| |
Long-Term Mortality in HIV-Infected Individuals 50 Years or Older: A Nationwide, Population-Based Cohort Study
|
| |
| |
Download the PDF here
JAIDS Feb 1 2016
Legarth, Rebecca A. MD, PhD*; Ahlstrom, Magnus G. MD*; Kronborg, Gitte MD, DMSc ; Larsen, Carsten S. MD, DMSc ; Pedersen, Court MD, DMSc ; Pedersen, Gitte MD, PhD ; Mohey, Rajesh MD, PhD ; Gerstoft, Jan MD, DMSc*; Obel, Niels MD, DMSc*
*Department of Infectious Diseases, Copenhagen University Hospital, Rigshospitalet, Copenhagen, Denmark; Department of Infectious Diseases, Copenhagen University Hospital, Hvidovre, Denmark; Department of Infectious Diseases, Aarhus University Hospital, Aarhus, Denmark; Department of Infectious Diseases, Odense University Hospital, Odense, Denmark; Department of Infectious Diseases, Aalborg University Hospital, Aalborg, Denmark; and Department of Internal Medicine, The Regional Hospital West Jutland, Herning, Denmark.
"Among HIV-infected individuals without comorbidity the estimated median survival time from age 50 years was 25.6 years (to age 75.6 years) (95% CI: 23.8 to NA) compared with 34.2 years (age 84.2 years) (95% CI: 29.6 to 38.3) among population controls without comorbidity. MRR was 1.7 (95% CI: 1.2 to 2.3) for HIV-infected individuals compared with population controls.
Ageing of the HIV-population emphasizes challenges in HIV-treatment, as HIV-infected individuals ≥50 years more often faces complicating factors such as higher prevalence of comorbidity, more advanced HIV-infection at time of diagnosis and polypharmacy.
In our study the proportion of HIV-infected individuals ≥50 years with CCI ≥ 1 at study inclusion was higher than among the age-matched population controls. Lohse et al19 has previously shown that comorbidity achieved before HIV-diagnosis substantially influences mortality following HIV-diagnosis, and that HIV-infection and comorbidity interact synergistically with the risk of death among HIV-infected individuals. In our study we included a well-treated subpopulation of HIV-infected individuals ≥50 years with undetectable HIV RNA and CD4 cell count ≥350 cells per microliter after 1 year of cART. We detected a 1.6-fold increased risk of death of among HIV-infected individuals ≥ 50 years without comorbidity compared with population controls without comorbidity, which is in line with the findings of Lohse et al who found a 1.7-fold increased mortality in HIV-infected individuals with CCI = 0 compared with population controls with CCI = 0.
HIV-infected individuals and population controls were categorized with comorbidity at study inclusion if they: Had Charlson Comorbidity Index (CCI) ≥1 point, were diagnosed with hepatitis C virus- (HCV) in DNRP or positive HCV antibody test or a positive HCV RNA test in DHCS, diagnosed with alcohol- or drug-related abuse in DNPR, or were registered as intravenous drug abusers in DHCS. CCI was calculated at date of study inclusion based on recordings in DNRP, and includes 19 specific diseases: myocardial infarction, congestive heart failure, peripheral vascular disease, cerebrovascular disease, dementia, chronic pulmonary disease, connective tissue disease, ulcer disease, mild liver disease, diabetes, hemiplegia, moderate to severe renal disease, diabetes with end-organ failure, any tumor, leukemia, lymphoma, moderate to severe liver disease, metastatic solid tumor. AIDS was not included. CCI was calculated by methods described previously.19
HIV-infected individuals who were observed during 1996-1999 had increased mortality within all age-strata compared with HIV-infected individuals observed during 2006-2014: MRR 1.5 (95% CI: 1.0 to 2.4) for 50-55 years, 1.8 (95% CI: 1.0 to 3.1) for 55-60 years, 2.5 (95% CI: 1.3 to 4.7) for 60-65 years, 1.2 (95% CI: 0.5 to 3.0) for 65-70 years, 1.2 (95% CI: 0.3 to 5.5) for 70-75 years, and 9.5 (95% CI: 1.7 to 53.7) for 75-80 years. For HIV-infected individuals observed during 2000-2005 MRRs were 1.5 (95% CI: 1.0 to 2.2) for 50-55 years, 1.2 (95% CI: 0.8 to 1.8) for 55-60 years, 1.3 (95% CI: 0.8 to 2.0) for 60-65 years, 1.6 (95% CI: 0.9 to 2.8) for 65-70 years, 1.9 (95% CI: 0.0 to 3.9) for 70-75 years, and 1.8 (95% CI: 0.5 to 6.3) for 75-80 years compared with HIV-infected individuals observed during 2006-2014.
