 |
 |
 |
| |
Less Bone Loss With DRV/RAL Than DRV/TDF/FTC at 48 and 96 Weeks
|
| |
| |
ICAAC 2014. September 5-9, 2014. Washington, DC
Mark Mascolini
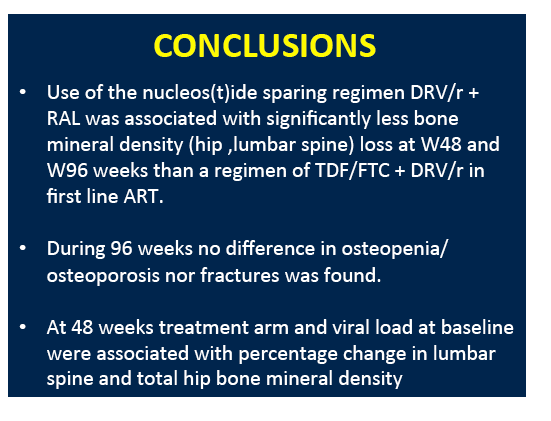
People randomized to darunavir/ritonavir plus raltegravir lost significantly less bone mineral density (BMD) at three sites than people randomized to darunavir/ritonavir plus tenofovir/emtricitabine (TDF/FTC) at 48 and 96 weeks in the NEAT trial [1]. But rates of osteopenia, osteoporosis, and fracture did not differ between the two treatment arms.
NEAT randomized antiretroviral-naive people in 15 European countries to once-daily darunavir/ritonavir plus either twice-daily raltegravir or once-daily TDF/FTC [2]. After 48 weeks, Kaplan-Meier estimated treatment failure rates were 17.8% in the raltegravir arm and 13.8% in the TDF/FTC arm. The difference (4.0%, 95% CI -0·8 to 8·8).established the noninferiority of the raltegravir regimen to the TDF/FTC regimen in this naive population.
In a previous trial comparing the same combinations, RADAR, the 48-week response rate was better with the TDF/FTC combination, largely because of premature discontinuations in the raltegravir arm (20% versus 7%) [3]. Bone changes were significantly worse in the TDF/FTC arm, but kidney outcomes were similar in RADAR. In the single-arm ACTG A5262 trial, virologic failure rates with darunavir/ritonavir plus raltegravir were 16% at week 24 and 26% at week 48 [4].
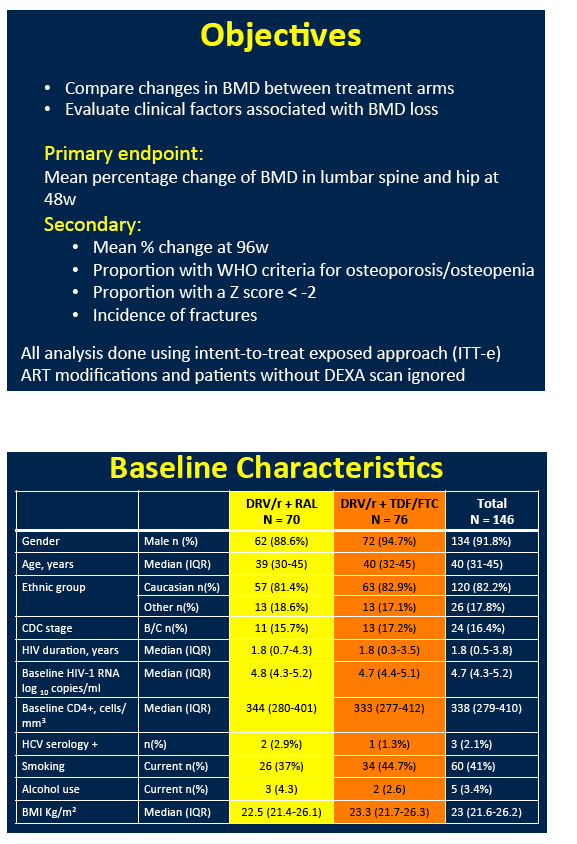
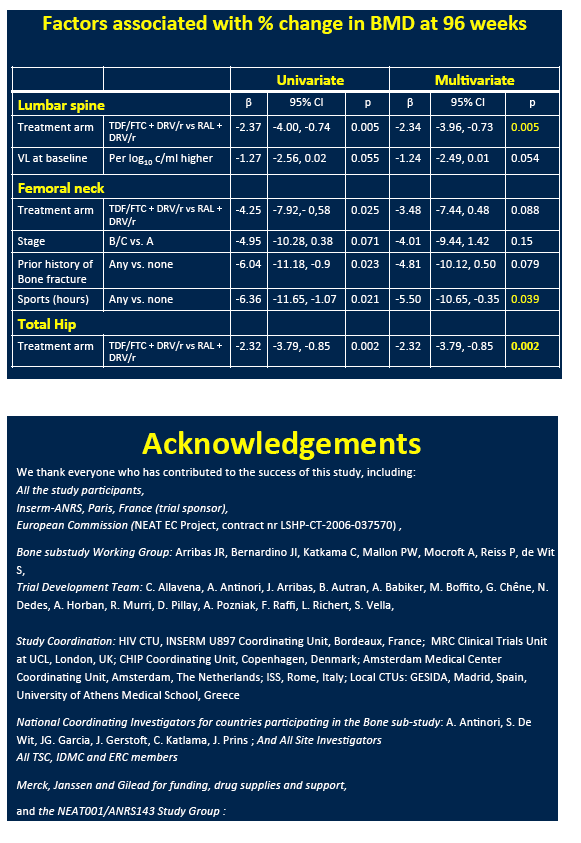
NEAT investigators conducted a substudy of 146 participants to assess DXA-measured BMD changes at the lumbar spine, total hip, and femoral neck through 48 and 96 weeks [1]. BMD declines with TDF are well known. The NEAT team used multivariate linear regression analysis to assess associations between percent BMD change and baseline variables.
