 |
 |
 |
| |
Dolutegravir Superior to Efavirenz at 144 Weeks in SINGLE Trial
|
| |
| |
ICAAC 2014. September 5-9, 2014. Washington, DC
Mark Mascolini
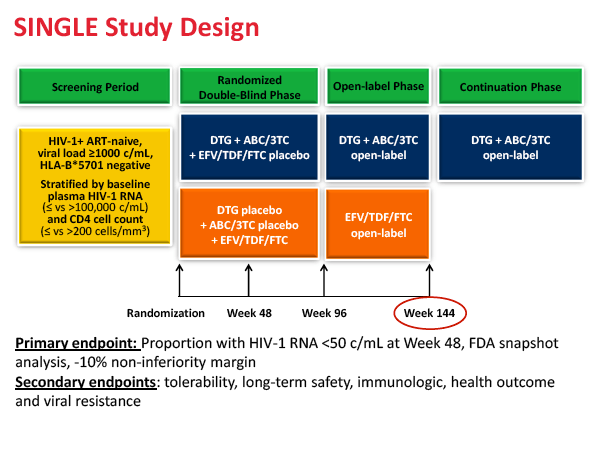
Dolutegravir plus abacavir/lamivudine maintained superiority over efavirenz plus tenofovir/emtricitabine through 144 weeks in previously untreated people in the SINGLE trial [1]. Published 48-week results of SINGLE established the virologic superiority of the once-daily dolutegravir regimen over once-daily Atripla [2]. Since this trial began, Triumeq, a single-tablet formulation of the integrase inhibitor with abacavir/lamivudine, has been licensed for first-line therapy in the United States and Europe.
SINGLE enrolled antiretroviral-naive people with a viral load at or above 1000 copies and randomized them to dolutegravir plus single-tablet abacavir/lamivudine or to Atripla. No one had genotypic evidence of resistance to antiretrovirals. This double-blind, double-dummy, phase 3 trial figured efficacy by the FDA snapshot analysis of people exposed to study drugs. The new analysis extends data from week 96 to 144, an open-label phase in which participants continued their assigned combination.
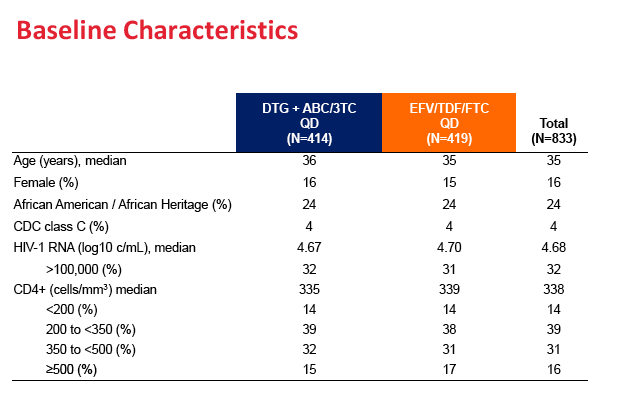
Among the 833 people who received study drug, 84% were men and 32% were nonwhite. Median age stood at 35 years. Before treatment began, median CD4 count measured 338 and median viral load 4.68 (about 48,000 copies). One third of study participants had a pretreatment load above 100,000 copies.
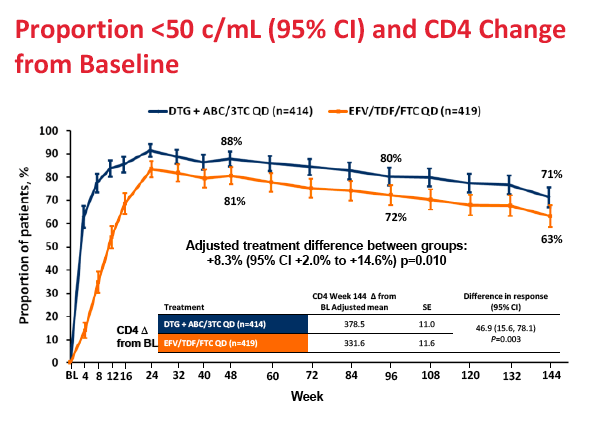
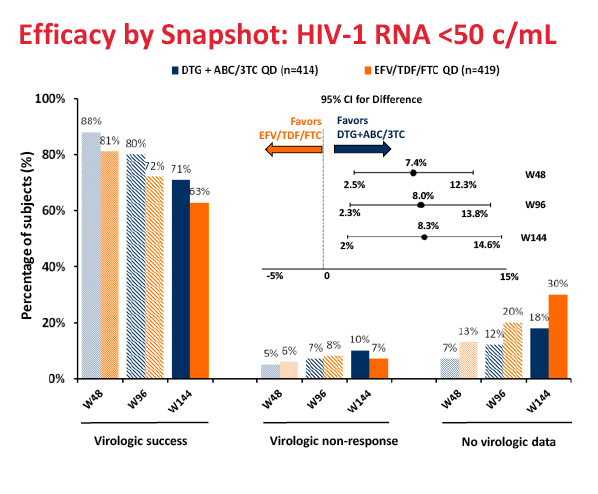
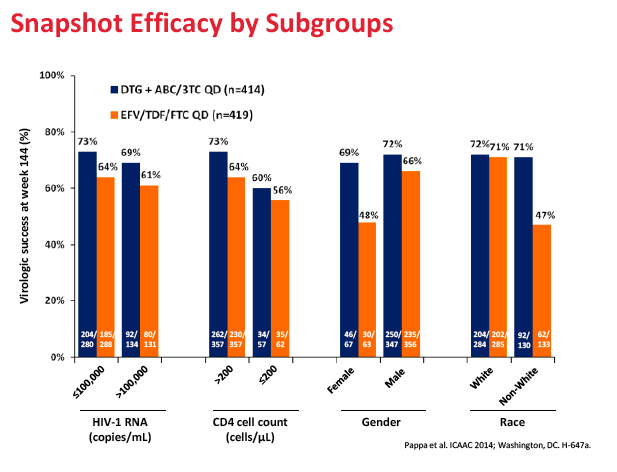
After 144 weeks, 71% randomized to the dolutegravir regimen and 63% randomized to Atripla had a viral load below 50 copies, a significant difference confirming the long-term superiority of the dolutegravir combination (difference 8.3%, 95% confidence interval 2.0 to 14.6, P = 0.010). Among people who began treatment with a viral load above 100,000 copies, 144-week response rates were 69% in the dolutegravir arm and 61% in the Atripla arm. Among women 69% assigned to dolutegravir and 48% assigned to Atripla had a week-144 viral load below 50 copies. Among nonwhites respective 144-week response rates were 71% and 47%.
