 |
 |
 |
| |
Tenofovir Nanoparticles in Vaginal Gel
Protect 10 of 10 BLT Mice From HIV Challenge
|
| |
| |
see full poster report below
ICAAC 2014. September 5-9, 2014. Washington, DC
Mark Mascolini
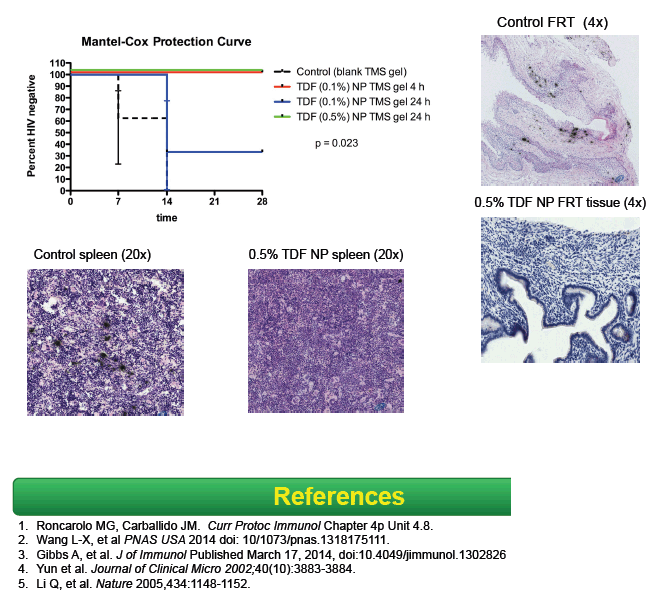
Tenofovir (TDF) nanoparticles suspended in a thermosensitive intravaginal gel protected 10 of 10 humanized BLT mice from HIV challenge for 4 weeks, while all 3 control animals who received a gel lacking TDF became infected [1].
Intravaginal 1% tenofovir gel applied before and after sex lowered HIV acquisition risk 39% in the CAPRISA 004 trial [2], but daily 1% tenofovir gel did not protect women in the VOICE trial [3]. Researchers at Creighton University and colleagues at other centers noted that vaginal gel leakage and poor tenofovir penetration of vaginal tissue may contribute to these inconsistent results [1]. To overcome these problems they suspended TDF nanoparticles in a thermosensitive gel that remains liquid at room temperature but becomes viscous at body temperature.
To test the TDF nanoparticles in thermosensitive gel, they randomly assigned 13 humanized BLT mice with good human immune reconstitution to receive TDF-loaded gel or a blank gel. Six mice received nanoparticles containing 0.1% weight/volume TDF, 4 received nanoparticles containing 0.5% weight/volume TDF, and 3 received blank gel. The TDF doses are equivalent to 1 and 5 mg/mL TDF. Gel volume was 30 uL.
Four hours and 24 hours after gel application, treated mice were challenged with two HIV-1 viruses, WITO.c/2474 and SUMA.c/2821, at a dose of 500,000 TCID50 each. Control mice were challenged with the same viruses 4 hours after application. The investigators withdrew plasma weekly for viral load testing for 4 weeks after HIV-1 challenge. Four weeks after challenge, they measured viral DNA in peripheral blood mononuclear cells (PBMCs). After sacrificing animals, they assessed HIV-1 p24 antigen in vaginal tissues.
All 3 control mice had detectable HIV RNA in plasma 1 to 2 weeks after viral challenge. None of the 10 treated mice had detectable HIV RNA in plasma through 4 weeks of follow-up (P < 0.02 versus control animals). Viral DNA remained undetectable in PBMCs of treated mice 4 weeks after HIV-1 challenge, and HIV-1 p24 could not be detected in vaginal tissues. HIV RNA could not be detected in mouse spleen.
Particle size of TDF nanoparticles averaged 68.2 +/- 2.9 nM and surface charge averaged -4.5 +/- 0.9 mV. TDF nanoparticle encapsulation efficiency determined by HPLC was 16.1% without isopropyl alcohol and 95.8% in the presence of isopropyl alcohol.
The researchers concluded that TDF nanoparticles suspended in a thermosensitive intravaginal gel offer "sustained and coitus-independent prevention from vaginal contraction of HIV-1 in a hu-BLT mouse model." They proposed that their findings "are the first step in assessing long-acting efficacy of TDF nanoparticles plus thermosensitive gel" for HIV prevention.
References
1. Destache CJ, Date AA, Zhe Y, et al. Topical application of tenofovir TDF nanoparticles prevents HIV-1 vaginal transmission in humanized-BLT mice. ICAAC 2014. September 5-9, 2014. Washington, DC. Abstract H-997.
2. Abdool Karim Q, Abdool Karim SS, Frohlich JA, et al. Effectiveness and safety of tenofovir gel, an antiretroviral microbicide, for the prevention of HIV infection in women. Science. 2010;329:1168-1174.
3. Marrazzo J, Ramjee G, Nair G, et al. Pre--exposure prophylaxis for HIV in Women: daily oral tenofovir, oral tenofovir/emtricitabine, or vaginal tenofovir gel in the VOICE Study (MTN 003). 20th Conference on Retroviruses and Opportunistic Infections. March 3-6, 2013. Atlanta. Abstract 26LB. http://www.natap.org/2013/CROI/croi_09.htm
------------------------------
Reported by Jules Levin
Topical Application of Tenofovir DF Nanoparticles Prevents HIV-1 Vaginal Transmission in Humanized-BLT Mice......"for the first time, we encapsulated tenofovir disoproxil fumarate (TDF) into nanoparticles (NPs)"
CJ Destache1, AA Date1, Y Zhe2, G Kang2, W Lu2, FY Tso2, C Wood2, and Q Li2.
1Creighton Univ Sch of Pharm, Omaha, NE 2Nebraska Center for Virology and Sch of Biological Sci, Univ of Nebraska-Lincoln, Lincoln, NE


|
| |
|
 |
 |
|
|