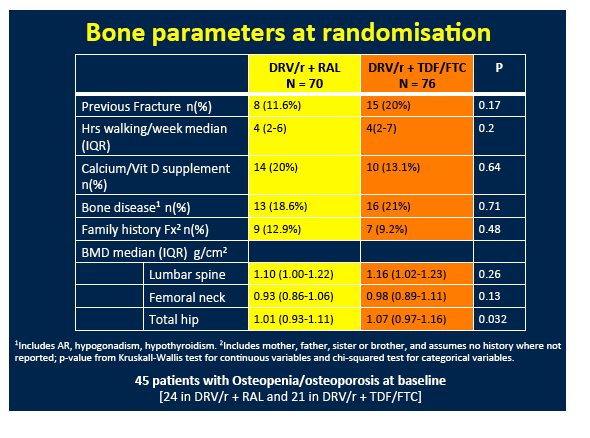
Most substudy participants, 92%, were men, 12% were black, and 41% currently smoked, but only 2% had HCV infection Median age stood at 40 years and median body mass index at 23 kg/m2. Median pretreatment CD4 count measured 338 and median viral load 4.7 log (about 50,000 copies). When entering the trial, 24 of 70 people (34%) randomized to raltegravir and 21 of 76 (28%) randomized to TDF/FTC had osteopenia or osteoporosis.
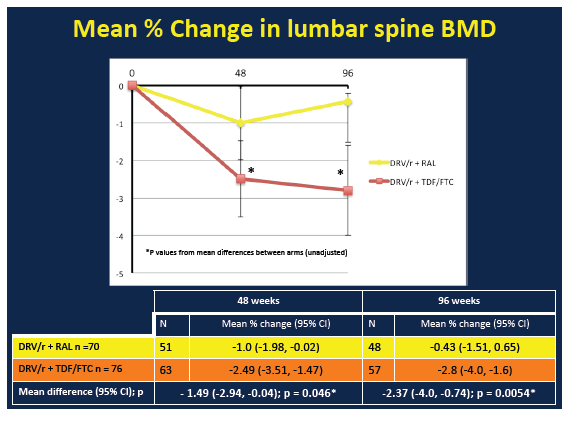
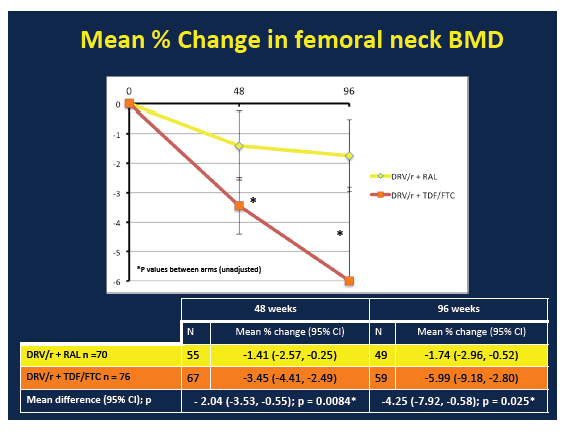
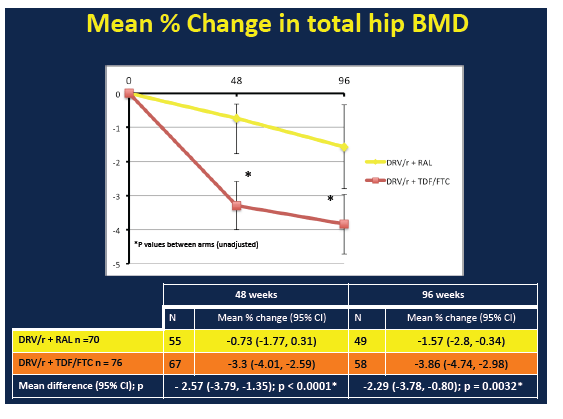
BMD declines through 48 and 96 weeks at all three sites were significantly greater in the TDF/FTC arm than the raltegravir arm:
Week 48 BMD change with raltegravir vs TDF/FTC (95% CI)
Lumbar spine: -1.0 (-1.98 to -0.02) vs -2.49 (-3.51 to -1.47), P = 0.046
Femoral neck: -1.41 (-2.57 to -0.25) vs -3.45 (-4.41 to -2.49), P = 0.0084
Total hip: -0.73 (-1.77 to 0.31) vs -3.30 (-4.01 to -2.59), P < 0.0001
Week 96 BMD change with raltegravir vs TDF/FTC (95% CI)
Lumbar spine: -0.43 (-1.51 to 0.65) vs -2.80 (-4.00 to -1.60), P = 0.0054
Femoral neck: -1.74 (-2.96 to -0.52) vs -5.99 (-9.18 to -2.80), P = 0.025
Total hip: -1.57 (-2.80 to -0.34) vs -3.86 (-4.74 to -2.98), P = 0.0032
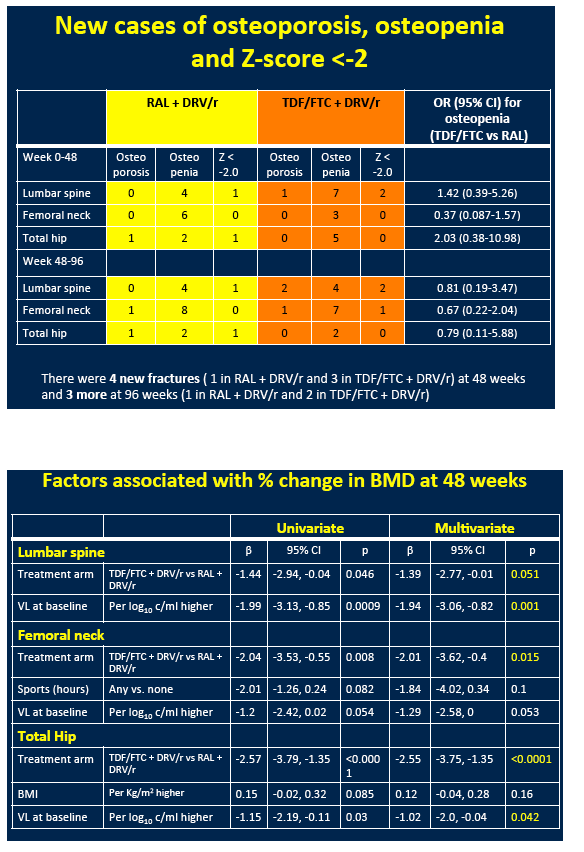
From week 48 to week 96, BMD continued to decline at the total hip but not the other two sites in the raltegravir arm. In the TDF/FTC arm, BMD continued to drop at the femoral neck but not at the other two sites. During the first 48 weeks of the study there were 4 new fractures, 1 in the raltegravir group and 3 in the TDF/FTC group. From week 48 to week 96, 1 more person randomized to raltegravir and 2 more randomized to TDF/FTC broke a bone. These differences were not statistically significant. Nor were changes in new osteopenia or osteoporosis.
