| |
New Study Reports HIV in Brain Very Early but other studies in the past few years have found HIV gets into the brain very soon after infection....AND studies report very early ART, as soon as possible, within days, is the best way to limit HIV's bad affect on the brain
|
| |
| |
Download the PDF here
New study from Serena Spudich (NIH) was just published in PLOS Pathogens see below following these additional data
at CROI 2013 this study was presented by the same author Serena Spudich
CROI/2013: Very Early Viral Infection of the Central Nervous System without Evidence of Compartmentalization During Acute HIV-1 Infection - (05/27/13)
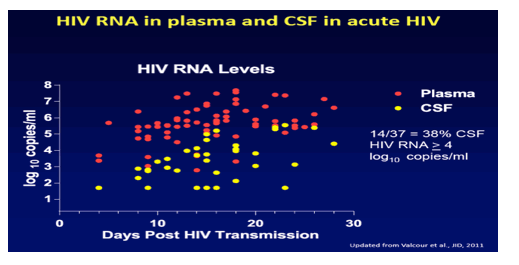
in this study at CROI/2015:
CROI: ART in Acute Infection, Inflammation/Activation "Largely Disappear" - (03/18/15) in this study ART started during Feibig stages 1/2, based on estimate within 10-20 days of infection vs Feibgig 3/4 the next 10 days results in better reservoir HIV reduction - lower HIV DNA in the colon - total DNA in PBMC - greater reduction in cytokines & inflammation - reduced CD8 activation - total HIV DNA in the plasma blood - and less HIV in the brain/CSF
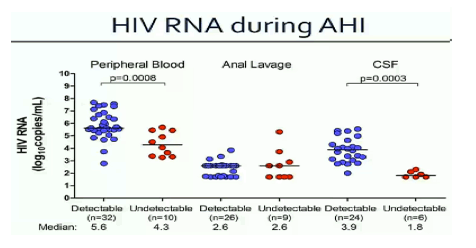
CROI/2015: THE EARLIER cART IS INITIATED DURING PHI, THE MORE INTRACELLULAR HIV-DNA DECREASES - (03/23/15)
at CROI/2011: HIV Affects Brain Immediately After Infection with HIV RNA in the CSF & Inflammation & Brain Injury Continues Unfolding During Chronic Infection & There is a Need for Routine Monitoring - (03/21/11).......within days HIV enters the brain, there is CSF RNA and inflammation is detectable.......at the 2-month timepoint and found damage: "intensive volumetric analysis finds consistent pattern of loss of brain gray matter early in the clinical course of HIV-infection."......brain injury in chronically infected patients. They found: HIV-associated brain injury continues to unfold in the setting of stable HIV disease & treatment. Neuroasympomatic on HAART are at significant risk for converting to subclinical or mild cognitive impairment.
CROI/2014: Brain Markers of Inflammation Drop to Normal With Early ART - (03/05/14)
CROI: Suppressive ART is Key to Reduce Neurocognitive Impairment in Aging HIV+ Individuals - (03/16/15)
CROI: Rates of Non-confounded HIV-Associated Neurocognitive Disorder after Early cART..... "Our findings suggest substantial neuroprotective benefit of initiating cART during primary infection" - (03/03/15)
---------------------------
full text publication below follows NIH press release
Findings add urgency to screening, treatment - NIH-funded study
NIH March 26, 2015 · Press Release
The AIDS virus can genetically evolve and independently replicate in patients' brains early in the illness process, researchers funded by the National Institutes of Health have discovered. An analysis of cerebral spinal fluid (CSF), a window into brain chemical activity, revealed that for a subset of patients HIV had started replicating within the brain within the first four months of infection. CSF in 30 percent of HIV-infected patients tracked showed at least transient signs of inflammation - suggesting an active infectious process - or viral replication within the first two years of infection. There was also evidence that the mutating virus can evolve a genome in the central nervous system that is distinct from that in the periphery.
