| |
HCV Could be a "Rare Disease" by 2026, "1-time HCV screening for all/test & treat"..... need for more aggressive screening strategies and higher treatment capacity.....1-time universal screening - new study projects
|
| |
| |
Download the PDF here
Download the PDF here
an Annals of Internal Medicine study [published today-full text below] shows what society could get for its billions. Widespread treatment could push hepatitis cases into the rare-disease realm, or one in about 1,500 patients. It could prevent 79,000 cases of liver cancer, more than 124,000 cases of cirrhosis and 126,500 deaths from hepatitis complications.
"Hepatitis C virus infection could become a rare disease in the United States by 2036......with ongoing therapeutic advancements and screening policy changes, HCV infection could become a rare disease within the next [13 to] 22 years......."Those are certainly reasonable predictions. I don't think they're overstating the situation at all," said Dr. Eugene Schiff, director of the Schiff Center for Liver Diseases at the University of Miami Miller School of Medicine......."Now we're moving toward an era of 'test and treat,'" Schiff said. "Cure rates are approaching 100 percent with these new regimens, and they're very well-tolerated."
.....more aggressive [screening] recommendations should be proposed in conjunction with an increase in the treatment capacity for this condition.......In the ideal scenario, HCV infection could become a rare disease by 2026 (that is, 10 years earlier than in the base-case scenario).....1-time universal screening could identify 933 700 cases of HCV infection in the next 10 years......Our study underscores the need for more aggressive screening strategies and higher treatment capacity to further reduce the burden of HCV infection......Our study underscores the need for more aggressive screening strategies and higher treatment capacity to further reduce the burden of HCV infection. Birth-cohort screening would decrease this burden but would fail to identify a large pool of existing patients with HCV infection who could advance to severe disease stages without treatment. In addition, the number of patients who are able to receive treatment greatly affects the potential disease burden. This number depends on treatment capacity, availability of new drugs, treatment cost, and insurance coverage.......
......With the launch of all-oral drugs that can simplify treatment, primary care physicians or infectious disease specialists also may take on the role of treating patients with HCV infection, thus alleviating the burden on hematologists.......
U.S. Federal Funding for HIV/AIDS: The President's FY 2015 Budget Request.....
http://kff.org/global-health-policy/fact-sheet/u-s-federal-funding-for-hivaids-the-presidents-fy-2015-budget-request/......President Obama's Fiscal Year (FY) 2015 federal budget request, released on March 4, 2014, includes an estimated $30.4 billion for combined domestic and global HIV efforts.1 Domestic HIV is funded at $24.2 billion and global at $6.2 billion in the request.2 The FY 2015 request represents a 2.3% increase ($685 million) over FY 2014 levels, which totaled $29.7 billion. .....Federal funding for HIV has increased significantly over the course of the epidemic, including by $5.4 billion (or 22%) since FY 2009.....Federal funding for HIV, however, represents just a small fraction (<1%) of the overall federal budget of the United States.......The federal HIV budget is generally organized into five broad categories: care & treatment; cash & housing assistance; prevention; research; and global/international. The first four categories are for domestic programs only. More than half (57%) of the FY 2015 request is for care and treatment programs in the U.S.; 10% is for domestic cash/housing assistance; 3% is for domestic HIV prevention; 9% is for domestic HIV research; and 20% is for the global epidemic, including funding for international research (See Figure 2).
