruction of virus-producing cells arising from latent infection, diminish deleterious, generalized immune activation, and facilitate the efficacy of T-cell-targeted therapeutic vaccines intended to destroy latent cells8. At the very least, these results will catalyse collaborations between the massive teams of experts who have for decades worked on HIV prevention and treatment in separate venues. As previously emphasized9, 10, true multidisciplinary science will be needed if we are to generate a cure for HIV.
|
|
| |
| |
|
|
|