"These results underscore the importance of early diagnosis and treatment with antiretroviral therapy," said Dianne Rausch, Ph.D., director of the Division of AIDS Research of the NIH's National Institute of Mental Health (NIMH). "Any delay runs the risk that the virus could find refuge and cause damage in the brain, where some medications are less effective - potentially enabling it to re-emerge, even after it is suppressed in the periphery."
NIMH grantees Serena Spudich, M.D., of Yale University, New Haven, Connecticut; Ronald Swanstrom, Ph.D. , of the University of North Carolina, Chapel Hill; Richard Price, M.D. , University of California, San Francisco; and Christa Buckheit Sturdevant, Ph.D., UNC (now at Duke), and colleagues, report on their findings March 26, 2015, in the journal PLoS Pathogens.
Prior to the study, it was known that HIV readily penetrates the brain and can trigger neurological problems and eventually cause dementia over the course of the infection. Yet there was little evidence about how quickly it can take hold and thrive there. Nor was it clear to what extent the brain serves as a hard-to-reach hideout from which the virus might re-infect the body - even if it is eliminated from peripheral blood and lymph node tissue by treatment.
To learn more, the researchers compared evidence of HIV activity in CSF versus blood from 72 untreated HIV-infected patients over the first two years of their infection. Overall, 10-22 percent of the patients showed evidence of HIV replication or inflammation in the brain at the different time points analyzed within the first two years - and the signs persisted over time in about 16 percent of the participants.
The evidence suggests that in most patients peripheral forms of the virus infect immune cells that spread to the brain via blood. Yet in some patients, genetic versions of the virus not found in blood evolve in the brain environment. So it could become an independent, compartmentalized viral reservoir, capable of generating treatment-resistant mutant forms that could break out and re-infect the rest of the body after seemingly successful treatment, explained Rausch.
Whether the potential brain damage caused by early HIV replication and inflammation might be reversible with antiviral therapy awaits further research, said Swanstrom.
HIV, the AIDS virus (yellow) infecting a human immune cell.
Source: Seth Pincus, Elizabeth Fischer and Austin Athman, National Institute of Allergy and Infectious Diseases, National Institutes of Health.
Reference:
Compartmentalized replication of R5 T Cell-Tropic HIV-1 in the central nervous system early in the course of infection. Sturdevant CB, Joseph SB, Schnell G, Price RW, Swanstrom R, Spudich S. PLoS Pathogens, March 26, 2015.
----------------------------
http://journals.plos.org/plospathogens/article?id=10.1371/journal.ppat.1004720
Compartmentalized Replication of R5 T Cell-Tropic HIV-1 in the Central Nervous System Early in the Course of Infection
Abstract
Compartmentalized HIV-1 replication within the central nervous system (CNS) likely provides a foundation for neurocognitive impairment and a potentially important tissue reservoir. The timing of emergence and character of this local CNS replication has not been defined in a population of subjects. We examined the frequency of elevated cerebrospinal fluid (CSF) HIV-1 RNA concentration, the nature of CSF viral populations compared to the blood, and the presence of a cellular inflammatory response (with the potential to bring infected cells into the CNS) using paired CSF and blood samples obtained over the first two years of infection from 72 ART-naïve subjects. Using single genome amplification (SGA) and phylodynamics analysis of full-length env sequences, we compared CSF and blood viral populations in 33 of the 72 subjects. Independent HIV-1 replication in the CNS (compartmentalization) was detected in 20% of sample pairs analyzed by SGA, or 7% of all sample pairs, and was exclusively observed after four months of infection. In subjects with longitudinal sampling, 30% showed evidence of CNS viral replication or pleocytosis/inflammation in at least one time point, and in approximately 16% of subjects we observed evolving CSF/CNS compartmentalized viral replication and/or a marked CSF inflammatory response at multiple time points suggesting an ongoing or recurrent impact of the infection in the CNS. Two subjects had one of two transmitted lineages (or their recombinant) largely sequestered within the CNS shortly after transmission, indicating an additional mechanism for establishing early CNS replication. Transmitted variants were R5 T cell-tropic. Overall, examination of the relationships between CSF viral populations, blood and CSF HIV-1 RNA concentrations, and inflammatory responses suggested four distinct states of viral population dynamics, with associated mechanisms of local viral replication and the early influx of virus into the CNS. This study considerably enhances the generalizability of our results and greatly expands our knowledge of the early interactions of HIV-1 in the CNS.