Our results indicate that further incentives to reduce mortality among HIV-infected individuals ≥50 years are needed."
Abstract
Background: Although the prevalence of HIV-infection among individuals ≥50 years of age has increased, the impact of HIV-infection on risk of death in this population remains to be established. Our aim was to estimate long-term mortality among HIV-infected individuals who were 50 years or older, when compared with an individually-matched cohort from the background population.
Methods: Population-based cohort-study including HIV-infected individuals ≥50 years, who were alive 1 year after HIV-diagnosis (n = 2440) and a comparison cohort individually-matched by age and gender extracted from the background population (n = 14,588). Cumulative survival was evaluated using Kaplan-Meier method and Mortality Rate Ratios (MRRs) were estimated using Cox Regression Models. Study period 1996-2014.
Results: Estimated median survival time from age 50 years for HIV-infected individuals increased from 11.8 years (95% CI: 10.2 to 14.5) during 1996-1999 to 22.8 years (20.0-24.2) in 2006-2014. MRR decreased with increasing age from 3.8 (3.1-4.7) for 50-55 years to 1.6 (1.0-2.6) for 75-80 years. In a cohort of well-treated HIV-infected individuals ≥50 years without AIDS-defining events or comorbidity at study inclusion (n = 517). MRR was 1.7 (1.2-2.3) compared with population controls without comorbidity.
Conclusion: Among HIV-infected individuals estimated median survival time from age 50 years has increased by more than 10 years from 1996-1999 to 2006-2014, but is still substantially lower than in the background population. Even among well-treated HIV-infected individuals ≥50 years without comorbidity or AIDS-defining events the estimated median survival time remains lower than in the general population.
INTRODUCTION
In high-income countries approximately 30%-50% of HIV-infected individuals are 50 years or older, and the prevalence of HIV-infection among individuals older than 50 years of age is increasing regardless of geographical regions.1,2 Ageing of the HIV-population is the result of the tremendous decline in mortality after the introduction of combination antiretroviral therapy (cART) in the mid-1990,3 and an increased proportion of newly HIV-infected individuals diagnosed after 50 years of age.4-6 HIV-infection in older individuals is often associated with several complicating factors such as higher prevalence of comorbidity, higher risk of late HIV-diagnosis, increased mortality6-12 and reduced CD4-cell response to cART.13-15 With the increased prevalence of HIV-infections among older individuals it has become important to determine, whether the effect of cART among older HIV-infected individuals are reflected by mortality rates approaching those in the background population.
We used a nationwide, population-based cohort of HIV-infected individuals 50 years or older and an individually-matched comparison cohort from the background population to estimate all-cause mortality stratified by age-intervals and calendar period. We further compared mortality among well-treated HIV-infected individuals ≥50 years without comorbidity with an individually-matched comparison cohort from the background population also without comorbidity.
METHODS
Setting
The study period was from 1 January 1996 until 31 May 2014. The estimated HIV prevalence in Denmark is approximately 0.1% in the adult population, and HIV-treatment is centralized to 8 specialized HIV-centers where HIV-infected individuals are seen on an outpatient basis at intended intervals of 12-24 weeks. The Danish Healthcare system provides free tax-supported HIV-treatment including cART to all HIV-infected individuals that are residents in Denmark.
Data Sources
The Danish HIV Cohort Study (DHCS)
DHCS is a population-based, nationwide cohort study including all HIV-infected individuals treated at Danish HIV centers since 1 January 1995. Individuals are consecutively enrolled at first contact with an HIV center. Collected data includes demographics, date of HIV diagnosis, route of transmission, AIDS-defining events, and antiretroviral treatment. CD4 cell counts and HIV RNA measurements are extracted electronically from laboratory data files. Data are updated annually.16
We used the unique 10-digit civil registration number assigned to all Danish residents to link the following registries:
Civil Registration System (CRS)
CRS includes information on vital status, date of death, and migration for all Danish residents. Data is updated continuously.17
The Danish National Registry of Patients (DNRP)
DNRP includes information on all patients discharged from Danish hospitals. Records for each in-patient admission include dates of admission and discharge and diagnoses, categorized according to the International Classification of Diseases (ICD-8/ICD-10).18
Study Populations
All HIV-1 infected individuals who reached an age of 50 years between 1 January 1996 and 31 May 2014 were included in study. Study inclusion was one year following HIV-diagnosis, one year following immigration, or date of 50th birthday, whichever came last. For each HIV-infected individual 6 population controls individually matched on age and gender were identified from CRS to constitute a background population cohort. All population controls were living in Denmark at time of study inclusion of their corresponding HIV-infected individual. Population controls were assigned same date of study inclusion as their corresponding HIV-infected individual.
