| |
ASM/ICAAC: Pharmacokinetics-Pharmacodynamics of Emtricitabine/Tenofovir Alafenamide Demonstrated Wide Exposure Range Associated With Clinical Safety
|
| |
| |
Reported by Jules Levin
ASM/ICAAC 2016 June 16-20 Boston, MA
Joseph M. Custodio, Lillian S. Ting, Julia Z. Zack, Sophia R. Majeed, Mingjin Yan, Lijie Zhong, Martin S. Rhee
Gilead Sciences, Inc., Foster City, CA


*Study drugs taken without regard to food; †FTC/TAF dose: 200/10 mg with boosted protease inhibitors (PIs) and 200/25 mg with unboosted 3rd agents; ‡FTC/TAF 200/10 mg: ATV+RTV (n=53), DRV+RTV (n=84), LPV/r (n=18); FTC/TAF 200/25 mg: DTG (n=26), NVP (n=74), RAL (n=66), EFV (n=8), RPV (n=3), MVC (n=1); §FTC/TDF 200/300 mg: ATV+RTV (n=50), DRV+RTV (n=82), LPV/r (n=18); FTC/TAF 200/25 mg: DTG (n=23), NVP (n=66), RAL (n=73), EFV (n=6), RPV (n=6), MVC (n=6). BL, baseline; eGFR, estimated glomerular filtration rate.
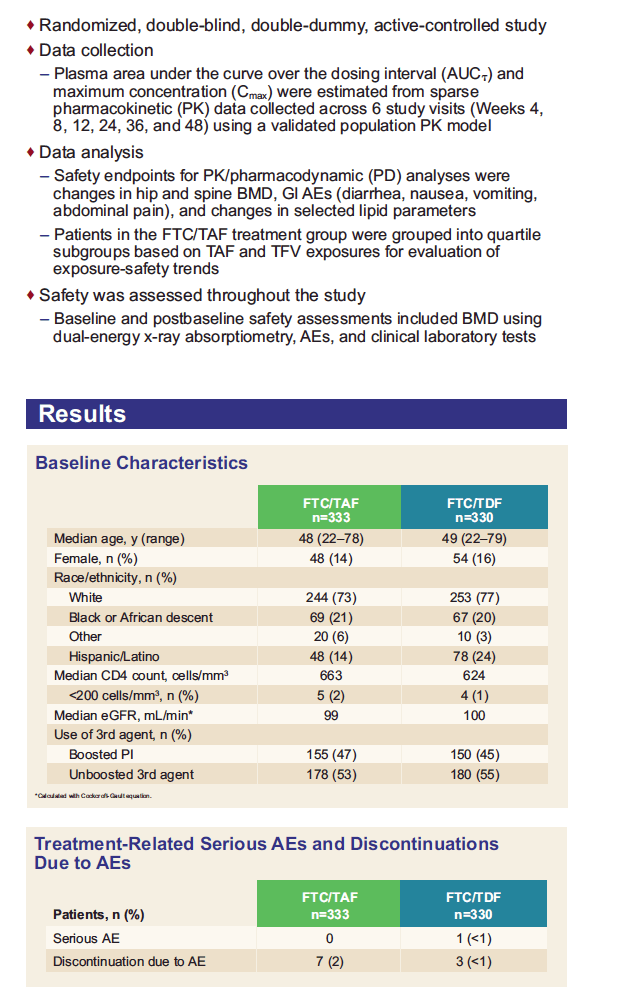
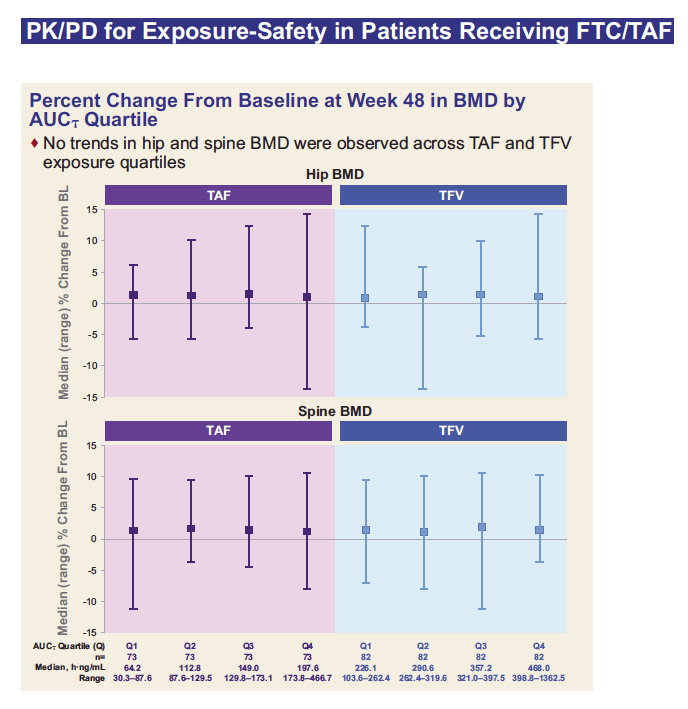
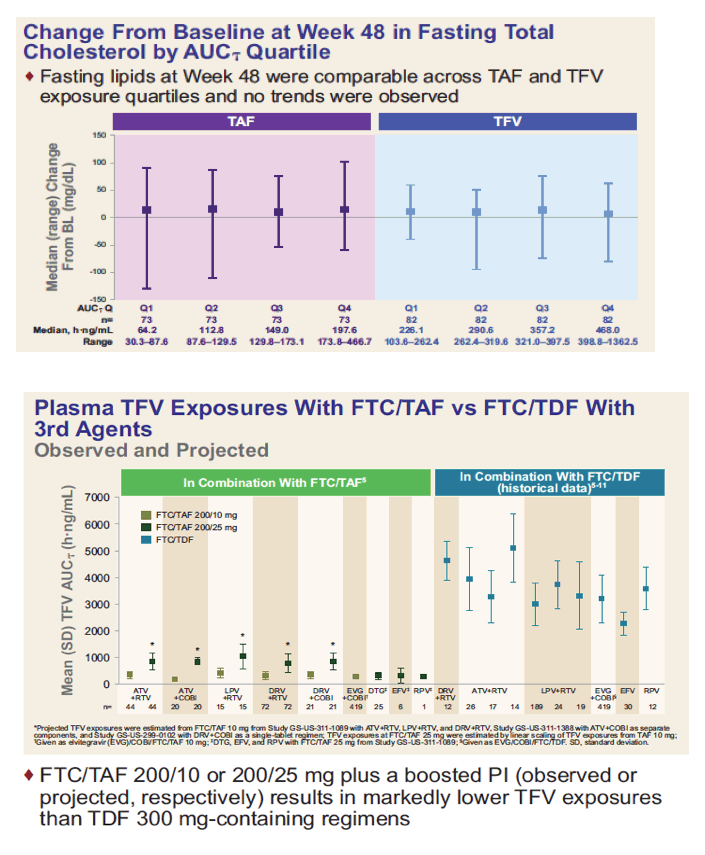
*Projected TFV exposures were estimated from FTC/TAF 10 mg from Study GS-US-311-1089 with ATV+RTV, LPV+RTV, and DRV+RTV, Study GS-US-311-1388 with ATV+COBI as separate components, and Study GS-US-299-0102 with DRV+COBI as a single-tablet regimen; TFV exposures at FTC/TAF 25 mg were estimated by linear scaling of TFV exposures from TAF 10 mg;
†Given as elvitegravir (EVG)/COBI/FTC/TAF 10 mg; ‡DTG, EFV, and RPV with FTC/TAF 25 mg from Study GS-US-311-1089; §Given as EVG/COBI/FTC/TDF. SD, standard deviation.

|
|
| |
| |
|
|
|