| |
Will a new tenofovir prodrug for the treatment of
HIV reduce the risk of nephrotoxicity?
|
| |
| |
Download the PDF here
Kidney Intl Jan 2016
Christina Wyatt
1Division of Nephrology, Department of Medicine, Icahn School of Medicine at Mount Sinai, New York, New York, USA
The widely used antiretroviral agent tenofovir disoproxil fumarate has been associated with proximal tubular injury and decreased glomerular filtration rate in HIV-infected individuals. Phase 3 trials of a new prodrug, tenofovir alafenamide, suggest a lower potential for kidney injury.
The nucleotide reverse transcriptase inhibitor tenofovir disoproxil fumarate (TDF) is one of the most widely used antiretroviral agents worldwide; it is also approved for the treatment of chronic hepatitis B virus infection and as a component of pre- and post-exposure prophylaxis to prevent HIV infection. Although generally well tolerated, TDF causes severe proximal tubulopathy and acute kidney injury in a minority of treated individuals. TDF has also been associated with decreased glomerular filtration rate (GFR) and loss of bone mineral density.1, 2, 3 A new prodrug of tenofovir, tenofovir alafenamide (TAF), was developed with the goal of reducing kidney and bone toxicity. TAF has a longer half-life in plasma, allowing for increased uptake of tenofovir into immune cells at lower doses and lower plasma concentrations than TDF. Because tenofovir is cleared by a combination of glomerular filtration and active tubular secretion, the lower plasma concentrations are anticipated to result in lower intracellular accumulation of tenofovir in proximal tubular epithelial cells, the primary target of TDF nephrotoxicity.
Applications for regulatory approval were filed with the US Food and Drug Administration (FDA) and the European Commission based on the results of 2 phase 3 studies comparing TAF versus TDF, each coformulated into a once-daily fixed-dose combination pill with elvitegravir, cobicistat, and emtricitabine.4 In addition to demonstrating non-inferiority of the TAF-containing regimen for initial treatment of HIV, the combined study results were notable for a smaller decline in Cockcroft-Gault creatinine clearance (CrCl) in participants assigned to TAF (Figure 1). Randomization to TAF was also associated with smaller increases in proteinuria, albuminuria, and the urine biomarkers retinol-binding protein (RBP) and β2-microglobulin. The fixed-dose combination including TAF received FDA approval for marketing in November 2015; 2 other TAF-containing regimens are currently under review.
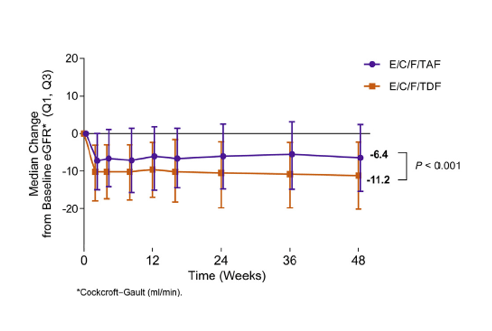
Figure 1 | Median change in Cockcroft-Gault creatinine clearance (CrCl) from baseline in participants
randomized to tenofovir alafenamide (TAF) versus tenofovir disoproxil fumarate (TDF), each in fixeddose
combination with elvitegravir, cobicistat, and emtricitabine (E/C/F). The initial decline in CrCl noted
in both arms between 0 and 12 weeks is consistent with the effect of cobicistat on the tubular secretion of
creatinine. The small but statistically significant difference in CrCl decline noted between the arms may suggest
a more favorable safety profile for TAF versus TDF. (Reprinted with permission from Sax PE, Wohl D, Yin MT, et
al. Tenofovir alafenamide versus tenofovir disoproxil fumarate, coformulated with elvitegravir, cobicistat, and
emtricitabine, for initial treatment of HIV-1 infection: two randomised, doubleblind, phase 3, non-inferiority
trials. Lancet. 2015;385:2606-2615,4 Supplementary Figure 2a.)
Because the trials were of limited duration and were not powered to demonstrate a difference in clinical outcomes such as acute kidney injury or chronic kidney disease, only postmarketing experience will determine the clinical relevance of the small but statistically significant differences observed between TAF and TDF. Postmarketing surveillance of renal safety will be complicated by the coadministration of TAF with medications known to interfere with the tubular secretion of creatinine, including the pharmacologic enhancers cobicistat and ritonavir and the antiretroviral agents dolutegravir and rilpivirine.5 Each of these agents has been associated with a rapid and sustained increase in serum creatinine that is independent of measured GFR, confounding the interpretation of creatine-based estimates. Cystatin C has been suggested as an alternative to serum creatinine in this setting, but is less accurate as a marker of GFR when used alone in HIV-infected individuals.6
Another important unanswered question is whether any safety benefits of TAF would extend to individuals with reduced GFR. Individuals with CrCl <50 ml/min were excluded from the primary trials, consistent with dosing guidelines for the TDF-containing comparator. Based on the results of a small single-dose pharmacokinetic study, TAF does not appear to require dose reduction in adults with CrCl 30 ml/min; exposure to tenofovir as measured by area under the curve in adults with CrCl 15 to 30 ml/min was similar to that previously observed in healthy volunteers following the administration of full-dose TDF. The fixed-dose combination of TAF with elvitegravir, cobicistat, and emtricitabine is FDA-approved for use in adults with CrCl >30 ml/min, making it the first single-pill antiretroviral therapy regimen recommended for patients with CrCl <50 ml/min.
An ongoing open-label study is evaluating the safety of this regimen in adults with CrCl 30 to 69 ml/min.7 Preliminary results demonstrated no significant change in CrCl or measured GFR, but a rapid and sustained reduction in proteinuria, albuminuria, RBP, and β2-microglobulin in participants switching from a TDF-containing regimen. The safety of TAF in patients with a prior history of TDF nephrotoxicity will be the subject of future studies.
|
|
| |
| |
|
|
|