Further, from the study population we identified a cohort of well-treated HIV-infected individuals observed during 2006-2014 who: (1) had received at least 1 year of cART, (2) had HIV RNA < 500 copies per milliliter and CD4 cell count ≥350 cells per microliter following 1 year of cART, (3) had no AIDS-defining events at study inclusion or after 1 year of cART, and (4) had no comorbidity (as defined below) after 1 year of cART. Study inclusion was 1 year after start of cART.
For each well-treated HIV-infected individual 6 population controls individually matched on age and gender were identified in CRS. Population controls had to be living in Denmark at time of study inclusion of their corresponding HIV-infected individual, and be without any record of comorbidity at the time of study inclusion. Population controls were assigned the same date of study inclusion as their corresponding HIV-infected individual.
Description and Categorization of Exposure Variables, Outcomes, and Covariates
Primary outcome was time to death from all causes. Date of death was extracted from CRS.
HIV-infected individuals and population controls were categorized with comorbidity at study inclusion if they: Had Charlson Comorbidity Index (CCI) ≥1 point, were diagnosed with hepatitis C virus- (HCV) in DNRP or positive HCV antibody test or a positive HCV RNA test in DHCS, diagnosed with alcohol- or drug-related abuse in DNPR, or were registered as intravenous drug abusers in DHCS. CCI was calculated at date of study inclusion based on recordings in DNRP, and includes 19 specific diseases: myocardial infarction, congestive heart failure, peripheral vascular disease, cerebrovascular disease, dementia, chronic pulmonary disease, connective tissue disease, ulcer disease, mild liver disease, diabetes, hemiplegia, moderate to severe renal disease, diabetes with end-organ failure, any tumor, leukemia, lymphoma, moderate to severe liver disease, metastatic solid tumor. AIDS was not included. CCI was calculated by methods described previously.19
cART was defined as a treatment regimen of at least 3 antiretroviral drugs or a regimen including a nonnucleoside reverse-transcriptase inhibitor, an integrase inhibitor or a protease inhibitor. HIV RNA below <500 copies per milliliter was categorized as undetectable viral load.
Statistics
Observation time was calculated from study inclusion until death, emigration, lost to follow-up or 31 May 2014.
Cumulative survival was evaluated with Kaplan-Meier methods using age as time scale. Analyses were performed for 3 observation periods: 1996-1999 (early period of cART), 2000-2005 and 2006-2014 (later periods of cART). HIV-infected individuals could provide observation time in different observation periods depending on length of follow-up time. In analyses among well-treated HIV-infected individuals without comorbidity or AIDS-defining events observation time was estimated from January 1, 2006, date of 50th birthday or date of HIV diagnosis, which ever came last. As no substantial differences in survival was observed between calendar periods among population controls ≥50 years, survival curves for population controls were collapsed into one curve depicting 1996-2014. All-cause mortality rates (MR) per 1000 Person Years at Risk (PYR), excess mortality rates (EMR) per 1000 PYR and mortality rate ratios (MRR) for 5-years age intervals for HIV-infected individuals and population controls were calculated for the complete study period and the calendar periods: 1996-1999, 2000-2005, and 2006-2014. MRRs were estimated using Cox Proportional Hazards Model. The proportional-hazards assumption was tested using nonzero slope.
Data analysis was performed using SPSS statistical software, version 19.0 (Norusis; SPSS Inc., Chicago, IL), and Stata software, version 11.0 (STATA Corp., College Station, TX).
RESULTS
We identified 2440 HIV-infected individuals, who fulfilled the inclusion criteria and who were followed for a total of 16,637 PYR (Table 1). Among HIV-infected individuals 530 (21.7%) died, 50 (2.1%) emigrated, and 4 (0.2%) were registered as lost to follow up. We identified 14,588 population controls that were followed for a total of 115,817 PYR. Among population controls 1388 (9.5%) died, 185 (1.3%) emigrated, and 8 (0.1%) were recorded lost to follow up. Further baseline characteristic are summarized in Table 1.