Author Summary
Early HIV-1 CNS replication likely provides a foundation for brain injury and a potentially important tissue reservoir. To explore the character and timing of emergence of early HIV-1 CNS replication, we examined paired cerebrospinal fluid (CSF) and blood samples from 72 ART-naïve adults, with one-half having longitudinal samples, during the first two years following HIV-1 subtype B infection. In a cross sectional analysis over the first two years of infection, 10-25% of subjects had evidence of either local viral replication in the CNS, defined by the presence of CSF compartmentalization, or a robust inflammatory response, and in approximately 16% of subjects this CNS involvement persisted over time. In some subjects, one of two transmitted viruses replicated predominantly within the CNS, providing insight into how HIV-1 can establish independently replicating populations early in different parts of the body. Based on their entry phenotype, all viruses were selected for replication in CD4+ T cells, although this phenotype was slightly altered in the compartmentalized virus. Overall, we suggest four states to model the nature of HIV-1 CNS infection, which imply distinct mechanisms of virus/host interaction within the CNS during early infection.
Introduction
While HIV-1 can be detected in both the cerebrospinal fluid (CSF) and brain tissue during the weeks after initial exposure [1-7], it is unknown when the virus actually begins replicating independently in the central nervous system (CNS). Independent viral replication within the CNS has two important implications. First, HIV-1 replication can lead to CNS dysfunction and injury, and while combination antiretroviral therapy (cART) has markedly reduced the incidence of HIV-associated dementia (HAD), the prevalence of milder HIV-associated neurological disorders (HAND) has increased [8,9] in the cART era. Second, independent CNS replication may also provide a reservoir distinct from that found in CD4+ T cells in the blood and lymphoid tissue. We do not know the time course of the virologic events that lead to neurological dysfunction and the potential establishment of a CNS reservoir, or the extent to which these long-term outcomes are predicted by the initial aspects of virus-host interaction.
While extensive independent, or compartmentalized, CSF/CNS replication is associated with severe HIV-1 clinical CNS dysfunction [1,10-13], genetically distinct virus can be detected in the CNS throughout the course of infection [4,10]. Thus far, two types of compartmentalization have been defined: one in which a few variants are rapidly expanded giving a CSF viral population of low complexity (clonal amplification) consisting of variants that require high levels of CD4 for entry (R5 T cell-tropic). The second type is characterized by a complex CSF viral population consisting of variants that can enter cells expressing low levels of CD4 (macrophage-tropic), indicative of a more prolonged period of isolated replication and evolution of the entry phenotype.
Additionally, we have recently shown that after vertical transmission to children, CNS compartmentalization can be established via two mechanisms: the early sequestration of one of multiple transmitted variants in the CNS, or the later establishment of compartmentalized CNS virus originating from the periphery [14]. In a previous study, we demonstrated CSF HIV-1 compartmentalization in human subjects during the first year of HIV-1 infection in adults [4]. However, that study only examined viral population sequences from CSF and plasma in a small number of subjects (eight), limiting the generalizability of our findings. Furthermore, the previous study had sparse assessment of longitudinal samples, and included no analysis of sources of compartmentalized HIV-1 within the CSF.