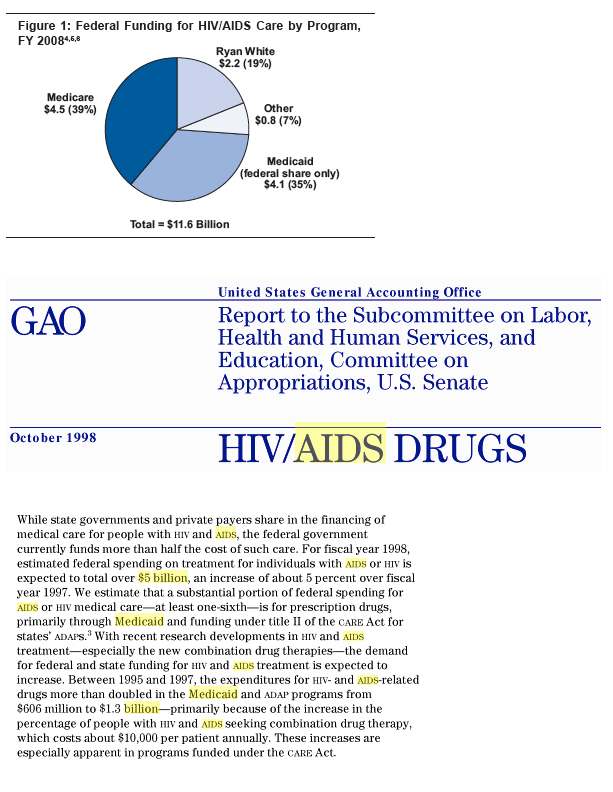
Medicaid spends over $5 billion a year on HIV/AIDS......Between 1997 and 2003 the number of Medicare beneficiaries with HIV increased by 80% (from 42,520 to 76,500), and rose more rapidly among women than men......In FY 2008, Medicare spending on HIV totaled $4.5 billion, representing 39 percent of federal spending on HIV care (Figure 1); it is expected to reach $4.8 billion in FY 2009.4,5,6 HIV spending under Medicare has increased over time, as the number of beneficiaries has grown. Still, Medicare spending on HIV represents less than 1% of total Medicare spending.7
----------------------
{......The actuarial firm Milliman [http://www.natap.org/2014/HCV/080114_01.htm]forecasts a federal spending increase somewhere in the range of $2.9 billion to $5.8 billion in 2015 at the hands of hepatitis C high-flyers, it said in a recent report commissioned by U.S. payers. And as a result, Part D premiums could rise by as much as 8.6%....."The study shows taxpayers will bear the lion's share of the increased drug costs for Medicare Part D beneficiaries taking hepatitis C drugs.......Average monthly premiums for Medicare PDPs are projected to increase 4.3 percent to 8.6 percent next year as a result of new hepatitis C drugs, an increase of $17 to $33 per beneficiary per year."........A recent JAMA commentary, written by two CVS Caremark execs, predicted that HCV patients could add $200 to $300 per year to every insured American's health insurance premium for each of the next 5 years.......Medicaid programs have demanded additional state funding to cover Sovaldi's cost.....The treatment cures 95% of all patients, saving the cost of further therapy--like liver replacements, in some cases--down the line......."Over time, these new medicines can transform the face of hep C in the U.S. by dramatically reducing the sickness and death associated with the disease," the California Healthcare Institute, a pharma-backed group, wrote in a recent report. "Treating the U.S. hep C population with these new drugs, as opposed to the previous standard of care, could save the lives of thousands of people.}.......UnitedHealthCare's PBM Optum says in a report they issued the new HCV are are cost-effective http://www.natap.org/2014/HCV/052914_01.htm
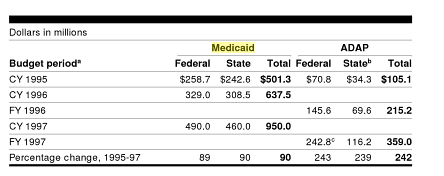
............The study finds that the majority of new Federal spending will come from increased federal reinsurance subsidies. Medicare payments for Part D catastrophic coverage have grown considerably faster than other components of Part D spending. According to the Medicare Payment Advisory Commission, between 2007 and 2012, payments have increased at an average annual rate of 14 percent while nearly doubling from $8 billion to $15.6 billion.