All-cause Mortality Among HIV-Infected Individuals Compared With Population Controls Aged 50 Years or Older
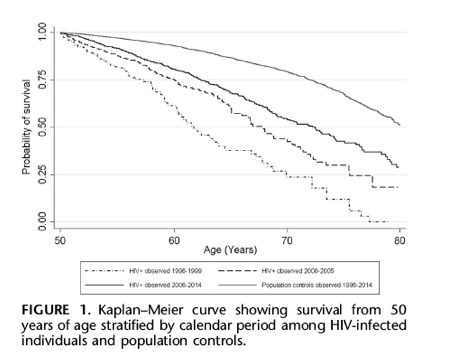
Figure 1 shows Kaplan-Meier survival curves from 50 years of age for HIV-infected individuals and population controls stratified by calendar period of 1996-1999, 2000-2005, and 2006-2014.
Among HIV-infected individuals estimated median survival time increased during the study period; 11.8 years (95% CI: 10.2 to 14.5) in 1996-1999, 17.8 years (95% CI: 15.1 to 20.5) in 2000-2005, and 22.5 years (95% CI: 20.0 to 24.2) in 2006-2014. For population controls estimated median survival time was 30.2 years (95% CI: 29.5 to 31.1) in 1996-2014.
MR, EMR, and MRR in 5-year age-intervals for HIV-infected individuals and population controls are summarized in Table 2.
The overall-MRR decreased with increasing age; from 3.8 (95% CI: 3.1 to 4.7) for 50-55 years to 1.6 (95% CI: 1.0 to 2.6) for 75-80 years of age (Table 2). The overall-EMR increased with increasing age; from 16.3 (95% CI: 12.6 to 19.9) for 50-55 years to 48.8 (95% CI: 7.2 to 90.4) for 75-80 years (Table 2). Highest EMR was observed during the period 1996-1999, and remained lower within all age-intervals in the 2 subsequent periods (see Table S1, Supplemental Digital Content, http://links.lww.com/QAI/A739). In line with this, MRRs were highest in the early period 1996-1999, and remained lower in all age-intervals in the 2 subsequent periods (see Table S1, Supplemental Digital Content, http://links.lww.com/QAI/A739).
HIV-infected individuals who were observed during 1996-1999 had increased mortality within all age-strata compared with HIV-infected individuals observed during 2006-2014: MRR 1.5 (95% CI: 1.0 to 2.4) for 50-55 years, 1.8 (95% CI: 1.0 to 3.1) for 55-60 years, 2.5 (95% CI: 1.3 to 4.7) for 60-65 years, 1.2 (95% CI: 0.5 to 3.0) for 65-70 years, 1.2 (95% CI: 0.3 to 5.5) for 70-75 years, and 9.5 (95% CI: 1.7 to 53.7) for 75-80 years. For HIV-infected individuals observed during 2000-2005 MRRs were 1.5 (95% CI: 1.0 to 2.2) for 50-55 years, 1.2 (95% CI: 0.8 to 1.8) for 55-60 years, 1.3 (95% CI: 0.8 to 2.0) for 60-65 years, 1.6 (95% CI: 0.9 to 2.8) for 65-70 years, 1.9 (95% CI: 0.0 to 3.9) for 70-75 years, and 1.8 (95% CI: 0.5 to 6.3) for 75-80 years compared with HIV-infected individuals observed during 2006-2014.
To determine whether age at time of HIV diagnosis influenced risk of death, we compared mortality in patients diagnosed before or after 50 years for all calendar periods, and observed no substantial difference: MRR 1.0 (95% CI: 0.5 to 1.9) for 1996-1999, 1.2 (95% CI: 0.8 to 1.8) for 2000-2005 and 1.0 (95% CI: 0.8 to 1.4) for 2006-2014 (Figure 3).
Mortality Among Well-treated HIV-infected Individuals >50 Years Observed During 2006-2014
We included 517 well-treated HIV-infected individuals without AIDS-defining events or comorbidity at time of study inclusion. Median age at HIV diagnosis vas 45 (IQR: 39-52) and 170 (32.9%) were older than 50 years of age when diagnosed. Further baseline characteristics for the cohort of well-treated HIV-infected individuals are summarized in Table 1. In addition, 3192 individually-matched population controls without comorbidity were included.
Figure 2 shows Kaplan-Meier survival curves for HIV-infected individuals ≥50 years without comorbidity and population controls ≥50 years without comorbidity for the period 2006-2014.
Among HIV-infected individuals without comorbidity the estimated median survival time from age 50 years was 25.6 years (to age 75.6 years) (95% CI: 23.8 to NA) compared with 34.2 years (age 84.2 years) (95% CI: 29.6 to 38.3) among population controls without comorbidity. MRR was 1.7 (95% CI: 1.2 to 2.3) for HIV-infected individuals compared with population controls.
|
|
| |
| |
|
|
|