For the current study, we used single genome amplification (SGA) and phylogenetic analysis to assess viral populations in the CSF in the presence and absence of cellular inflammation (i.e. pleocytosis) in cross-sectional and often longitudinal paired blood plasma and CSF samples obtained during the first two years of HIV-infection in ART-naïve subjects. We also extended our analytical approach to include Bayesian Evolutionary Analysis by Sampling Trees (BEAST) to assess time to most recent common ancestor (TMRCA) of CSF and blood HIV-1 populations [15], providing new insights on the timing of establishment of early compartmentalized populations, and we assessed the entry phenotypes of selected clones, further confirming the nature of the transmitted virus as R5 T cell-tropic [16]. Based upon a complex interplay between HIV-1 RNA concentration, viral compartmentalization, and CSF white blood cell (WBC) count, we suggest at least four different patterns to characterize the relationship between virus in the blood and virus in the CSF/CNS during the early period after infection. The current study considerably enhances the generalizability of our results and provides an unprecedented view of the early interactions of HIV-1 in the CNS.
Discussion
Independent HIV-1 replication in the CNS has been associated with neurological disorders [10,40,41] and may represent a distinct reservoir from that found in the blood and lymphoid tissue [42,43]. We examined the virologic characteristics associated with early CNS infection through analysis of paired cross-sectional and longitudinal blood plasma and CSF samples from a large cohort of 72 ART-naïve subjects infected with HIV-1 for less than two years. Our current study significantly builds upon a previous preliminary study by our group [4], enabling us to propose a model with four distinct states to describe the relationship between viral populations in the CSF/CNS and viral populations within the peripheral blood during early HIV-1 infection. These states are based upon details revealed by the current study on mechanisms of establishment of viral compartmentalization within the CNS, relationships between cellular inflammation, HIV-1 RNA levels and phylogenetic state, and insight into longitudinal maintenance and evolution of compartmentalization.
The first state (Fig. 6A) was observed in subjects with little evidence of CNS replication or pleocytosis, with CSF HIV-1 RNA concentrations proportionally 1-2% of the viral load in the periphery (Fig. 2B). In many of these subjects, the CSF HIV-1 RNA level was very low, below the limit of detection of standard assays. Minimal CSF viral burden has been observed in a prior report on a portion of this primary infection cohort [5]. With little or no pleocytosis, HIV-1 is likely entering the CSF/CNS at low levels via incomplete partitioning of virus at the blood-brain barrier, or low level trafficking of immune cells, including small numbers of infected CD4+ T cells. In this circumstance the viral population is very similar to the population in the blood. It is possible that some HIV-1 is replicating independently in the CNS at low levels in these subjects, but we were not able to detect these putative genetic variants above the low level background of virus recently imported from the periphery into the CSF/CNS. An argument in favor of even this low level viremia in the CSF being the result of T cell trafficking is the observation that in neuro-asymptomatic subjects with CD4+ T cells below 50 cells/ul in the blood the viral load in the CSF is on average lower than in subjects with higher CD4+ T cell counts [44,45].
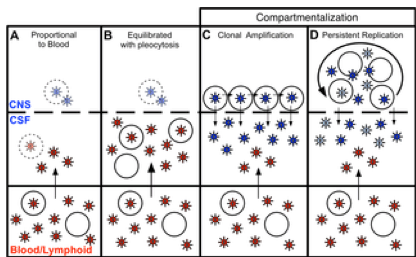
Fig 6. Four states can define the relationship between virus in the CSF/CNS and blood early during infection.
Blood and CSF/CNS compartments are indicated. Blood plasma viral variants are represented by the red virus particles; and CNS viral variants are represented by the blue virus particles. CD4+ T cells are represented by open circles. Arrows indicate direction of virus movement between compartments. (A) State with a CSF HIV-1 RNA level 1-2% of the viral load in the blood and defined by minimal to no local CNS replication or inflammation, resulting in an equilibrated state between the two compartments (when CSF viral load high enough for analysis). Transparent infected CD4+ T cell represents potential local CNS replication that may be obscured by the import of virus into the CNS from the periphery. (B) State of equilibration between CSF and blood accompanied by high levels of pleocytosis, potentially caused by local CNS replication. Transparent infected CD4+ T cell represents potential local CNS replication likely obscured by the high levels of virus secreted by the infiltrating CD4+ T cells. (C) State of CSF/CNS clonal amplification of identical or nearly identical variants within CD4+ T cells. These clonally amplified populations are characterized by low diversity, signified by all CSF viruses in a single shade of blue. (D) State of genetically complex, compartmentalized viral replication within CD4+ T cells in the CSF/CNS indicative of persistent replication beyond a single clonal amplification event; this complexity is signified by CNS viral variants in multiple shades of blue.