The most expensive drug in the world is Alexion's Soliris, which is used to treat a rare form of anemia and also a rare kidney disorder. Alexion charges $440,000/year per patient. This price is really not related to the R&D costs needed to bring this drug to the market. Yet, private insurers and national health agencies in Europe willingly pay for this drug. Why? Because the costs of caring for patients with these conditions can run into millions each year. Soliris, even at this high price, actually saves the healthcare system money because using it results in dramatic decreases in other healthcare system expenses generated by these patients.
---------------------
.....more aggressive [screening] recommendations should be proposed in conjunction with an increase in the treatment capacity for this condition.......In the ideal scenario, HCV infection could become a rare disease by 2026 (that is, 10 years earlier than in the base-case scenario).....1-time universal screening could identify 933 700 cases of HCV infection in the next 10 years......Our study underscores the need for more aggressive screening strategies and higher treatment capacity to further reduce the burden of HCV infection......Our study underscores the need for more aggressive screening strategies and higher treatment capacity to further reduce the burden of HCV infection. Birth-cohort screening would decrease this burden but would fail to identify a large pool of existing patients with HCV infection who could advance to severe disease stages without treatment. In addition, the number of patients who are able to receive treatment greatly affects the potential disease burden. This number depends on treatment capacity, availability of new drugs, treatment cost, and insurance coverage.......
......With the launch of all-oral drugs that can simplify treatment, primary care physicians or infectious disease specialists also may take on the role of treating patients with HCV infection, thus alleviating the burden on hematologists.......
.....Objective of study: To predict the effect of new therapies and screening on chronic HCV infection and associated disease outcomes.......Our model estimated that 2.3 million persons were chronically infected with HCV in the beginning of 2013 compared with 3.2 million persons in 2001.....Institutionalized patients with HCV infection were excluded......We developed an individual-level state-transition model (18) that simulated the HCV-infected population of the United States from 2001 to 2050. We used a nationally representative distribution of patient age, sex, awareness of HCV infection status, HCV genotype, disease stage, and treatment history using data from NHANES (National Health and Nutrition Examination Survey) 1999-2002 and published clinical studies.....Prevalence decreased mainly because of deaths and successful treatments in this cohort. Also, our model projected that fewer patients are eligible for birth-cohort screening than estimated in a previously published study.....
.....From 2001 to 2013, a total of 157 300 HCV-infected persons died of liver-related complications, 415 000 died of other causes, and 589 100 achieved SVR. During the same period, 251 000 new persons became chronically infected with HCV. Considering the population growth in the United States (50), we projected that HCV infection would become a rare disease by 2036 (that is, it would affect approximately 1 in 1500 persons, or less.) (51)......
.......One-time birth-cohort screening beginning in 2013 is expected to identify 487 000 cases of HCV infection in the next 10 years. In contrast, 1-time universal screening could identify 933 700 cases. With the availability of highly effective therapies, HCV infection could become a rare disease in the next 22 years. Recently approved therapies for HCV infection and 1-time birth-cohort screening could prevent approximately 124 200 cases of decompensated cirrhosis, 78 800 cases of hepatocellular carcinoma, 126 500 liver-related deaths, and 9900 liver transplantations by 2050. Increasing the treatment capacity would further reduce the burden of HCV disease.....In the base-case scenario, our model projected that the prevalence of DC, HCC, and liver-related deaths will peak during 2019 to 2020 and start declining afterward (Figure 3)."
.......In the ideal scenario, HCV infection could become a rare disease by 2026 (that is, 10 years earlier than in the base-case scenario) (Figure 2). The implementation of 1-time universal screening could identify 933 700 cases of HCV infection in the next 10 years. Compared with the base-case scenario (current clinical practice), the ideal scenario could reduce the total number of cases of DC by 135 800 (46%), cases of HCC by 96 300 (40%), liver-related deaths by 161 500 (37%), and liver transplantations by 13 900 (37%) during 2014 to 2050 (Table 3)."