Multivariate analysis identified randomization to TDF/FTC as a predictor of greater spine, hip, and femoral neck BMD decline at week 48. Higher pretreatment viral load independently predicted lower 48-week BMD at the total hip and lumbar spine. Randomization to TDF/FTC independently predicted a greater BMD drop at the lumbar spine (beta -3.96, P = 0.005) and total hip (beta -3.79, P = 0.002) at 96 weeks.
References
1. Bernardino JI, Mocroft A, Mallon PW, et al. changes in bone mineral density (BMD) over 96 weeks on darunavir/ritonavir (DRV/r) + raltegravir (RAL) or DRV/r + tenofovir/emtricitabine (TDF/FTC). ICAAC 2014. September 5-9, 2014. Washington, DC. Abstract H-1198.
2. Raffi F, Babiker AG, Richert L, et al. Ritonavir-boosted darunavir combined with raltegravir or tenofovir-emtricitabine in antiretroviral-naive adults infected with HIV-1: 96 week results from the NEAT001/ANRS143 randomised non-inferiority trial. Lancet. 2014; pii S0140-6736(14)61170-3.
3. Bedimo R, Drechsler H, Cutrell J, Jain M, Tebas P, Maalouf N. RADAR study: week 48 safety and efficacy of raltegravir combined with boosted darunavir compared to tenofovir/emtricitabine combined with boosted darunavir in antiretroviral-naive patients: impact on bone health. 7th IAS Conference on HIV Pathogenesis, Treatment and Prevention, June 30-July 3, 2013, Kuala Lumpur. Abstract WEPE512. http://www.natap.org/2013/IAS/IAS_64.htm
4. Taiwo B, Zheng L, Gallien S, et al. Efficacy of a nucleoside-sparing regimen of darunavir/ritonavir plus raltegravir in treatment-naive HIV-1-infected patients (ACTG A5262). AIDS. 2011;25:2113-2122. http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3515052/
--------------------------
CROI: First-Line Raltegravir (RAL) + Darunavir/Ritonavir (DRV/r) is Non-inferior to Tenofovir/Emtricitabine (TDF/FTC) + DRV/r: The NEAT 001/ANRS 143 Randomised Trial - (03/05/14)



|
| |
|
 |
 |
|
|