doi:10.1371/journal.ppat.1004720.g006
In a second state (Fig. 6B), we observed a relationship between equilibrated viral populations with elevated viral load and high levels of pleocytosis (Fig. 2C). These equilibrated populations were most likely the result of the release of virus from increased numbers of infected CD4+ T cells trafficking from the periphery into the CNS. Though pleocytosis of > 10 cells/ul might have been due to a variety of inflammatory conditions (e.g. neurosyphilis as documented in one individual), it is most likely in response to HIV-1 replication in these PHI subjects. We screened for syphilis in this cohort, and detailed clinical and imaging assessment did not reveal other contributing causes of pleocytosis.
Furthermore, other 'background' non-HIV causes of CSF WBC ≥ 10 cells/ul are unlikely in these subjects, as our parallel studies of 54 HIV-uninfected volunteers recruited from the similar local community demonstrated median CSF WBC counts of 1 cell/ul (IQR 0-2), and none of these 54 subjects had a CSF WBC as high as 10 [5]. In the setting of pleocytosis, while low levels of local CNS replication may have been occurring, the virus imported from the periphery by infected CD4+ T cells dominated the population as it raised the CSF HIV-1 RNA concentration by release of virus imported from the blood. If the inflammatory immune response was successful, pleocytosis might eventually result in low CSF viral loads, a condition observed in a small subset of subjects with pleocytosis but very low levels of virus in the CSF (S1 Table). Pleocytosis was also observed in several subjects with an intermediate viral population phenotype and half of the subjects with compartmentalized viral populations (Fig. 2D), suggesting pleocytosis may result in dynamic changes in the viral population in the CSF. An association between equilibrated compartments and high pleocytosis was also observed in a previous study analyzing four HIV-infected subjects during therapy interruption [46].
In a third state (Fig. 6C), we observed clonally amplified CSF populations of low complexity (Fig. 1 and 4) representing the recent expansion of identical or nearly identical variants that required high levels of CD4 for entry (R5 T cell-tropic; Fig. 5). High levels of pleocytosis were observed in approximately half of the subjects with clonally amplified CSF populations, making it possible that the influx of activated CD4+ T cells may also have provided cellular targets for further transient amplification of a CSF variant. We [47] (Dukhovlinova et al., in preparation) and others [48-50] have observed clonal amplification in the genital tract as well as in the CSF both early [4,14] and at later times in infection [41]. Clonal amplification appears to be a distinct type of virus-host interaction where infection of a population of CD4+ T cells in a compartment is a low probability event and when it occurs there is transient rapid expansion of the viral population. Due to the daily rapid turnover of the CSF viral RNA load, the elevated CSF viral RNA load that is often observed during clonal amplification, and the fact that these clonally amplified lineages generate their own diversity that can persist within the CNS, it is highly unlikely that clonally amplified virus represents virus produced from a single cell. The detection of clonally amplified populations in the CSF within the first year of infection has allowed us to estimate the establishment of these populations within the CNS to within the first 2-6 months (Table 2, Fig. 4), and such amplified populations were detected in 8% of subjects in this study within the first year.