---------------------
The Changing Burden of Hepatitis C Virus Infection in the United States: Model-Based Predictions
Ann Intern Med. Aug 5 2014
Mina Kabiri, MS; Alison B. Jazwinski, MD; Mark S. Roberts, MD; Andrew J. Schaefer, PhD; and Jagpreet Chhatwal, PhD
2 pdfs attached for download above
Abstract
Background: Chronic hepatitis C virus (HCV) infection causes a substantial health and economic burden in the United States. With the availability of direct-acting antiviral agents, recently approved therapies and those under development, and 1-time birth-cohort screening, the burden of this disease is expected to decrease.
Objective: To predict the effect of new therapies and screening on chronic HCV infection and associated disease outcomes.
Design: Individual-level state-transition model.
Setting:Existing and anticipated therapies and screening for HCV infection in the United States.
Patients: Total HCV-infected population in the United States.
Measurements: The number of cases of chronic HCV infection and outcomes of advanced-stage HCV infection.
Results: The number of cases of chronic HCV infection decreased from 3.2 million in 2001 to 2.3 million in 2013. One-time birth-cohort screening beginning in 2013 is expected to identify 487 000 cases of HCV infection in the next 10 years. In contrast, 1-time universal screening could identify 933 700 cases.
With the availability of highly effective therapies, HCV infection could become a rare disease in the next 22 years. Recently approved therapies for HCV infection and 1-time birth-cohort screening could prevent approximately 124 200 cases of decompensated cirrhosis, 78 800 cases of hepatocellular carcinoma, 126 500 liver-related deaths, and 9900 liver transplantations by 2050. Increasing the treatment capacity would further reduce the burden of HCV disease.
Limitation: Institutionalized patients with HCV infection were excluded, and empirical data on the effectiveness of future therapies and on the future annual incidence and treatment capacity of HCV infection are lacking.
Conclusion: New therapies for HCV infection and widespread implementation of screening and treatment will play an important role in reducing the burden of HCV disease. More aggressive screening recommendations are needed to identify a large pool of infected patients.
Primary Funding Source: National Institutes of Health.
---------------------------
Editors' Notes
Context Strategies for screening and treating hepatitis C virus (HCV) infection are rapidly evolving.
Contribution A model was developed that shows dramatic declines in the prevalence of HCV infection and the incidence of advanced-stage outcomes, such as cirrhosis, hepatocellular carcinoma, liver transplantations, and liver-related deaths, with the implementation of current screening policies and use of newly developed antiviral therapies. Adoption of 1-time universal screening and improved treatment capacity would add to declines in these outcomes.
Caution Estimates about the long-term efficacy and toxicity of recently identified antiviral drugs for HCV infection used in the model are uncertain.
Implication Hepatitis C virus infection could become a rare disease in the United States by 2036
-The Editors
---------------------------
Chronic hepatitis C virus (HCV) infection is a major health problem in the United States, affecting 3.2 million persons (1). This condition is the leading cause of chronic liver disease and hepatocellular carcinoma (HCC) and the leading indication for liver transplantation in the United States (2). The number of deaths from HCV infection in the United States surpassed that from HIV infection in 2007 (3). In 2011, the estimated economic burden associated with chronic HCV infection in the United States was $6.5 billion (4).
Treatment of HCV infection has rapidly evolved over the past 2 decades. The launch of direct-acting antivirals (DAAs) in 2011 and recent availability of the first all-oral regimens for HCV infection represent an important shift in the treatment paradigm for this condition (5). The sustained virologic response (SVR) rates for certain patients increased to 97% (6). New treatments under investigation have shown potential to further increase response rates, decrease treatment duration, and improve adverse effect profiles. These therapies are being studied as combinations of DAAs, with and without ribavirin and interferon (7-8).
In addition to advances in treatment, key changes in screening recommendations for HCV infection have taken place. The Centers for Disease Control and Prevention and the U.S. Preventive Services Task Force expanded their screening recommendations for HCV infection to include 1-time screening for anyone born between 1945 and 1965 (9-10). Modeling studies have shown that this strategy can be cost-effective and can reduce the burden of HCV disease (11-13). Finally, the Patient Protection and Affordable Care Act might facilitate the implementation of recommended screening strategies for HCV infection and linkage to care and treatment (14).