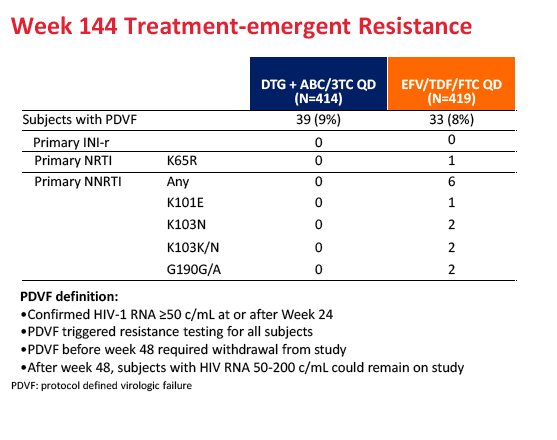
Protocol-defined virologic failure did not differ between treatment arms--9% with dolutegravir and 8% with Atripla. Among people genotyped after failure, no integrase inhibitor or nucleoside mutations could be detected in the dolutegravir arm, while nucleoside mutations could be detected in 1 person and nonnucleoside mutations in 6 people randomized to Atripla.
People randomized to the dolutegravir regimen gained more CD4 cells than those randomized to Atripla through 144 weeks (average 379 versus 332, P = 0.003), though this difference may not be clinically meaningful.
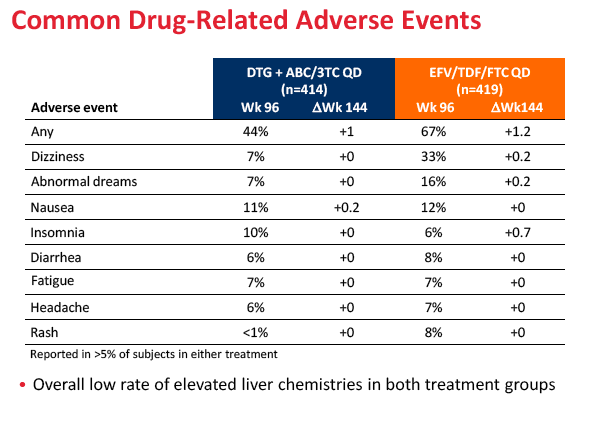
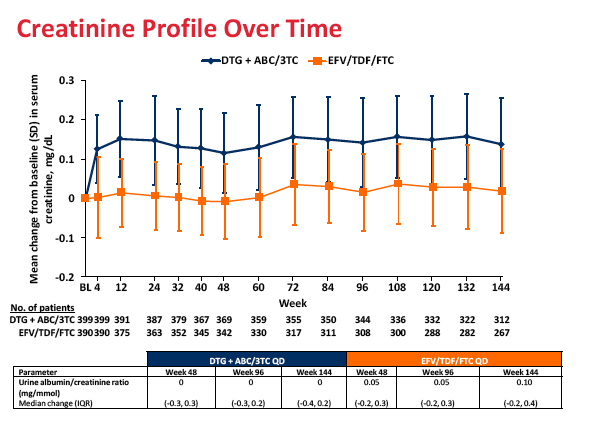
Sixteen people randomized to dolutegravir versus 58 randomized to Atripla withdrew from the study because of adverse events (4% versus 14%). Rates of psychiatric disorders (6% versus under 1%), nervous system disorders (4% versus under 1%), skin and subcutaneous tissue disorders (2% versus under 1%), and gastrointestinal disorders (2% versus 0) were higher with Atripla than with the dolutegravir combination. Urine protein-to-creatinine ratio did not change substantially through 144 weeks.
References
1. Pappa K, Baumgarten A, Felizarta F, et al. Dolutegravir + abacavir/lamivudine once daily superior to tenofovir/emtricitabine/efavirenz in treatment naive HIV subjects: 144-week results from SINGLE (ING114467). ICAAC 2014. September 5-9, 2014. Washington, DC. Abstract H-647a.
2. Walmsley SL, Antela A, Clumeck N, et al. Dolutegravir plus abacavir-lamivudine for the treatment of HIV-1 infection. N Engl J Med. 2013;369:1807-1818. http://www.nejm.org/doi/full/10.1056/NEJMoa1215541




|
| |
|
 |
 |
|
|