In the final state, we observed more genetically complex compartmentalized viral replication within the CSF/CNS (Fig. 6D) indicative of persistent replication beyond a single clonal amplification event. In an effort to get a more complete view of the interaction of the virus within the CNS at these early times of infection we have interpreted the presence of persistent replication in the CNS based on four criteria: i) sequential clonal amplification events that indicated a permissive CNS environment for viral replication (Fig. 4B); ii) overlapping clonal amplification events that gave rise to compartmentalized recombinants showing continuous replication between the sampled time points (Fig. 4A); iii) intermittent compartmentalization and pleocytosis suggesting an inflammatory immune response to ongoing replication (Fig. 3B); and iv) sequestration of a transmitted variant within the CNS (Fig. 4C). Collectively these markers defined approximately 30% of subjects in the first two years as having evidence of viral replication in the CNS in at least one time point, and 16% having evidence of replication and/or inflammation at multiple time points within this period. This suggests that the CNS compartment is permissive for HIV-1 replication in at least a subset of subjects from a very early period after infection.
Entry tropism analysis revealed that all compartmentalized variants required high levels of CD4 for entry. It is now widely described in the literature that macrophage-tropic variants utilize low levels of CD4 for entry [24-31], are not transmitted, and that the transmitted virus is R5 T cell-tropic [16,27,28,34-39], an understanding further supported by our phenotypic analysis (Fig. 5). Our finding that the viruses involved in this early persistent CNS replication were adapted to replication in CD4+ T cells is distinct from previous studies of individuals with HAD where genetically complex compartmentalized CSF populations that had been replicating as an isolated population had evolved to replicate in macrophages/microglia [41]. Thus, adaptation to use low levels of CD4 for entry, a hallmark of macrophage tropism, is not a feature of the transmitted virus and does not evolve during the early stages of CNS infection in adults, at least as reflected in the compartmentalized virus detected in the CSF. However, we do note that the compartmentalized viruses from the CSF show a small but statistically significant increase in the ability to enter cells with low levels of CD4 compared to CSF virus from equilibrated subjects (Fig. 5B). One explanation for this small difference is that the virus in the CNS is carrying out at least a portion of its replication in a cell with low levels of CD4 which allows for at least a low level of selection for a low CD4 entry phenotype. We found no consistent differences in glycosylation site count or positions, or consistent sequence changes in sites previously described as being associated with macrophage tropism in comparing the viral sequences from the plasma to the compartmentalized sequences in the CSF (S2 Table and S3).
The only CNS tissue that is readily sampled in volunteer human subjects is CSF, which, though not identical to brain, is produced within the brain in the choroid plexus, and reflects brain inflammation and infection in the context of CNS infections including HIV-1. Measures of immune activation, HIV-1 burden, and neural injury detected in CSF are markers of brain involvement in HIV-1 that correlate to clinical and pathologic disease [51]. While the cellular source of HIV-1 RNA detected in the CSF is not certain and may differ during different stages of infection [52], compartmentalization of HIV-1 detected in CSF associates with clinical dementia in humans [10] and immunopathology in the brain in rhesus macaques [53]. Despite limitations of generalizing CSF findings to those of the CNS more broadly, our studies have used the best methods available in living humans to assess HIV-1 populations derived from the CNS in a unique cohort of subjects enrolled during primary HIV-1 infection. Our results show that in cross-sectional analysis over the first two years of HIV-1 infection, 30% of subjects have evidence of either local viral replication in the CNS or a robust CNS inflammatory response, and that in approximately 16% of subjects this CNS involvement can persist over time. We have found that the viral population in the CSF is dynamic as the result of local replication and/or the influx of virus in infected CD4+ T cells as part of an inflammatory response. This early viral replication in a subset of subjects may represent an inability to protect the CNS from infection, potentially leading to HAND later in infection, and may also define a distinct reservoir of infected cells within the body. Longitudinal follow-up of these subjects to examine the long-term impact of the presence of early active HIV-1 replication in the CNS will help to define the significance of these findings for clinical neurologic disease outcomes and compartmentalized viral reservoirs in the setting of HIV-1
|
|
| |
| |
|
|
|