The launch of DAA therapies and the new screening recommendations are collectively expected to substantially reduce the burden of HCV infection in the United States; however, the effect of these changes has not yet been quantified. Previous studies did not project the burden of infection under these changing dynamics but instead limited the analyses to the old standard of care-peginterferon and ribavirin (PEG-RBV) without screening for HCV infection (4, 15)-or evaluated only the cost-effectiveness of screening without projecting the changing burden of infection (11, 16-17). The effect of limited treatment capacity on the burden of infection also has not been studied. Our objective was to project the burden of HCV disease in the United States by considering recent therapeutic advances, treatment capacity, and the implementation of 1-time birth-cohort or universal screening.
Methods
Characterization of the HCV-Infected Population
We developed an individual-level state-transition model (18) that simulated the HCV-infected population of the United States from 2001 to 2050. We used a nationally representative distribution of patient age, sex, awareness of HCV infection status, HCV genotype, disease stage, and treatment history using data from NHANES (National Health and Nutrition Examination Survey) 1999-2002 and published clinical studies (Table 1 of the Supplement) (11, 15, 19-22). We added new HCV infections to the model on the basis of the annual number of new HCV infections reported by the Centers for Disease Control and Prevention (Table 2 of the Supplement) (23). Each newly infected patient was added as an acute case that could progress to the chronic phase (19).
Patients could become aware of their HCV infection status in the course of disease progression (Table 3 of the Supplement). At any given time, patients occupied one of the health states (Figure 1) and could transition to another state with a predefined probability depending on their current state (Table 4 of the Supplement).
Natural History of HCV Infection
The chronic phase of the infection was defined using the Meta-analysis of Histologic Data in Viral Hepatitis (METAVIR) scoring system: F0 indicates no fibrosis of the liver, F1 indicates portal fibrosis without septa, F2 indicates portal fibrosis with few septa, F3 indicates many septa without cirrhosis, and F4 indicates cirrhosis. Patients could progress to decompensated cirrhosis (DC) or HCC, receive a liver transplant, or die of liver-related complications (Figure 1). The model assumed an age limit on liver transplantation of 75 years (24). Table 1 of the Supplement shows all probabilities of disease progression. Patients who achieved SVR in F0 to F3 states were assumed to be cured of HCV infection; however, those who achieved SVR in an F4 state could progress to DC, HCC, or both but at a slower rate than HCV-infected patients.
Simulation Scenario: Current Clinical Practice
We simulated the current clinical practice as our base case, that is, 1-time birth-cohort screening for HCV infection starting in 2013 and treatment with PEG-RBV or protease inhibitor (PI)-based triple therapy before 2014, sofosbuvir- and simeprevir-based therapies starting in 2014, and future drugs as they become available. We implemented 1-time birth-cohort screening for HCV infection in persons born between 1945 and 1965 that detected prevalent cases unaware of their infection status. We also included risk-based screening in this scenario.
We assumed that 91% of these patients would accept screening and 90% of those who tested positive would receive their results (11). We assigned the uptake of screening such that most of these patients would gradually receive screening during 5 years beginning in 2013. We estimated that 80% of the patients aware of their HCV infection status would initiate treatment (11, 25-26). We assigned treatment regimens on the basis of patients' treatment history, HCV genotype, and contraindication to interferon and the standard of care at the time of treatment.
For patients with genotype 1 HCV, we assigned PEG-RBV during 2001 to 2011, followed by a combination of a first-generation PI (boceprevir or telaprevir) and PEG-RBV in 2012 to 2013. For patients with non-genotype 1 HCV, we assigned PEG-RBV during the entire period of 2001 to 2013. We assumed that patients in whom PEG-RBV treatment failed could be re-treated once more at the most with PEG-RBV or PI-based therapy. We also assumed that patients in whom PI-based therapy failed were not eligible for re-treatment with a drug from the same class.
Based on recently published evidence, higher treatment response rates would be expected in all patients after 2013 because of the availability of new therapies, albeit at different intervals (27-40). Therefore, we assumed that these therapies could be divided into 2 major waves on the basis of therapy availability, cure rates, and target populations (Table 1). We also assumed that during 2012 to 2013, 75% of the eligible patients with mild fibrosis (F0 to F2) and 25% of the eligible patients with bridging fibrosis (F3) waited for newer therapies (41).
We assumed that wave 1 of new treatments would start in 2014 and that, with wave 1, the SVR rates would increase to 90% in the groups of patients with genotypes 1 to 6 HCV without cirrhosis (Table 1). Although the reported SVR rates were as high as 97% in some patients, we used a conservative estimate of 90% in certain patients to account for lower SVR rates in real life (42). The first wave also included therapies for patients with genotypes 1 to 6 HCV who had cirrhosis, but we assumed that the response rates among these patients would remain suboptimal (Figure 1 of the Supplement) (43).
We assumed that the second wave of treatment would begin in 2017 and increase the response rates to 90% in all patients. We included re-treatment with wave 1 or 2 therapies of patients in whom PEG-RBV or PI-based therapy failed before 2014. Table 1 and the Supplement show the SVR rates by treatment history, genotype, fibrosis stage, and interferon contraindication. Figure 1 of the Supplement shows the treatment used for each category of patients at different intervals.
Because treating all HCV-infected patients within a year is impracticable, we introduced an annual constraint on the number of persons who could access treatment. Our rationale was to model the effect of limited treatment uptake and limited resources (for example, budget and number of physicians) available to treat all eligible patients. For our base case, we used historical data to determine the national treatment uptake (44) and did sensitivity analyses.
Simulation Scenario: Ideal Case
We simulated the effect of a hypothetically ideal scenario that represented an upper limit of the benefits that ongoing advancements in therapies and policy-level changes could achieve. We simulated the best possible combination of 1-time universal screening in all adults, adoption of new drugs as they become available, and unlimited treatment capacity. We distributed the uptake of screening proportionally over a 5-year period beginning in 2013.
Simulation Scenario: Pre-DAAs and Natural History
For the purpose of estimating the incremental benefits of therapeutic advancements and policy-level changes, we simulated 2 comparator scenarios: a pre-DAA scenario and a natural-history scenario. The pre-DAA scenario represented screening and treatment practice until the launch of DAAs. It simulated treatment of HCV infection with PEG-RBV only, from 2001 onward, solely with risk-based screening. The natural-history scenario simulated the burden of HCV infection with no screening and no treatment. Table 2 summarizes the 4 clinical scenarios that were simulated.
Data Synthesis and Analysis
Model Outcomes
We projected the prevalence of HCV infection from 2001 to 2050. We also projected the prevalence and incidence of early stages of HCV infection (fibrosis states F0 to F4), advanced stages of disease, DC and HCC, and the number of liver transplantations and liver-related deaths.
Model Validation
Using the model outcomes from 2001 to 2013, we validated our model with several published studies. First, we compared the predicted prevalence of HCV infection with that from a recently published NHANES 2003-2010 (45). Second, we compared the predicted incidence and prevalence by stages of HCV disease with those from published studies and reports from the Centers for Disease Control and Prevention (15, 22, 46-48). Third, we compared the natural history of HCV infection according to our model with that from a multicenter follow-up study of patients with advanced fibrosis (49). Finally, we cross-validated our model with earlier modeling studies (4, 15) by comparing the results of the natural-history and pre-DAA scenarios.
Sensitivity Analyses
We tested the effect of the SVR rates, the timing of the availability of future therapies, treatment capacity, patients' decision to wait for new drugs, and changing annual incidence of HCV infection on the burden of HCV disease. We did deterministic sensitivity analyses on the natural-history parameters of HCV infection and patient characteristics (Table 4 and Table 5 of the Supplement). We also evaluated the effect of treatment capacity on the burden of HCV infection (Table 6 of the Supplement).
Role of the Funding Source
The study was supported by the National Center for Advancing Translational Sciences of the National Institutes of Health. The funding source played no role in study design, conduct, or analysis or the decision to submit the manuscript for publication.
Results
Validation
Our model projected that the average number of cases of chronic HCV infection in 2003 to 2010 was 2.7 million, which is equal to the reported values in NHANES 2003-2010 (45) (Table 7 of the Supplement). The projected average prevalence of HCC in 2001 to 2004 was within 3% of the reported values (46). The incidence of HCC and liver-related deaths in 2005 was within 1% to 15% of the reported values (22). The projected distribution of different stages of chronic HCV infection closely matched that of another modeling study (15). Finally, our model's 10-year cumulative incidence rates of DC, HCC, and combined liver-related deaths and liver transplantations closely matched those of a recently published multicenter follow-up study (Table 8 of the Supplement) (49).
Burden of HCV Infection
Our model projected that the cases of chronic HCV infection in the United States decreased from 3.2 million in 2001 to 2.3 million in 2013 (Figure 2). From 2001 to 2013, a total of 157 300 HCV-infected persons died of liver-related complications, 415 000 died of other causes, and 589 100 achieved SVR. During the same period, 251 000 new persons became chronically infected with HCV. Considering the population growth in the United States (50), we projected that HCV infection would become a rare disease by 2036 (that is, it would affect approximately 1 in 1500 persons, or less.) (51).
Ideal Scenario
In the ideal scenario, HCV infection could become a rare disease by 2026 (that is, 10 years earlier than in the base-case scenario) (Figure 2). The implementation of 1-time universal screening could identify 933 700 cases of HCV infection in the next 10 years. Compared with the base-case scenario (current clinical practice), the ideal scenario could reduce the total number of cases of DC by 135 800 (46%), cases of HCC by 96 300 (40%), liver-related deaths by 161 500 (37%), and liver transplantations by 13 900 (37%) during 2014 to 2050 (Table 3).
Pre-DAA Scenario
In the pre-DAA scenario, HCV infection did not become a rare disease. Compared with the base-case scenario, the pre-DAA scenario would have increased the number of cases of DC by 124 200 (30%), cases of HCC by 78 700 (25%), liver-related deaths by 126 500 (23%), and liver transplantations by 9900 (21%) during 2014 to 2050 (Table 3).
Sensitivity Analyses
We evaluated the effect of increased treatment capacity on disease burden (Table 6 of the Supplement). Compared with the base-case scenario, a 10% increase in treatment capacity in 2012 and a 50% increase beyond 2014 (scenario 1) would reduce the number of cases of DC and HCC, liver-related deaths, and liver transplantations by 9% to 14%; a 20% decrease in treatment capacity beyond 2014 (scenario 2) would increase the corresponding adverse outcomes by 16% to 22%. Compared with the base-case scenario, unlimited treatment capacity from 2014 onward (scenario 3) would prevent 128 800 cases of DC, 91 000 cases of HCC, 153 200 liver-related deaths, and 13 400 liver transplantations.
When we reduced the SVR rates of available and future drugs by 10%, the cumulative incidence of DC and HCC and the number of liver-related deaths and liver transplantations increased by 4% to 23%, depending on the simulation scenario (Table 9 of the Supplement). Delayed or early launch of the second wave of therapies for HCV infection did not substantially change the disease burden (Table 10 of the Supplement). In addition, we found that the results were not sensitive to the percentage of patients in states F0 to F3 who might choose to wait for future therapies instead of initiating treatment with PI-based therapies (Table 11 of the Supplement). Among the natural-history parameters, the probability of developing DC and HCC in patients with cirrhosis had the greatest effect on the disease burden.
We also did a sensitivity analysis on the prevalence of HCV infection. Assuming that 4.9 million persons were infected with HCV in 2001, which was the upper limit of the 95% CI of the NHANES 1999-2002 estimate (19), we determined that the cumulative incidence of DC, HCC, and liver-related deaths increased by 23% to 25% compared with that of the base-case scenario (Table 12 and Table 13 of the Supplement). Finally, we evaluated the impact of decreasing and increasing annual incidence of HCV infection and found no substantial effect on the outcomes (Table 14 of the Supplement).
Discussion
Our model estimated that 2.3 million persons were chronically infected with HCV in the beginning of 2013 compared with 3.2 million persons in 2001. With the implementation of birth-cohort screening and the availability of highly effective new therapies, HCV infection could become a rare disease by 2036. In addition, these changes could substantially decrease the overall clinical burden associated with HCV infection in the United States.
Our study also identified trends in HCV disease burden that have not been previously reported. We estimated that the current number of chronic cases in the United States is actually lower than the commonly reported estimate of 3.2 million, as corroborated by recently published data from NHANES 2003-2010. Prevalence decreased mainly because of deaths and successful treatments in this cohort. Also, our model projected that fewer patients are eligible for birth-cohort screening than estimated in a previously published study (11). Our results differed because we accounted for the possibility that patients in the birth cohort progressed beyond cirrhosis or became aware of their disease before the implementation of screening in 2013.
Our study underscores the need for more aggressive screening strategies and higher treatment capacity to further reduce the burden of HCV infection. Birth-cohort screening would decrease this burden but would fail to identify a large pool of existing patients with HCV infection who could advance to severe disease stages without treatment. In addition, the number of patients who are able to receive treatment greatly affects the potential disease burden. This number depends on treatment capacity, availability of new drugs, treatment cost, and insurance coverage.
With the launch of all-oral drugs that can simplify treatment, primary care physicians or infectious disease specialists also may take on the role of treating patients with HCV infection, thus alleviating the burden on hepatologists (52). Furthermore, such programs as the Extension for Community Healthcare Outcomes can help to increase the treatment capacity by improving access to care for underserved populations (53). However, the high price of new therapies could become a barrier to the timely treatment of HCV infection and inhibit the full potential of therapeutic advances and screening recommendations (54).
Our study has limitations. First, the historical number of cases of HCV infection in the model was based on data from NHANES 1999-2002, which underestimate the prevalence of HCV infection in the United States by excluding the institutionalized population. However, we tested the effect of higher prevalence of HCV infection on the future burden of this disease in a sensitivity analysis. Second, we estimated the total number of patients who received treatment from data on drug prescriptions reported by insurance companies (44), which may underestimate the number of patients who received treatment. Third, our model did not account for co-infections and other risk factors, such as alcohol consumption, that affect disease progression (55-56). These limitations may have resulted in an underestimation of the projected burden of HCV infection. Fourth, we do not consider the potential effect of treatment on disease transmission. Although improved treatment would be expected to decrease transmission of HCV infection, new cases of this disease are a small proportion of the number of existing cases.
Information about SVR rates and the launch time of new therapies is limited. We based our SVR rates on results from several phase 2 and 3 clinical studies, but real rates may differ. We also based our assumptions about the launch time of new therapies on the end dates of clinical trials. Finally, due to the lack of information on re-treatment of patients in whom recently approved and future therapies will fail, the analysis of the re-treatment of these patients is beyond the scope of our analysis.
In conclusion, we evaluated the effect of the availability of DAAs, recently approved and potential future therapies, and changes in screening recommendations for HCV infection on the future burden of HCV disease in the United States. We found that, with ongoing therapeutic advancements and screening policy changes, HCV infection could become a rare disease within the next 22 years. We also found that the current screening recommendations are helpful in decreasing the future burden, but more aggressive recommendations should be proposed in conjunction with an increase in the treatment capacity for this condition.
|
|
| |
| |
|
|
|